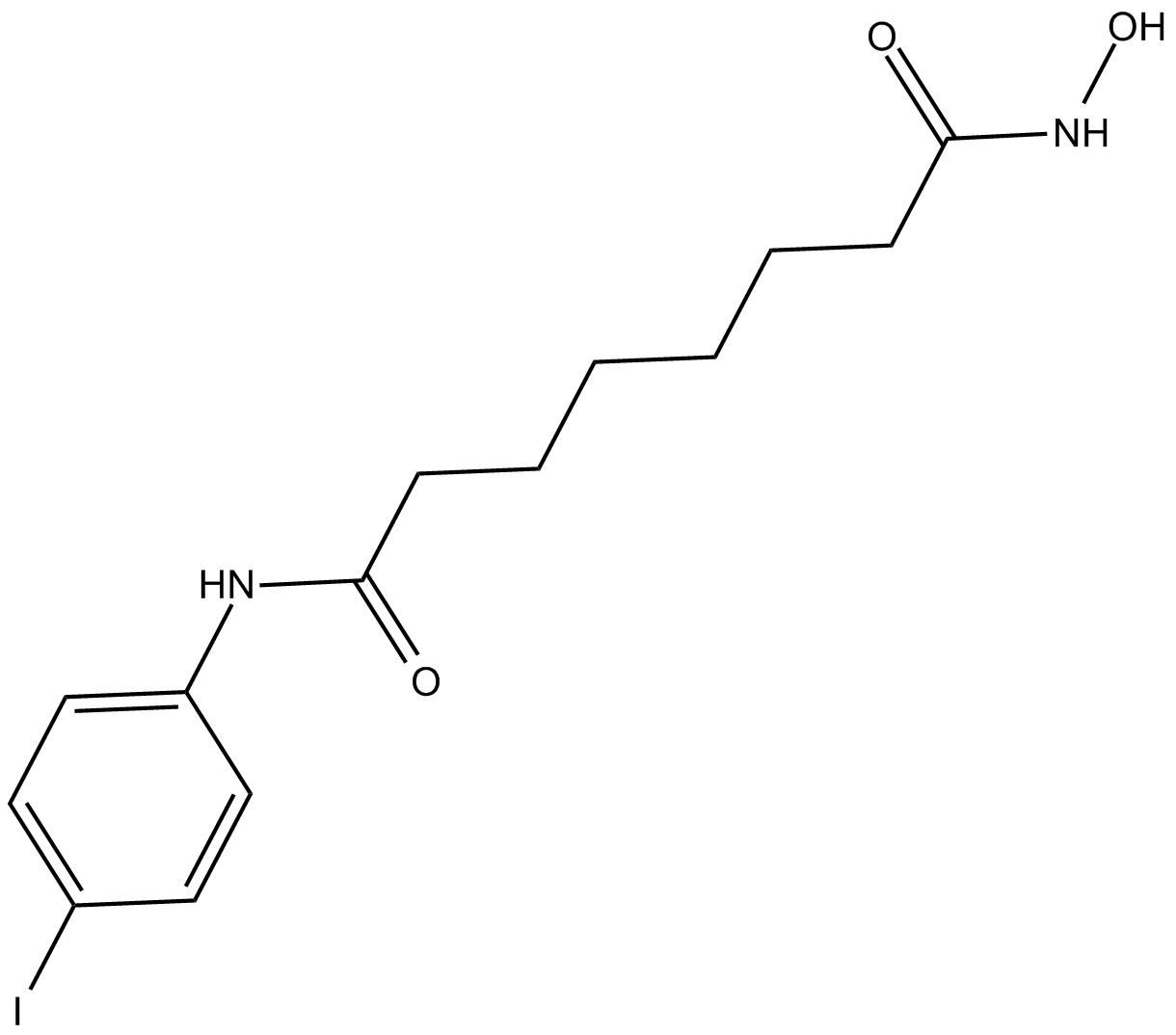
4-iodo-SAHA
4-iodo-SAHA is a hydrophobic derivative of SAHA, the class I and class II histone deacetylase (HDAC) inhibitor [1].
The reversible acetylation of lysine residues in histone plays an important role in transcriptional activation and repression. The regulation of these post-translational modifications is balanced by histone acetyltransferase (HAT) and histone deacetylase (HDAC) activities. HDACs are also involved in reversible acetylation of non-histone proteins [1].
4-iodo-SAHA is a histone deacetylase (HDAC) inhibitor. In SKBR3-breast-derived cell line, 4-iodo-SAHA inhibited cell proliferation with EC50 value of 1.1 μM. In HT29 colon-derived cell line, leukemia-derived U937 tumor cell line, JA16, HL60 and K562 cell lines, 4-iodo-SAHA inhibited cell proliferation with EC50 values of 0.95, 0.12, 0.24, 0.85 and 1.3 μM, respectively. 4-iodo-SAHA is 10-fold more potent as an inhibitor of U937 leukemia cell proliferation compared to SAHA (0.12 μM versus 1.2 μM). In SKBR3 cells, 4-iodo-SAHA reduced acetylated H4 and p21 levels [1].
Reference:
[1]. Salmi-Smail C, Fabre A, Dequiedt F, et al. Modified cap group suberoylanilide hydroxamic acid histone deacetylase inhibitor derivatives reveal improved selective antileukemic activity. J Med Chem. 2010 Apr 22;53(8):3038-47.
| Storage | Store at -20°C |
| M.Wt | 390.2 |
| Cas No. | 1219807-87-0 |
| Formula | C14H19IN2O3 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥1.67 mg/mL in DMSO |
| Chemical Name | N1-hydroxy-N8-(4-iodophenyl) octanediamide |
| SDF | Download SDF |
| Canonical SMILES | ONC(CCCCCCC(Nc(cc1)ccc1I)=O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure