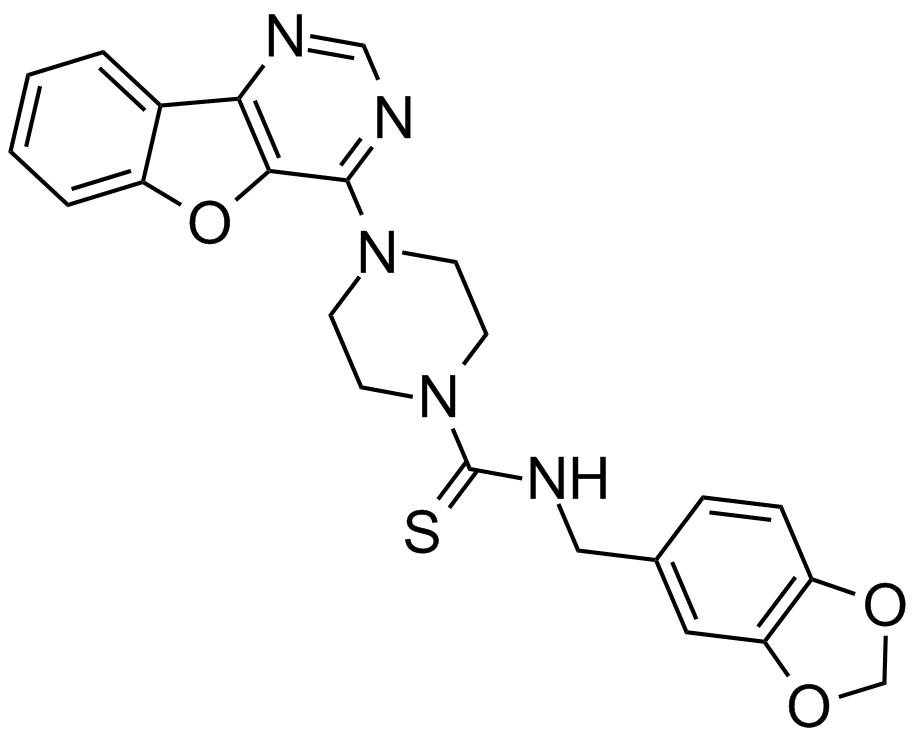
Amuvatinib (MP-470, HPK 56)
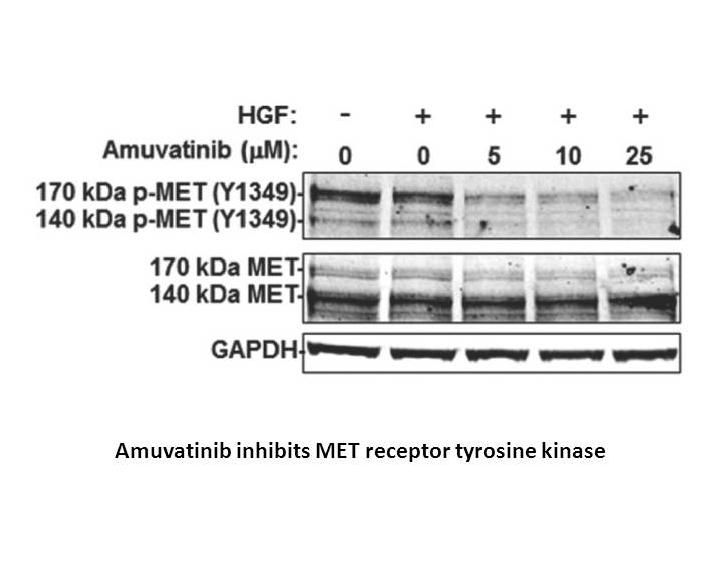
Amuvatinib (also known as MP-470 or HPK 56), a synthetic carbothioamide, is a novel and potent tyrosine kinase inhibitor, which exerts in vitro and in vivo activities against multiple targets including mutant KIT, platelet-derived growth factor receptor alpha (PDGFRα) and DNA repair. Μechanisticly, amuvatinib inhibits tyrosine kinase receptor KIT through occupying its ATP binding domain (IC50 < 0.1 μM) and disrupts DNA repair through suppression of homologous recombination protein Rad51 as well as synergistic effects in combination with double stranded DNA damaging agents. Recent studies have shown that amuvatinib exhibits antitumor activity against human cancer cell lines, especially GIST-48 human cell line, in which it strongly inhibits proliferation (IC50 = 0.20 μM).
Reference
Raoul Tibes, Gil Fine, Gavin Choy, Sanjeev Redkar, Pietro Taverna, Aram Oganesian, Amarpao Sahai, Mohammad Azab and Anthony W. Tolcher. A phase I, first-in-human dose-escalation study of amuvatinib, a multi-targeted tyrosine kinase inhibitor, in patients with advanced solid tumors. Cancer Chemother Pharmacol (2013) 71: 463-471
Gavin Choy, Rajashree Joshi-Hangal, Aram Oganesian, Gil Fine, Scott Rasmussen, Joanne Collier, James Kissling, Amarpal Sahai, Mohammad Azab and Sanjeev Redkar. Saftety, tolerability, and pharmacokinetics of amuvatinib from three phase 1 clinical studies in healthy volunteers. Cancer Chemother Pharmacol (2012) 70: 183-190
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 447.51 |
| Cas No. | 850879-09-3 |
| Formula | C23H21N5O3S |
| Synonyms | MP470,MP 470,HPK56 |
| Solubility | ≥22.4 mg/mL in DMSO; insoluble in H2O; insoluble in EtOH |
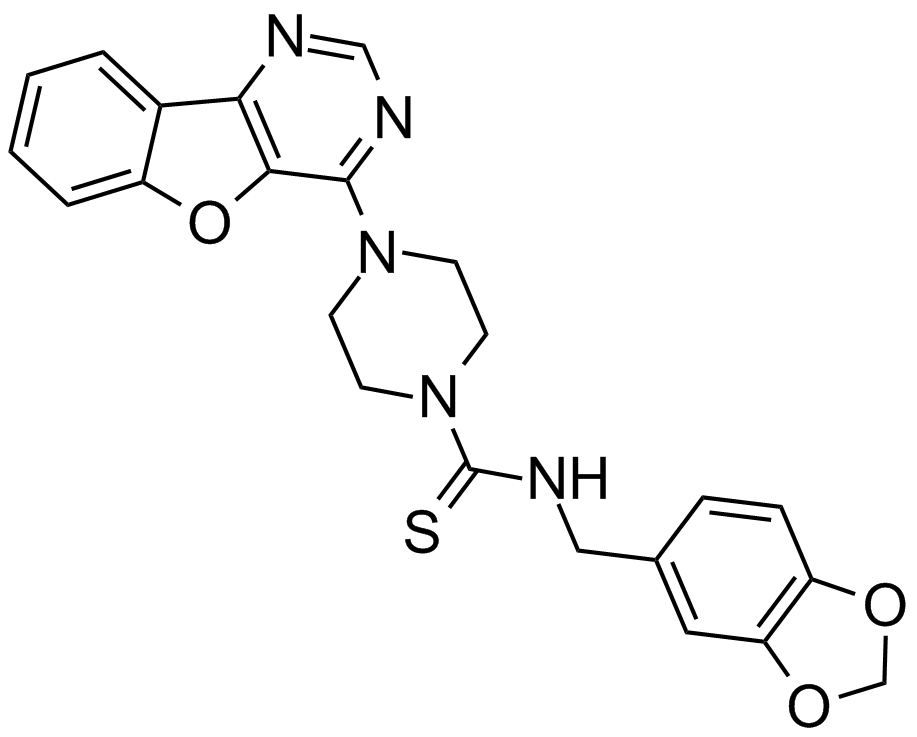
| Chemical Name | N-(1,3-benzodioxol-5-ylmethyl)-4-([1]benzofuro[3,2-d]pyrimidin-4-yl)piperazine-1-carbothioamide |
| SDF | Download SDF |
| Canonical SMILES | S=C(NCc(cc1)cc2c1OCO2)N(CC1)CCN1c1ncnc2c1[o]c1ccccc21 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Description | Amuvatinib (MP-470) is a potent and multi-targeted inhibitor of c-Kit, PDGFRα and Flt3 with IC50 of 10 nM, 40 nM and 81 nM, respectively. | |||||
| Targets | c-KitD816H | PDGFRαV561D | Flt3D835Y | |||
| IC50 | 10 nM | 40 nM | 81 nM | |||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
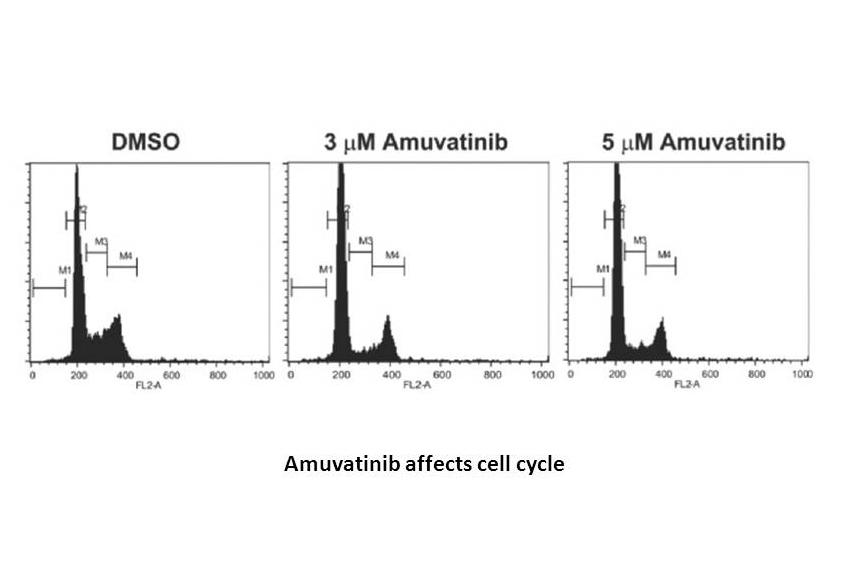
Related Biological Data