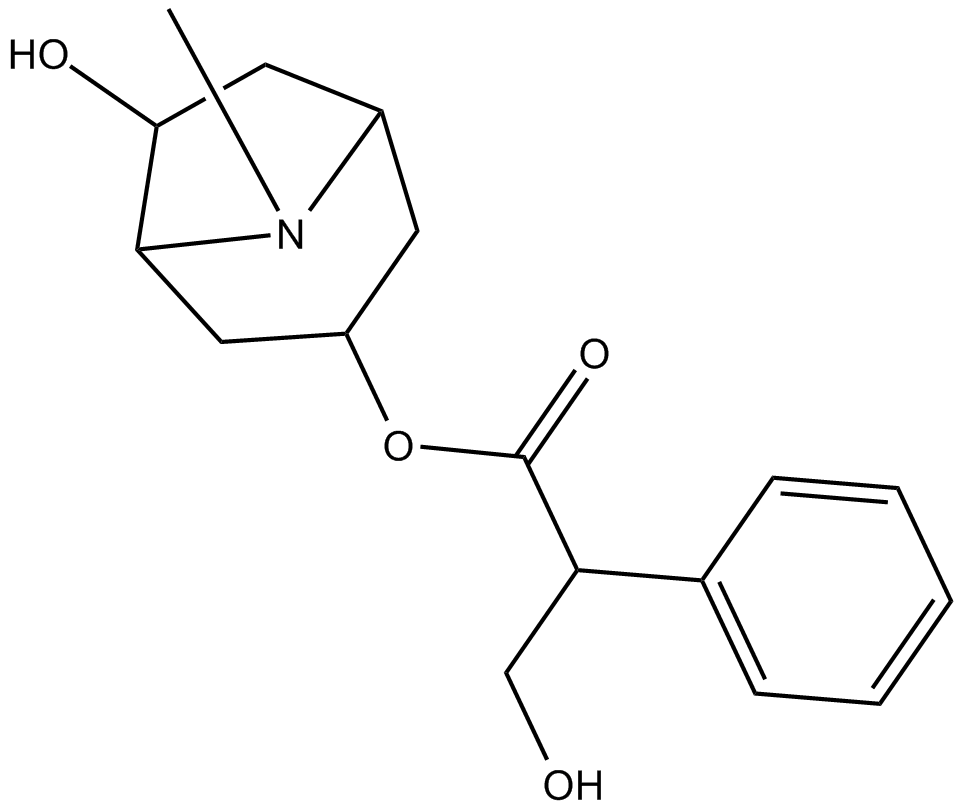
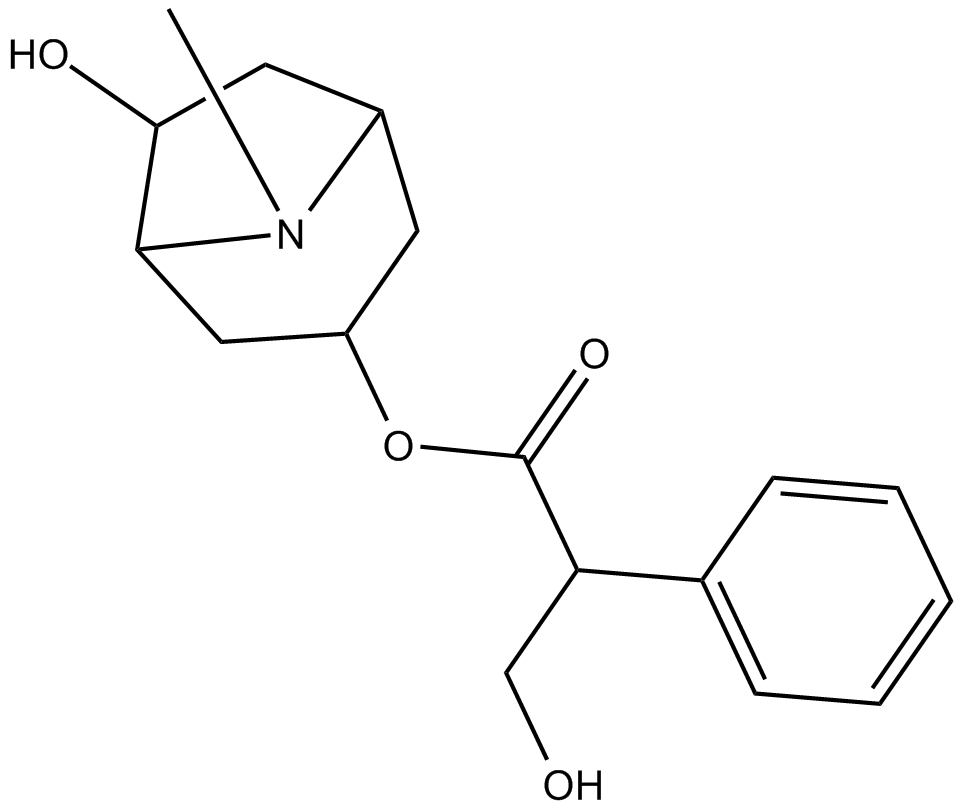
Raceanisodamine
Raceanisodamine is a weak antagonist of α1-adrenergic receptors [1].
The adrenergic receptors play an important role in modulating sympathetic nervous system activity as well as a site of action for many therapeutic agents. The α1-adrenergic receptors contain three types, termed as α1A-, α1B-, and α1D. The α1-adrenergic receptor plays an essential role in smooth muscle, growth, neurological, and cardiovascular function [2]. With the binding of the endogenous ligands, epinephrine and norepinephrine, the adrenergic receptors are the prime mediators of smooth muscle contraction and hypertrophic growth [2].
Raceanisodamine was a natural tropane alkaloid predominantly found in the roots of A. tanguticus. Raceanisodamine weakly antagonized the activity of α1-adrenoceptors and blocked WB-4101 and clonidine binding in brain membrane preparations with pKi values of 2.63 and 1.61, respectively [1]. Raceanisodamine showed antioxidant effects and protected against free radical-induced cellular damage [3].
References:
[1] Varma D R, Yue T L. Adrenoceptor blocking properties of atropine‐like agents anisodamine and anisodine on brain and cardiovascular tissues of rats[J]. British journal of pharmacology, 1986, 87(3): 587-594.
[2] Piascik M T, Perez D M. α1-Adrenergic receptors: new insights and directions[J]. Journal of Pharmacology and Experimental Therapeutics, 2001, 298(2): 403-410.
[3] Poupko J M, Baskin S I, Moore E. The pharmacological properties of anisodamine[J]. Journal of Applied Toxicology, 2007, 27(2): 116-121.
| Storage | Store at -20°C |
| M.Wt | 305.37 |
| Cas No. | 17659-49-3 |
| Formula | C17H23NO4 |
| Synonyms | 6-hydroxy Hyoscyamine |
| Solubility | ≥13.32 mg/mL in EtOH; ≥13.65 mg/mL in H2O; ≥51.3 mg/mL in DMSO |
| Chemical Name | 6-hydroxy-8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3-hydroxy-2-phenylpropanoate |
| SDF | Download SDF |
| Canonical SMILES | CN(C(C1)CC(C2)OC(C(CO)c3ccccc3)=O)C2C1O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure