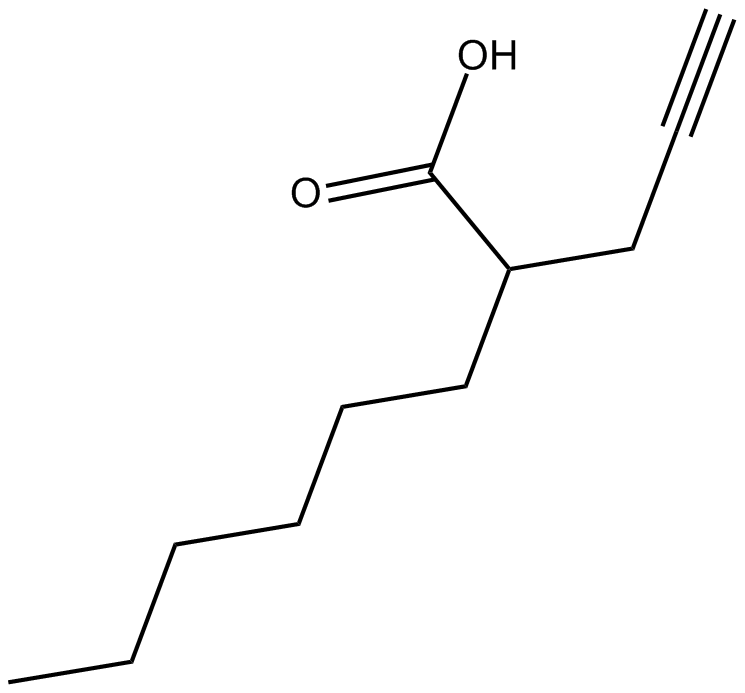
2-hexyl-4-Pentynoic Acid
2-hexyl-4-Pentynoic Acid is a potent and robust inhibitor of HDAC with IC50 value of 13 μM [1].
Histone deacetylases (HDACs) are a class of enzymes that remove acetyl groups from ε-N-acetyl lysines on histones, allowing the histones to wrap the DNA more tightly. DNA expression is regulated by de-acetylation and acetylation.
2-hexyl-4-Pentynoic Acid, a valproic acid (VPA) derivatives, is a potent and robust HDACs inhibitor. In cerebellar granule cells, 2-hexyl-4-Pentynoic Acid (5 μM) significantly and dose-dependently increased acetylated histone H3 (Ac-H3) levels, and at 50-100 μM led to a maximal increase of 600-700%, compared with only a 200% increase by VPA at 100 μM. 2-hexyl-4-Pentynoic Acid also completely blocked glutamate-induced cell death at 50-100 μM. Also, 2-hexyl-4-Pentynoic Acid restored glutamate-induced neuronal loss. At 50 μM, 2-hexyl-4-Pentynoic Acid effectively increased HSP70-1a and HSP70-1b mRNA levels through HDAC inhibition [1].
Reference:
[1]. Leng Y, Marinova Z, Reis-Fernandes MA, et al. Potent neuroprotective effects of novel structural derivatives of valproic acid: potential roles of HDAC inhibition and HSP70 induction. Neurosci Lett, 2010, 476(3): 127-132.
| Physical Appearance | A colorless liquid |
| Storage | Store at -20°C |
| M.Wt | 182.26 |
| Cas No. | 96017-59-3 |
| Formula | C11H18O2 |
| Solubility | insoluble in H2O; ≥15.45 mg/mL in DMSO; ≥51.8 mg/mL in EtOH |
| Chemical Name | 2-(prop-2-yn-1-yl)octanoic acid |
| SDF | Download SDF |
| Canonical SMILES | CCCCCCC(CC#C)C(O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure