BAY 80-6946 (Copanlisib)
Several phosphatidylinositol-3-kinase (PI3K) inhibitors are being investigated as a treatment for patients with B-cell malignancies. Such agents prevent activation of PI3K enzymes that are hyperactive in many B-cell malignancies and associated with tumor progression. Copanlisib is a novel pan-Class I phosphatidylinositol-3-kinase (PI3K) inhibitor with potent preclinical inhibitory activity against both PI3K-d and PI3K-α isoforms.
In vitro: BAY 80-6946 is a phosphoinositide 3-kinase (PI3K) inhibitor with potential antineoplastic activity, which inhibits proliferation with IC50 of 147 nM in HuCCT-1 (KRASG12D ) and 137 nM in EGI-1 (KRASG12D ) cell lines [1].
In vivo: BAY 80-6946 is generally well tolerated through the maximum tolerated dose (MTD) of 0.8 mg/kg. pharmacokinetics (PK) results support dosing weekly. Grade 2 or 3 hyperglycemia in the first 24 hrs after receiving a MTD dose. Pharmacokinetics, clinical SD as well as FDG-PET data are consistent with effective exposure and PI3K pathway inhibition. [2].
Clinical trial: Copanlisib (BAY 80-6946), developed by Bayer, is a selective Class I phosphoinositide 3-kinase inhibitor which has shown promise in Phase I/II clinical trials for the treatment of non-Hodgkin lymphoma and chronic lymphocytic leukemia. Phase II study shows that Copanlisib is active as a single-agent in heavily pretreated, advanced refractory/relapsed FL, MZL, , CLL and SLL. Copanlisib exhibited an acceptable toxicity profile, which was consistent with previous findings (https://ash.confex.com/ash/2014/webprogram/Paper70672.html).
References:
[1] Patnaik A, et al. J Clin Oncol, 29, 2011, (suppl, abstr 3035)
[2] Andrea H, et al. Cancer Res, 2012; 72(8), (suppl, Abstract 869)
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 480.52 |
| Cas No. | 1032568-63-0 |
| Formula | C23H28N8O4 |
| Solubility | insoluble in DMSO; insoluble in H2O; insoluble in EtOH |
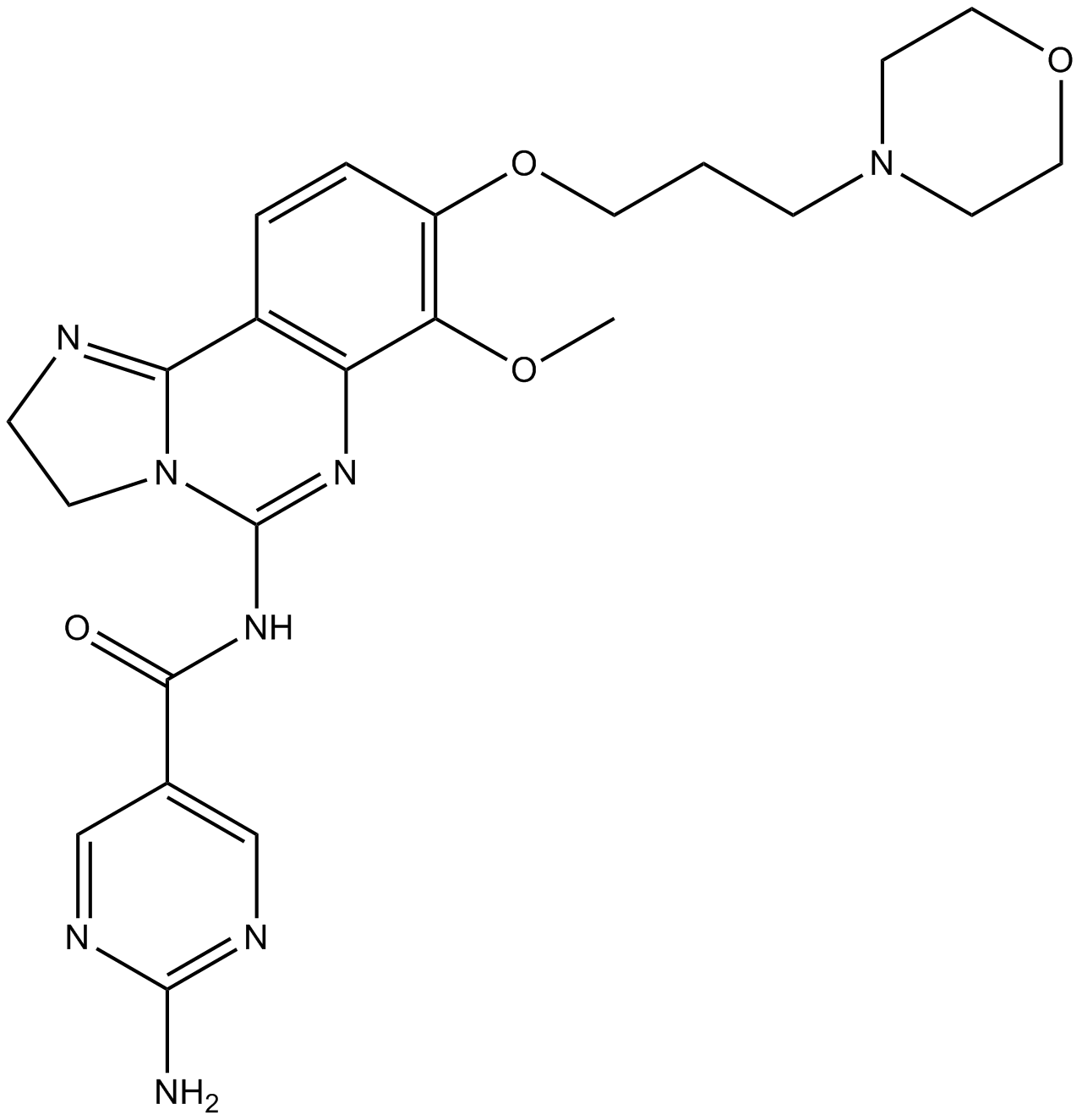
| Chemical Name | 2-amino-N-[7-methoxy-8-(3-morpholin-4-ylpropoxy)-2,3-dihydroimidazo[1,2-c]quinazolin-5-yl]pyrimidine-5-carboxamide |
| SDF | Download SDF |
| Canonical SMILES | COC1=C(C=CC2=C1N=C(N3C2=NCC3)NC(=O)C4=CN=C(N=C4)N)OCCCN5CCOCC5 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Biochemical lipid kinase assays |
The effect of BAY 80-6946 on PI3Kα, PI3Kβ and PI3Kγ activity was measured by the inhibition of 33P incorporation into phosphatidylinositol (PI) in 384-well MaxiSorp plates coated with 2 μg/well of PI and phosphatidylserine (PS) (1:1 molar ratio). In each PI3K isoform assay, 9 μL of reaction buffer (50 mM MOPSO, pH 7.0, 100 mM NaCl, 4 mM MgCl2, 0.1% BSA) containing 7.5 ng of His-tagged N-terminal truncated p110α or p110β protein or 25 ng of purified human p110γ protein was used. The reaction was started by adding 5 μL of 40 μM ATP solution containing 20 μCi/mL [γ-33P]-ATP. After 2-hr incubation at room temperature, the reaction was terminated by addition of 5 μL of 25 mM EDTA solution. The plates were washed and Ultima Gold scintillation cocktail (25 μL) was then added. The radioactivity incorporated into the immobilized PI substrate was determined with a BetaPlate Liquid Scintillation Counter. |
| Cell experiment [1]: | |
|
Cell lines |
A panel of cancer cell lines |
|
Preparation method |
The solubility of this compound in DMSO is limited. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
5 μM; 72 hrs |
|
Applications |
BAY 80-6946 showed significant anti-proliferative activity in a series of cancers cells exhibiting constitutively activated PI3K signaling. Several breast cancer, endometrial cancer and hematologic tumor cell lines were extremely sensitive to BAY 80-6946 (IC50 values < 10 nM). |
| Animal experiment [1]: | |
|
Animal models |
A rat KPL4 tumor xenograft model |
|
Dosage form |
0.5 ~ 6 mg/kg; i.v.; every 2 days for a total of 5 doses starting on day 14 after tumor cell implantation |
|
Applications |
On day 25 (i.e. 3 days after the last dose), BAY 80-6946 at doses of 0.5, 1, 3 and 6 mg/kg showed TGI rates of 77%, 84%, 99% and 100%, respectively. In addition, BAY 80-6946 at doses of 3 and 6 mg/kg resulted in complete tumor regression. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Liu N, Rowley BR, Bull CO, Schneider C, Haegebarth A, Schatz CA, Fracasso PR, Wilkie DP, Hentemann M, Wilhelm SM, Scott WJ, Mumberg D, Ziegelbauer K. BAY 80-6946 is a highly selective intravenous PI3K inhibitor with potent p110α and p110δ activities in tumor cell lines and xenograft models. Mol Cancer Ther. 2013 Nov;12(11):2319-30. |
|
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data