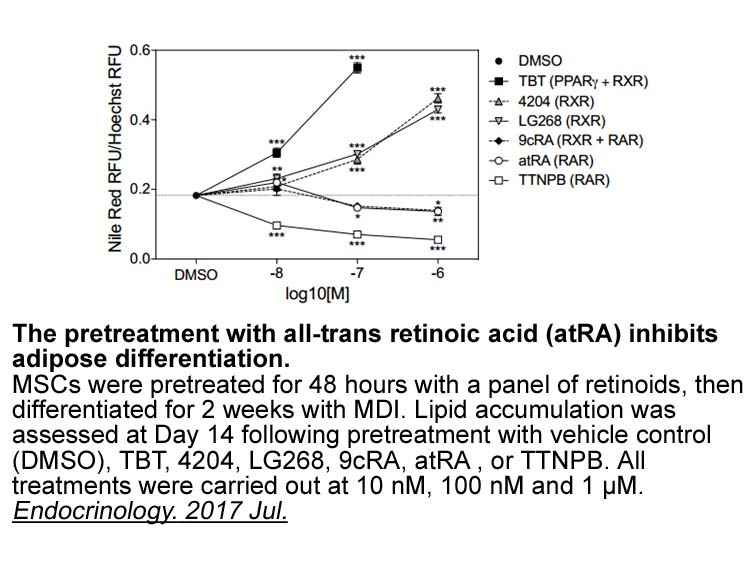
all-trans Retinal
All-trans Retinal, also known as Vitamin A aldehyde or Retinaldehyde, is one of the many forms of vitamin A and also the oxidation product of all-trans retinol [1]. All-trans Retinal are associated with one of the two isoforms of cellular retinol-binding proteins (CRBP-I and CRBP-II) with Kd values of 50 and 90 nM, respectively [1].
CRBP-I and CRBP-II were the first intracellular retinoid-binding proteins. Both proteins display a similar binding affinity towards retinal. They play important roles in retinoid biology and regulation of the metabolism of retinol and retinal. CRBP-I is used to regulate vitamin A storage and synthesis of retinoic acid. And CRBP-II has a role in the initial processing of retinol from food [1].
All-trans Retinal is one form of vitamin A. All-trans Retinal, the initial substrate of retinoid cycle, is a chemically reactive aldehyde that can form toxic conjugates with proteins and lipids, leading to degeneration of the retina [2].
References:
[1]. Noy N. Retinoid-binding proteins: mediators of retinoid action. Biochem J. 2000 Jun 15;348 Pt 3:481-95.
[2]. Kiser PD, Golczak M, Maeda A, et al. Key enzymes of the retinoid (visual) cycle in vertebrate retina. Biochim Biophys Acta. 2012 Jan;1821(1):137-51.
| Storage | Store at -20°C |
| M.Wt | 284.4 |
| Cas No. | 116-31-4 |
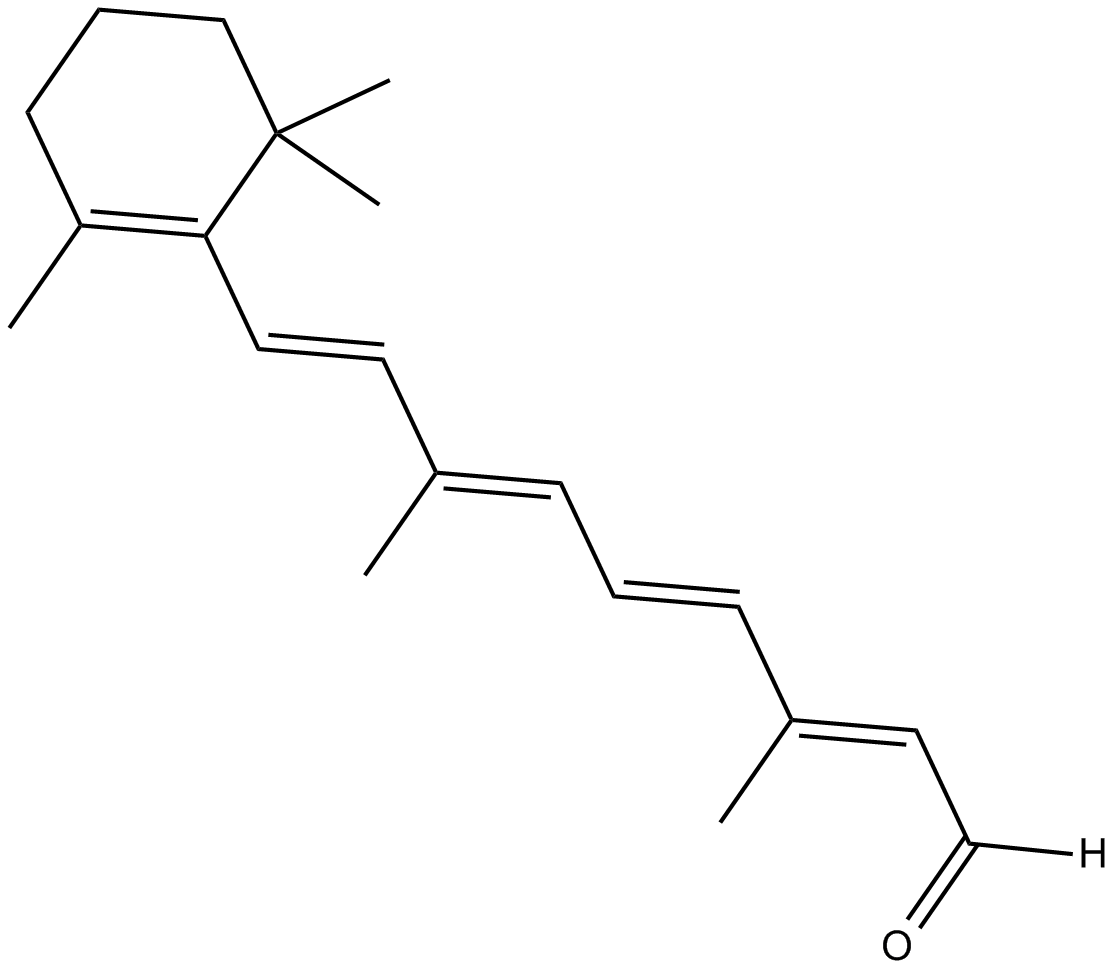
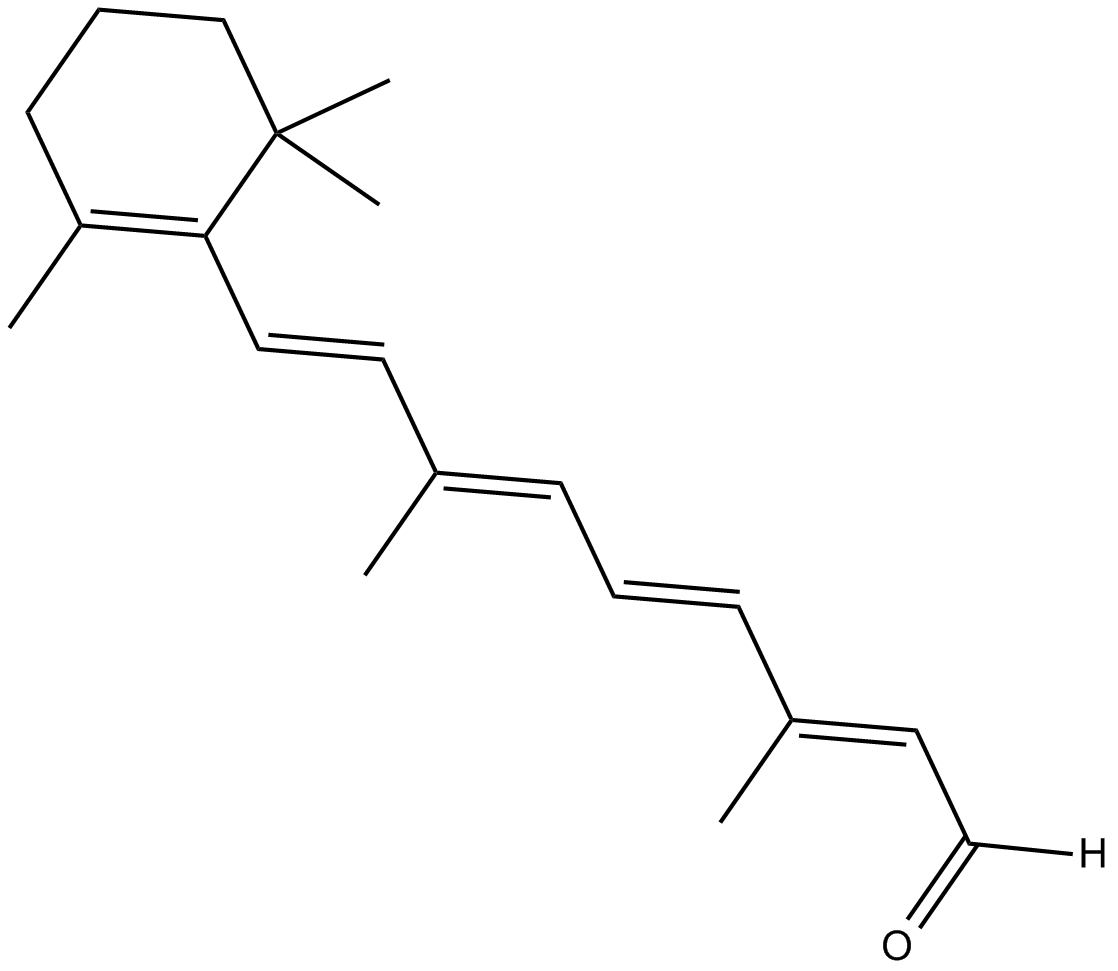
| Formula | C20H28O |
| Synonyms | Retinaldehyde,Vitamin A aldehyde |
| Solubility | insoluble in H2O; ≥56.6 mg/mL in EtOH; ≥57.4 mg/mL in DMSO |
| Chemical Name | retinal |
| SDF | Download SDF |
| Canonical SMILES | CC1=C(/C=C/C(C)=C/C=C/C(C)=C/C([H])=O)C(C)(C)CCC1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data