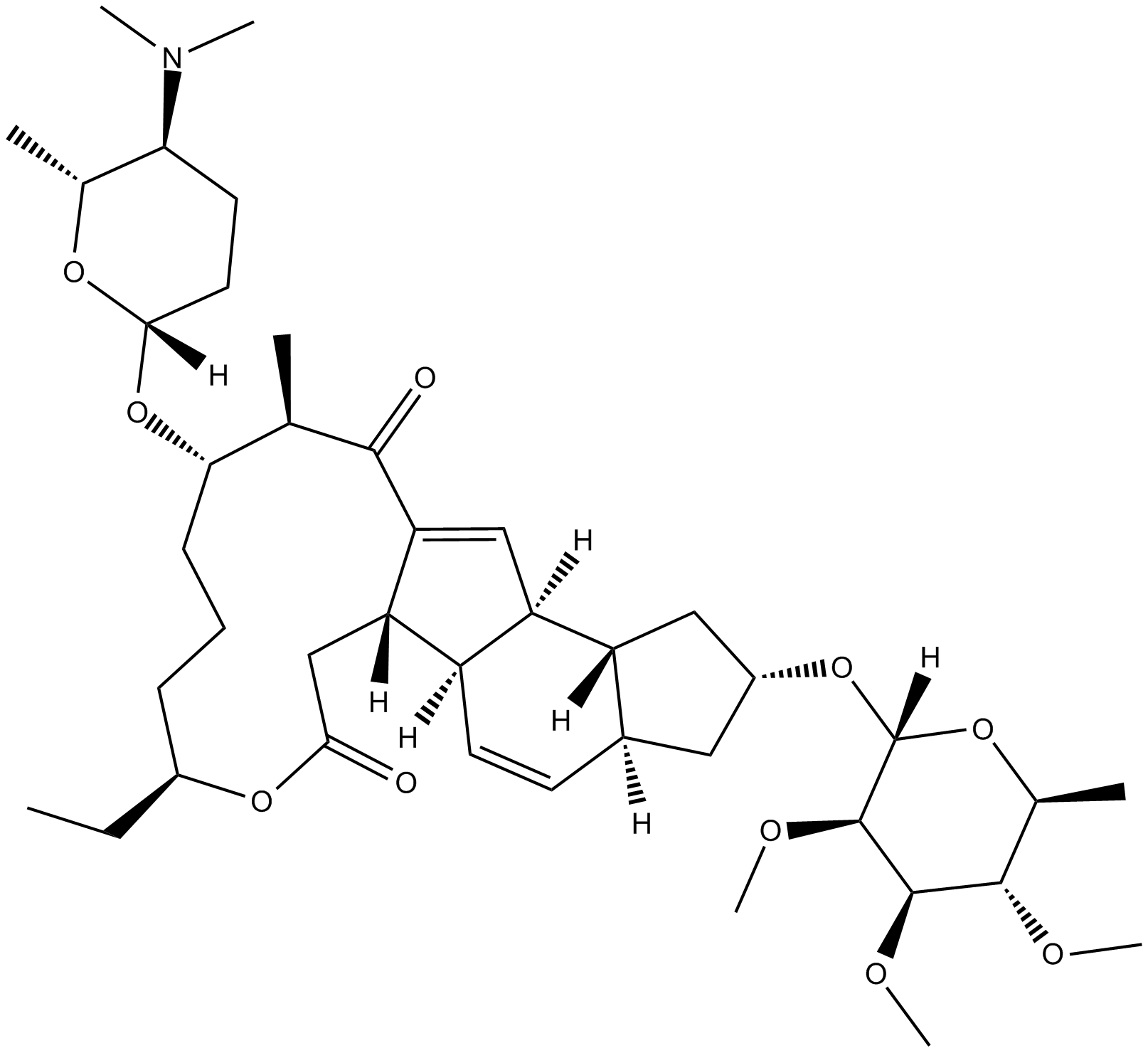
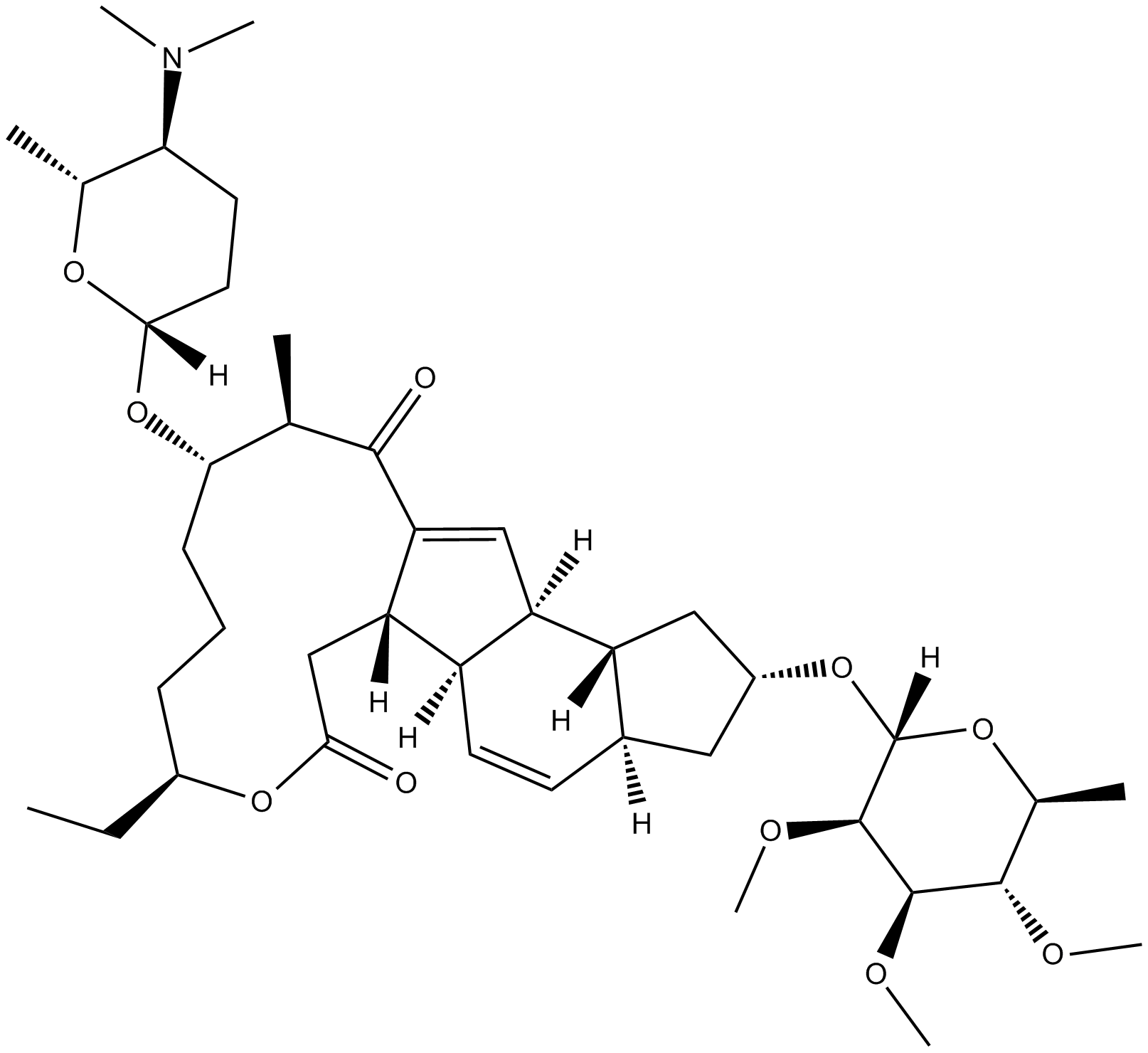
Spinosyn A
Spinosyn A is an insect nicotinic acetylcholinesterase receptors (nAChRs) agonist and potent insecticide.
The spinosyns are a family of macrolide natural products produced by the soil microorganism Saccharopolyspora spinosa. Spinosyn A is identified as a naturally-occurring macrocyclic lactone that is a potent insecticide.
In vitro: The mixture of spinosyns A and D, a commercial insecticide TracerTM (DowAgroSciences), is useful against various crop pests such as tobacco budworm. It was found that the deoxy analogs of spinosyns A were more potent insecticides than their respective parent factor. Moreover, the 2’-desmethoxy analogs of spinosyns A showed insecticidal potency against H. virescens greater than that of spinosyns A and D, suggesting that polarity was not well tolerated. Furthermore, the activity of 3'-deoxy spinosyn J was about the same as spinosyn A, and the activity of 2'-deoxy spinosyn H was found to be slightly greater than that of spinosyn A [1].
In vivo: Currently, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] L. C. Creemer, H. A. Kirst, J. W. Paschal, et al. Synthesis and insecticidal activity of spinosyn analogs functionally altered at the 2'-,3'- and 4'-positions of the rhamnose moiety. J.Antibiot.(Tokyo) 53(2), 171-178 (2000).
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 732.0 |
| Cas No. | 131929-60-7 |
| Formula | C41H65NO10 |
| Synonyms | A-83543A|Lepicidin A |
| Solubility | Soluble in DMSO |
| Chemical Name | (2R,3aS,5aR,5bS,9S,13S,14R,16aS,16bR)-2-[(6-deoxy-2,3,4-tri-O-methyl-α-L-mannopyranosyl)oxy]-13-[[(2R,5S,6R)-5-(dimethylamino)tetrahydro-6-methyl-2H-pyran-2-yl]oxy]-9-ethyl-2,3,3a,5a,5b,6,9,10,11,12,13,14,16a,16b-tetradecahydro-14-methyl-1H-as-indaceno[3, |
| Canonical SMILES | CN(C)[C@H]1CC[C@@](O[C@H]([C@@H](C)C2=O)CCC[C@H](CC)OC(C[C@]3([H])C2=C[C@]4([H])[C@@]3([H])C=C[C@@]5([H])[C@@]4([H])C[C@H](O[C@]6([H])O[C@@H](C)[C@H](OC)[C@@H](OC)[C@H]6OC)C5)=O)([H])O[C@@H]1C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure