7BIO
7-bromoindirubin-3'-oxime (7BIO), a derivative of indirubin, is a caspase independent nonapoptotic cell death inducer [1]. 7-bromoindirubin-3'-oxime is an inhibitor of FLT3, DYRK1A, DYRK2, Aurora B and Aurora C kinases.
FLT3 is a cytokine receptor expressed on the surface of many hematopoietic progenitor cells. Signalling of FLT3 is important for the normal development of haematopoietic stem cells and progenitor cells. DYRK1A and DYRK2 have been involved in catalyzing its autophosphorylation on serine/threonine and tyrosine residues and play a significant role in a signaling pathway regulating cell proliferation. Aurora kinases are serine/threonine kinases which are essential for cell proliferation and cellular division by controlling chromatid segregation.
7-bromoindirubin-3'-oxime (7BIO) showed a marginal inhibitory activity towards CDKs and GSK-3. 7BIO triggered a rapid cell death process distinct from apoptosis. 7BIO induced the appearance of large pycnotic nuclei, without classical features of apoptosis such as chromatin condensation and nuclear fragmentation. 7BIO-induced cell death was not accompanied by cytochrome c release neither by any measurable effector caspase activation. 7BIO triggered the activation of non-apoptotic cell death, possibly through necroptosis or autophagy. 7BIO inhibited FLT3, the dual-specificity tyrosine phosphorylation-regulated kinases, DYRK1A and DYRK2 with the IC50 values of 0.34 μM, 1.9 and 1.3 μM, respectively [2]. 7BIO also inhibited the activity of Aurora B and C kinases with IC50 values of 4.6 and 0.7 μM, respectively [3].
References:
[1] Ribas J, Bettayeb K, Ferandin Y, et al. 7-Bromoindirubin-3′-oxime induces caspase-independent cell death[J]. Oncogene, 2006, 25(47): 6304-6318.
[2] Myrianthopoulos V, Kritsanida M, Gaboriaud-Kolar N, et al. Novel inverse binding mode of indirubin derivatives yields improved selectivity for DYRK kinases[J]. ACS medicinal chemistry letters, 2012, 4(1): 22-26.
[3] Myrianthopoulos V, Magiatis P, Ferandin Y, et al. An integrated computational approach to the phenomenon of potent and selective inhibition of aurora kinases B and C by a series of 7-substituted indirubins[J]. Journal of medicinal chemistry, 2007, 50(17): 4027-4037.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 356.2 |
| Cas No. | 916440-85-2 |
| Formula | C16H10BrN3O2 |
| Synonyms | 7-Bromoindirubin-3’-oxime |
| Solubility | ≥41.9 mg/mL in DMSO; ≥2 mg/mL in EtOH with gentle warming and ultrasonic; insoluble in H2O |
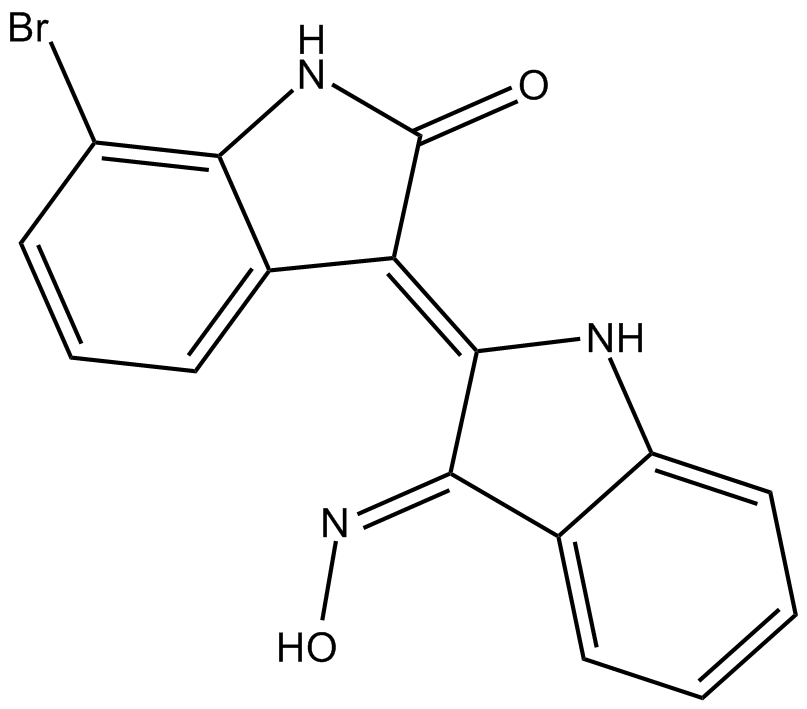
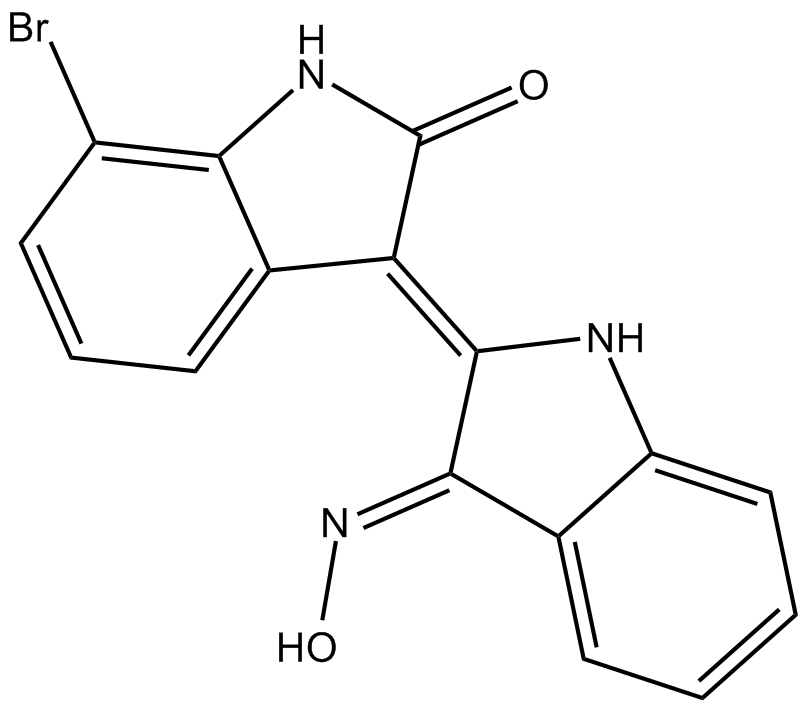
| Chemical Name | 7-bromo-3-[1,3-dihydro-3-(hydroxyimino)-2H-indol-2-ylidene]-1,3-dihydro-2H-indol-2-one |
| SDF | Download SDF |
| Canonical SMILES | O/N=C(\c1ccccc1N1)/C\1=C(\c1cccc(Br)c1N1)/C1=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure