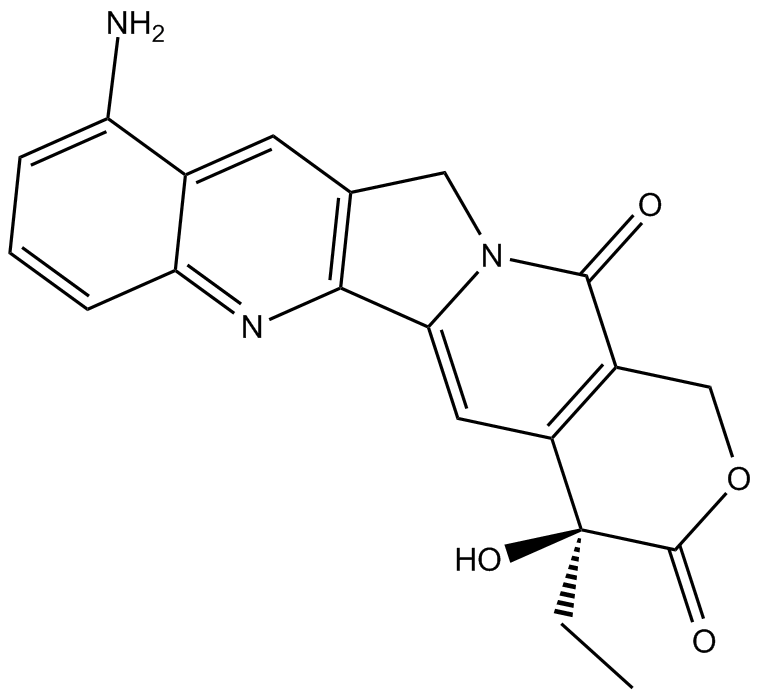
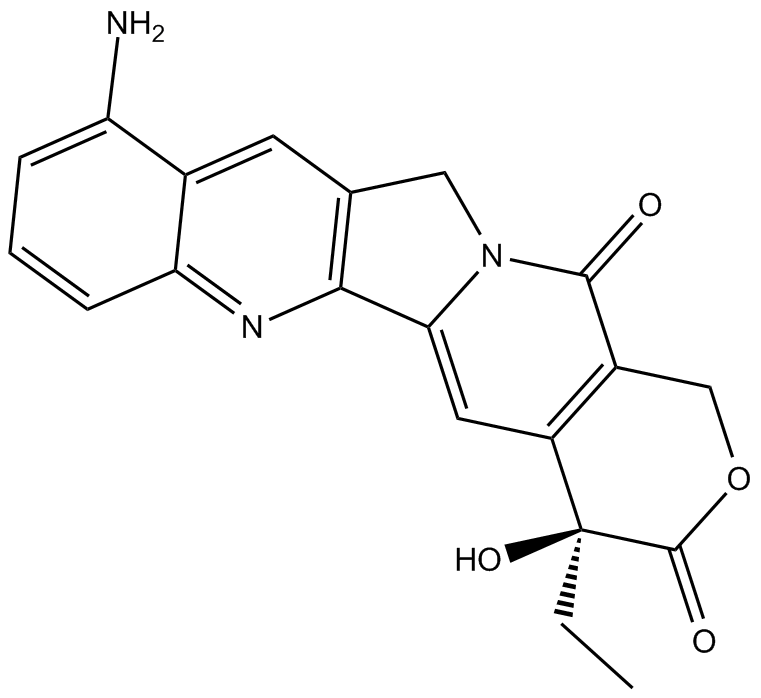
9-amino Camptothecin
9-amino Camptothecin is a topoisomerase I inhibitor [1][2].
DNA topoisomerases relax DNA torsional strain generated during replication, transcription, recombination, repair, and chromosome condensation. The relaxation of DNA supercoiling by topoisomerase I is enabled by a mechanism of controlled rotation around a transient DNA single-strand break. Camptothecin (CPT) is isolated from the bark of the Chinese tree Camptotheca accuminata [3].
9-amino Camptothecin, a water-soluble camptothecin analogue, is a topoisomerase I inhibitor. In human HT-29 colon adenocarcinoma, 9-amino Camptothecin (9-AC) exhibited cytotoxicity with IC50 value of 19 nM. 9-AC also induced DNA damage in whole cells and nuclei at a concenstration of 85 nM and 21 nM, respectively [1].
9-amino Camptothecin had greater activity than camptothecin against human tumour xenografts, including Lewis lung carcinoma and B16 melanoma. 9-AC had entered phase II trials. In patients with advanced solid tumours, 9-amino Camptothecin exhibited anti-tumor activity [1][2].
References:
[1]. Rothenberg, M.L. Topoisomerase I inhibitors: Review and update. Annals of Oncology 8(9), 837-855 (1997).
[2]. Dancey J, Eisenhauer EA. Current perspectives on camptothecins in cancer treatment. Br J Cancer. 1996 Aug;74(3):327-38.
[3]. Drwal MN1, Agama K, Wakelin LP, et al. Exploring DNA topoisomerase I ligand space in search of novel anticancer agents. PLoS One. 2011;6(9):e25150.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 363.4 |
| Cas No. | 91421-43-1 |
| Formula | C20H17N3O4 |
| Synonyms | NSC 603071 |
| Solubility | ≤1mg/ml in DMSO;1mg/ml in dimethyl formamide |
| Chemical Name | (4S)-10-amino-4-ethyl-4-hydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione |
| SDF | Download SDF |
| Canonical SMILES | CC[C@](C(C=C1N2Cc3cc(c(N)ccc4)c4nc13)=C(CO1)C2=O)(C1=O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity ≥ 95.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure