trans-AUCB
IC50: 0.5 nM
trans-AUCB is a potent inhibitor of soluble epoxide hydrolase (sEH).
Soluble epoxide hydrolase (sEH) can convert epoxides to their corresponding diols. Inhibitors of sEH have anti-hypertensive, anti-inflammatory, neuroprotective, and cardioprotective effects.
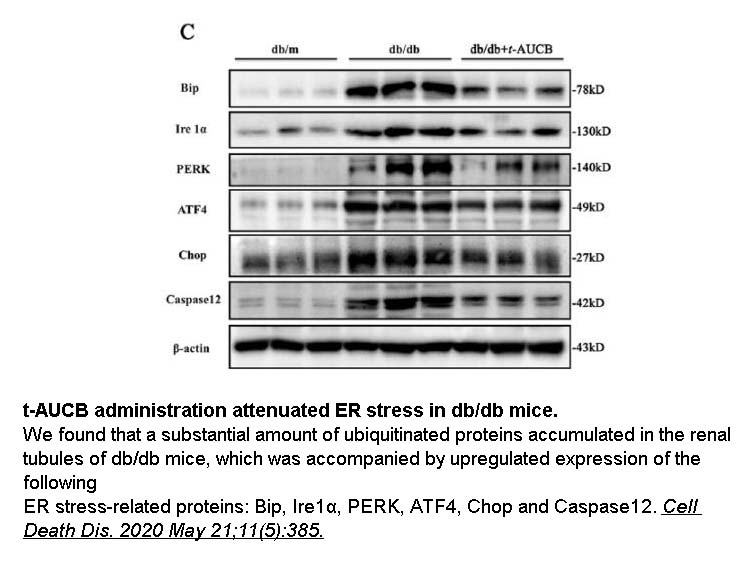
In vitro: A previous study showed that the pretreatment with DAPT could substantially potentiate the growth inhibition caused by t-AUCB in U251 and U87 cells. Moreover, the pretreatment with DAPT markedly increased t-AUCB-induced apoptosis of U251 and U87 cells. Moreover, T-AUCB alone did not obviously affect caspase-3 activity in the cells, but t-AUCB plus DAPT pretreatment caused significant increase of caspase-3 activity. In addition, the pretreatment with DAPT was able to completely block t-AUCB-induced phosphorylation of p38 MAPK, MAPKAPK2 and Hsp27 in the cells [1].
In vivo: A previous animal study was conducted to investigate the effects of acute sEH inhibition by t-AUCB on infarct volume, functional outcome, and changes in cerebral blood flow (CBF) in a rat model of ischemic stroke. It was found that t-AUCB could significantly reduce cortical infarct volume by 35%, elevate cumulative epoxyeicosatrienoic acids-to-dihydroxyeicosatrienoic acids ratio in brain cortex by twofold, and improve functional outcome in arm-flexion test when compared with that of the vehicle-treated group [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Li JY, Li RJ, Wang HD. γ-secretase inhibitor DAPT sensitizes t-AUCB-induced apoptosis of human glioblastoma cells in vitro via blocking the p38 MAPK/MAPKAPK2/Hsp27 pathway. Acta Pharmacol Sin. 2014 Jun;35(6):825-31.
[2] Shaik JS, Ahmad M, Li W, Rose ME, Foley LM, Hitchens TK, Graham SH, Hwang SH, Hammock BD, Poloyac SM. Soluble epoxide hydrolase inhibitor trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid is neuroprotective in rat model of ischemic stroke. Am J Physiol Heart Circ Physiol. 2013 Dec 1;305(11):H1605-13.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 412.5 |
| Cas No. | 885012-33-9 |
| Formula | C24H32N2O4 |
| Solubility | ≤30mg/ml in DMSO;20mg/ml in dimethyl formamide |
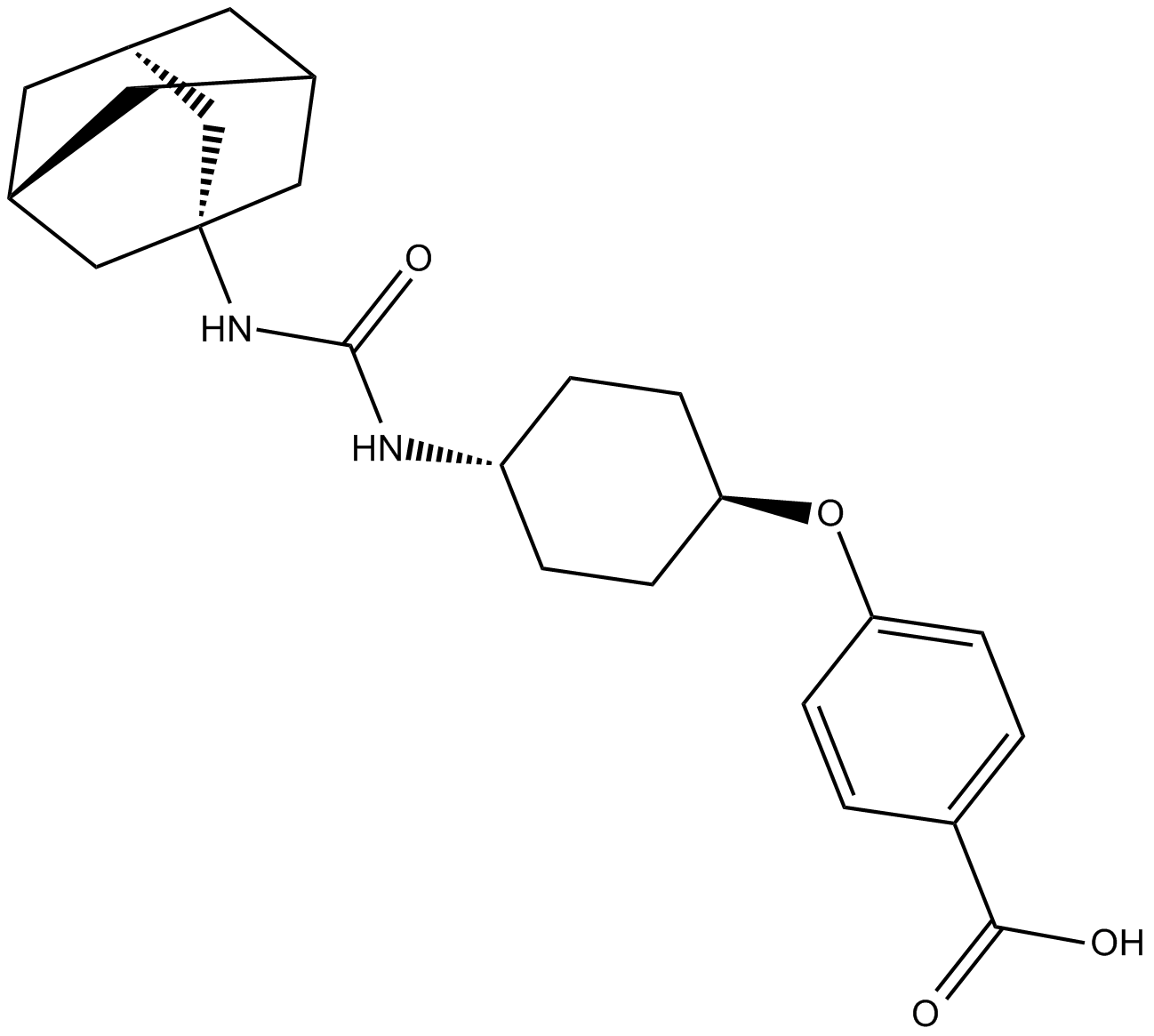
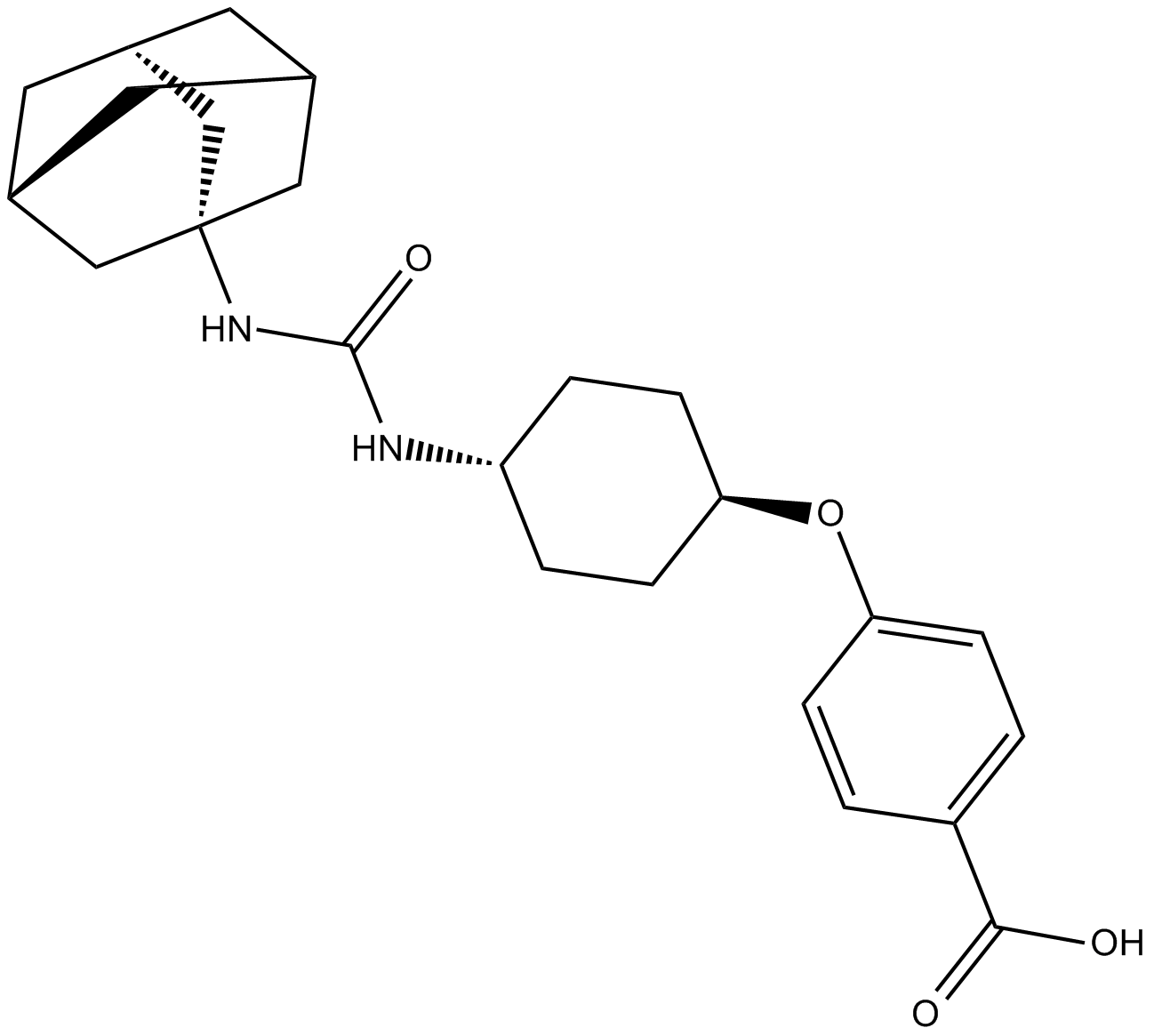
| Chemical Name | 4-[[trans-4-[[(tricyclo[3.3.1.13,7]dec-1-ylamino)carbonyl]amino]cyclohexyl]oxy]-benzoic acid |
| Canonical SMILES | OC(c(cc1)ccc1O[C@H](CC1)CC[C@@H]1NC(NC1(C[C@H](C2)C3)C[C@H]3CC2C1)=O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity ≥ 90.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure
Related Biological Data