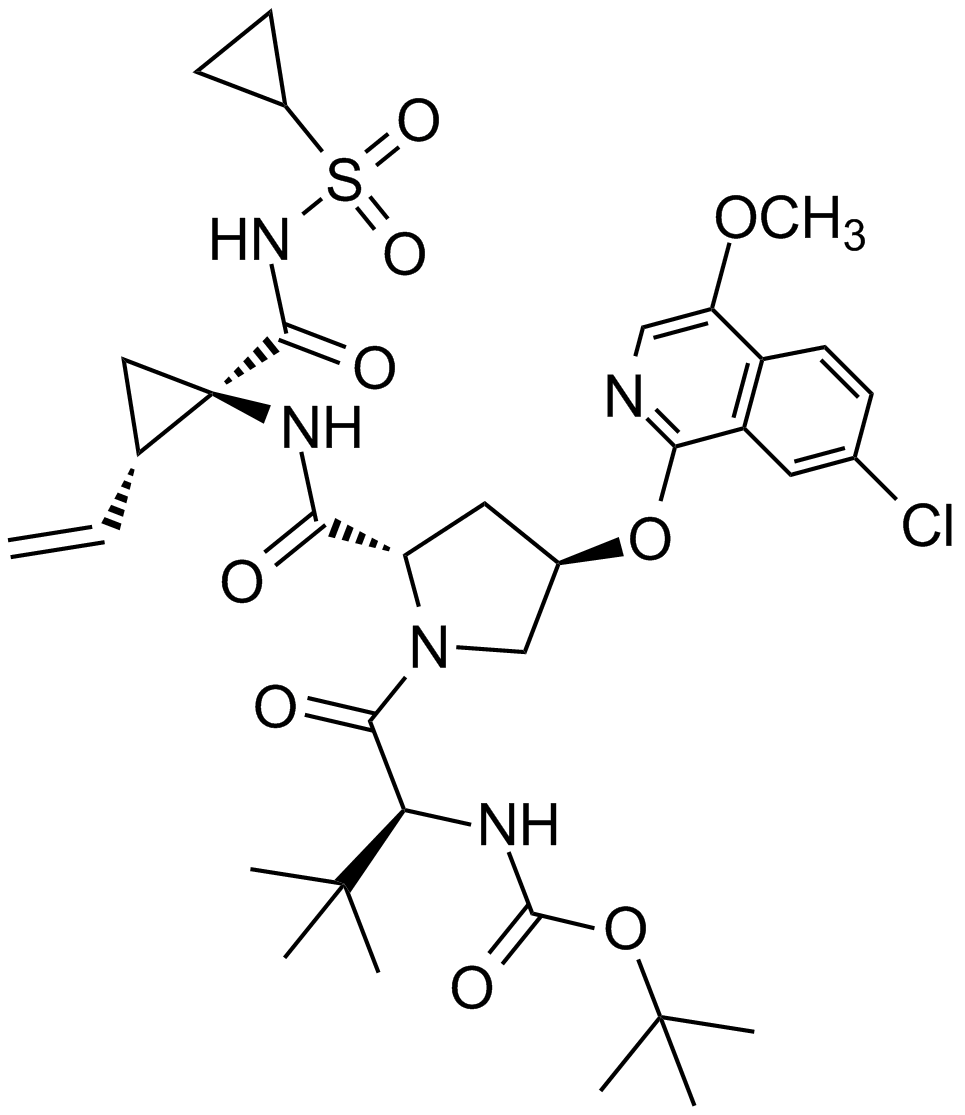
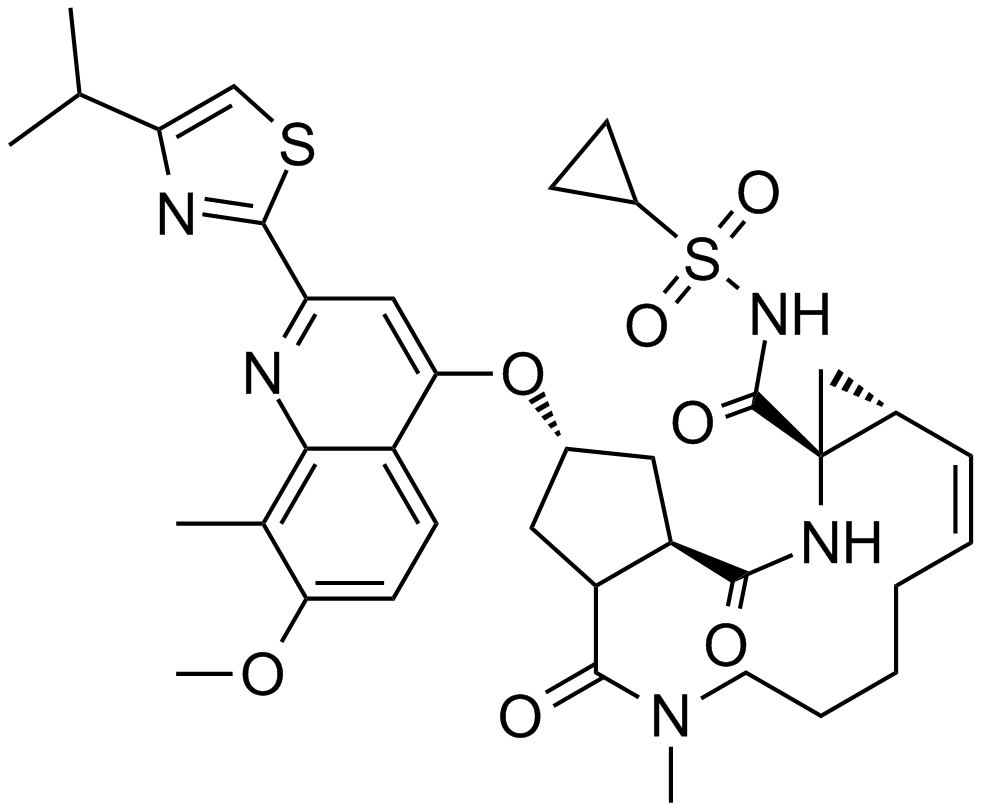
Asunaprevir (BMS-650032)
Asunaprevir is an orally efficacious inhibitor of NS3 protease with IC50 value of 1nM [1].
Asunaprevir is an inhibitor of hepatitis C virus (HCV) NS3 protease. It can inhibit 6 major genotypes of HCV NS3/4A protease with IC50 values of 0.7nM, 0.3nM, 15nM, 78nM, 320nM, 1.6nM, 1.7nM and 0.9nM, respectively for genotype 1a, 1b, 2a, 2b, 3a, 4a, 5a and 6a, respectively. When using the purified recombinant full-length HCV NS3/4A protease complexes, asunaprevir shows the Ki values of 0.4nM and 0.2nM, respectively for genotype 1a and genotype 1b. The mechanism of the inhibition is that the acylsulfonamide of asunaprevir interacts with the catalytic site of NS3 protease in a noncovalent manner. Asunaprevir inhibits HCV RNA replication in different cell lines, including liver, T lymphocytes, lung, cervix, and embryonic kidney. It shows no obvious activity against other RNA viruses. The permeability of asunaprevir is similar to the compound with good absorption in humans. The tests of metabolism rate show that asunaprevir exhibits low to intermediate metabolic clearance. Plasma and tissue exposures in vivo indicate that asunaprevir displays a hepatotropic disposition [2].
References:
[1] Scola PM, Sun LQ, Wang AX, Chen J, Sin N, Venables BL, Sit SY, Chen Y, Cocuzza A, Bilder DM, D'Andrea SV, Zheng B, Hewawasam P, Tu Y, Friborg J, Falk P, Hernandez D, Levine S, Chen C, Yu F, Sheaffer AK, Zhai G, Barry D, Knipe JO, Han YH, Schartman R, Donoso M, Mosure K, Sinz MW, Zvyaga T, Good AC, Rajamani R, Kish K, Tredup J, Klei HE, Gao Q, Mueller L, Colonno RJ, Grasela DM, Adams SP, Loy J, Levesque PC, Sun H, Shi H, Sun L, Warner W, Li D, Zhu J, Meanwell NA, McPhee F. The discovery of asunaprevir (BMS-650032), an orally efficacious NS3 protease inhibitor for the treatment of hepatitis C virus infection. J Med Chem. 2014 Mar 13;57(5):1730-52.
[2] McPhee F, Sheaffer AK, Friborg J, Hernandez D, Falk P, Zhai G, Levine S, Chaniewski S, Yu F, Barry D, Chen C, Lee MS, Mosure K, Sun LQ, Sinz M, Meanwell NA, Colonno RJ, Knipe J, Scola P. Preclinical Profile and Characterization of the Hepatitis C Virus NS3 Protease Inhibitor Asunaprevir (BMS-650032). Antimicrob Agents Chemother. 2012 Oct;56(10):5387-96.
- 1. Jason D Vevea, Edwin R Chapman. "Acute disruption of the synaptic vesicle membrane protein synaptotagmin 1 using knockoff in mouse hippocampal neurons." Elife. 2020;9:e56469. PMID: 32515733
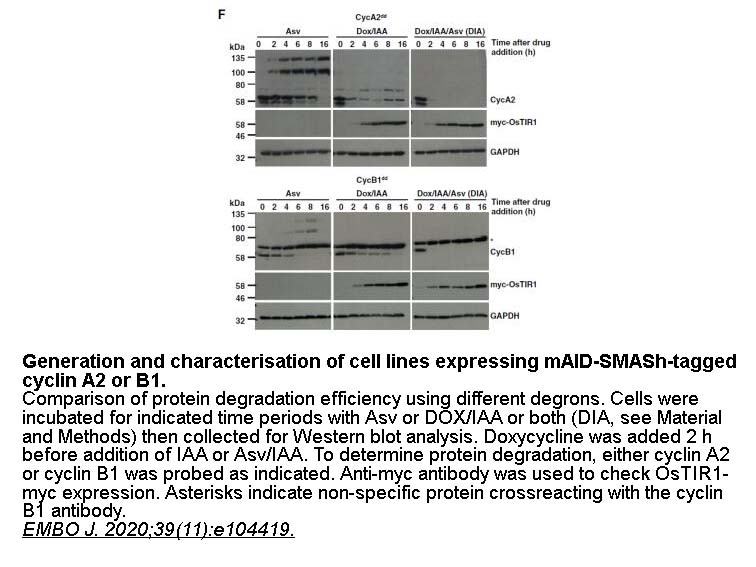
- 2. Hégarat N, Crncec A, et al. "Cyclin A triggers Mitosis either via the Greatwall kinase pathway or Cyclin B." EMBO J. 2020;39(11):e104419. PMID: 32350921
- 3. Foight GW, Wang Z, et al. "Multi-input chemical control of protein dimerization for programming graded cellular responses." Nat Biotechnol. 2019 Sep 9. PMID: 31501561
- 4. Juillerat A, Tkach D, et al. "Modulation of chimeric antigen receptor surface expression by a small molecule switch." BMC Biotechnol. 2019 Jul 3;19(1):44. PMID: 31269942
- 5. Rago F, DiMare MT, et al. "Degron mediated BRM/SMARCA2 depletion uncovers novel combination partners for treatment of BRG1/SMARCA4-mutant cancers." Biochem Biophys Res Commun. 2019 Jan 1;508(1):109-116. PMID: 30527810
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 748.29 |
| Cas No. | 630420-16-5 |
| Formula | C35H46ClN5O9S |
| Synonyms | BMS-650032;BMS 650032;BMS650032,Asunaprevir |
| Solubility | ≥37.41 mg/mL in DMSO; insoluble in H2O; ≥48.6 mg/mL in EtOH |
| Chemical Name | tert-butyl N-[(2S)-1-[(2S,4R)-4-(7-chloro-4-methoxyisoquinolin-1-yl)oxy-2-[[(1R,2S)-1-(cyclopropylsulfonylcarbamoyl)-2-ethenylcyclopropyl]carbamoyl]pyrrolidin-1-yl]-3,3-dimethyl-1-oxobutan-2-yl]carbamate |
| SDF | Download SDF |
| Canonical SMILES | CC(C)(C)C(C(=O)N1CC(CC1C(=O)NC2(CC2C=C)C(=O)NS(=O)(=O)C3CC3)OC4=NC=C(C5=C4C=C(C=C5)Cl)OC)NC(=O)OC(C)(C)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Hepatitis C virus (HCV) NS3/4A protease genotype assays |
Published methods for the construction, expression, and purification of recombinant full-length NS3/4A complexes representing HCV genotypes 1a (H77c) and 1b (J4L6S) were used to generate homogeneous full-length NS3/4A protease complexes representing the six major HCV genotypes (HC-J6, HC-J8, S52, ED43, SA13 and HK-6A). The susceptibility of purified recombinant NS3/4A protease complexes was assessed using fluorescence resonance energy transfer (FRET) assays. The IC50 value was calculated. |
| Cell experiment [1]: | |
|
Cell lines |
HuH-7, MRC5, MT-2, HepG2, HeLa and HEK293 cells |
|
Preparation method |
Soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reaction Conditions |
4 days |
|
Applications |
Asunaprevir inhibited HCV RNA replication in different cell lines, including liver, T lymphocytes, lung, cervix, and embryonic kidney. It showed no obvious activity against other RNA viruses. |
| Animal experiment [2]: | |
|
Animal models |
Rats |
|
Dosage form |
10 μM; p.o.; 60 mins |
|
Applications |
After oral dosing to the rat, Asunaprevir demonstrated modest oral bioavailability and a plasma AUC of 1.0 μM·h. However, at the 24th hrs after p.o. dosing, the liver levels of Asunaprevir were high at 15.2 μM, suggesting a hepatotropic distribution in vivo. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. McPhee F, Sheaffer AK, Friborg J, Hernandez D, Falk P, Zhai G, Levine S, Chaniewski S, Yu F, Barry D, Chen C, Lee MS, Mosure K, Sun LQ, Sinz M, Meanwell NA, Colonno RJ, Knipe J, Scola P. Preclinical Profile and Characterization of the Hepatitis C Virus NS3 Protease Inhibitor Asunaprevir (BMS-650032). Antimicrob Agents Chemother. 2012 Oct;56(10):5387-96. [2]. Scola PM, Sun LQ, Wang AX, Chen J, Sin N, Venables BL, Sit SY, Chen Y, Cocuzza A, Bilder DM, D'Andrea SV, Zheng B, Hewawasam P, Tu Y, Friborg J, Falk P, Hernandez D, Levine S, Chen C, Yu F, Sheaffer AK, Zhai G, Barry D, Knipe JO, Han YH, Schartman R, Donoso M, Mosure K, Sinz MW, Zvyaga T, Good AC, Rajamani R, Kish K, Tredup J, Klei HE, Gao Q, Mueller L, Colonno RJ, Grasela DM, Adams SP, Loy J, Levesque PC, Sun H, Shi H, Sun L, Warner W, Li D, Zhu J, Meanwell NA, McPhee F. The discovery of asunaprevir (BMS-650032), an orally efficacious NS3 protease inhibitor for the treatment of hepatitis C virus infection. J Med Chem. 2014 Mar 13;57(5):1730-52. |
|
Quality Control & MSDS
- View current batch:
Chemical structure

Related Biological Data

Related Biological Data

Related Biological Data

Related Biological Data
Related Biological Data