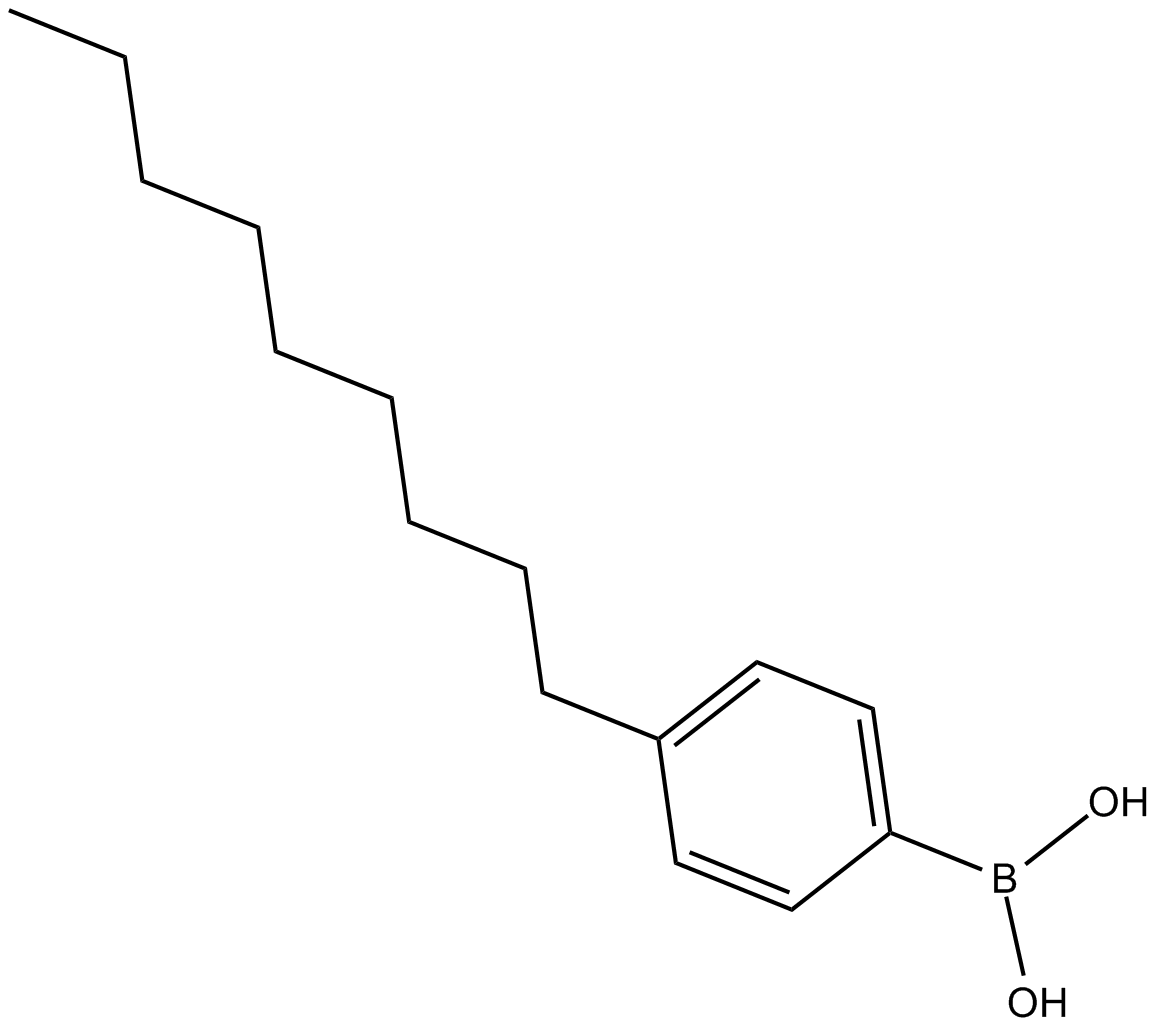
4-(n-nonyl) Benzeneboronic Acid
IC50: 9.1 nM
4-(n-nonyl) Benzeneboronic acid is a dual FAAH inhibitor.
Fatty acid amide hydrolase (FAAH), a membrane-bound enzyme of the endocannabinoid system, has been identified as a potential target for therapeutic agents in the treatment of various medical conditions, such as inflammation and pain. FAAH and monoglyceride lipase (MGL) have been reported to be the primary enzyme responsible for the hydrolysis of endocannabinoid N-arachidonoyl ethanolamide (AEA), which is a key lipid messenger in the brain and periphery.
In vitro: 4-(n-Nonyl) benzeneboronic acid was synthezed as a potent inhibitor of FAAH, with an IC50 of 9.1 nM. 4-(n-Nonyl) benzeneboronic acid was also found to be able to inhibit MAGL, which could hydrolyze 2-arachidonoyl glycerol, but at around 1000-fold higher concentration. Moreover, it was found that as the most potent para-substituted compound, 4-(n-Nonyl) benzeneboronic acid showed rather high pKa of 9.1. In addition, the molecular docking was utilized to gain insight on the FAAH binding mode of 4-(n-Nonyl) benzeneboronic acid and a putative binding mode was observed [1].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Minkkil, A. ,Saario, S.M.,Ksnnen, H., et al. Discovery of boronic acids as novel and potent inhibitors of fatty acid amide hydrolase. Journal of Medicinal Chemistry 51, 7057-7060 (2008).
| Storage | Store at -20°C |
| M.Wt | 248.2 |
| Cas No. | 256383-45-6 |
| Formula | C15H25BO2 |
| Solubility | insoluble in H2O; ≥11.75 mg/mL in DMSO; ≥25.1 mg/mL in EtOH |
| Chemical Name | B-(4-nonylphenyl)-boronic acid |
| SDF | Download SDF |
| Canonical SMILES | CCCCCCCCCc1ccc(B(O)O)cc1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure