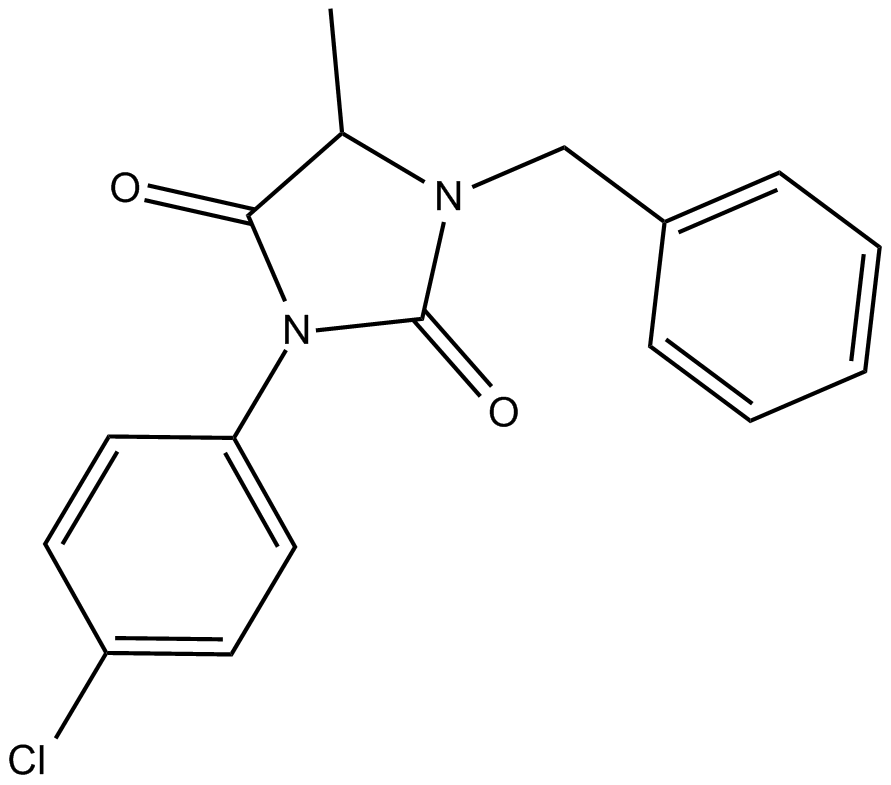
ALLO-1
IC50: 50 nM for wile type Smo
ALLO-1 is a SMO antagonist.
Hedgehog (Hh) proteins are important development regulators that bind the cell-surface protein, which allows the activation of a GPCR-like receptor, Smoothened (SMO). In vertebrates, the SMO activation finally results in the activation of the zinc-finger transcription factors of the Gli family. In addition, the overactivation of SMO may lead to certain cancers.
In vitro: Previous study found that ALLO-1 and its close analog ALLO-2 could inhibit Smo agonist Hh-Ag 1.5-induced luciferase expression in TM3-Gli-Luc cells. The potency of ALLO-1 did not change when either low dose or high dose of Hh-Ag 1.5 was used, in contrast to other known Smo antagonists that are strong SAG or Hh-Ag 1.5 competitors. Moreover, it was found that in contrast to GDC-0449, both ALLO-1 and ALLO-2 inhibited wild-type and the D477G mutant with only around2-fold shift in IC50, indicating that the D477G mutation did not significantly interfere with the binding of ALLO-1 and ALLO-2 to Smo. In addition, ALLO-1 as well as ALLO-2 were able to inhibit both wild-type and D473H mutant human SMO with similar potencies [1].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Tao, H. ,Jin, Q.,koo, D.I., et al. Small molecule antagonists in distinct binding modes inhibit drug-resistant mutant of smoothened. Chemistry & Biology 18, 432-437 (2011).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 314.8 |
| Cas No. | 37468-32-9 |
| Formula | C17H15ClN2O2 |
| Solubility | ≤20mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | 3-(4-chlorophenyl)-5-methyl-1-(phenylmethyl)-2,4-imidazolidinedione |
| SDF | Download SDF |
| Canonical SMILES | O=C1N(C2=CC=C(Cl)C=C2)C(N(CC3=CC=CC=C3)C1C)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure