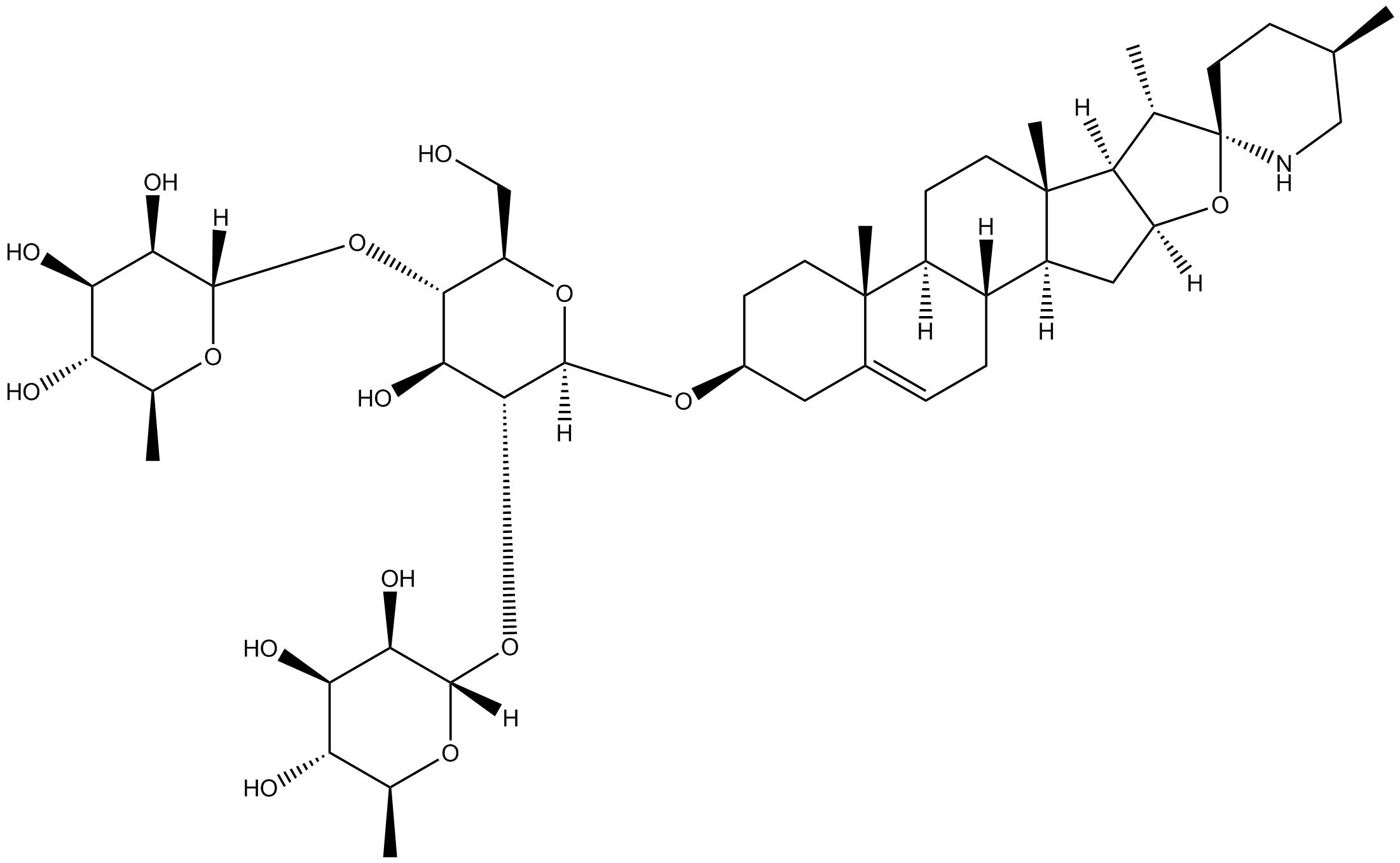
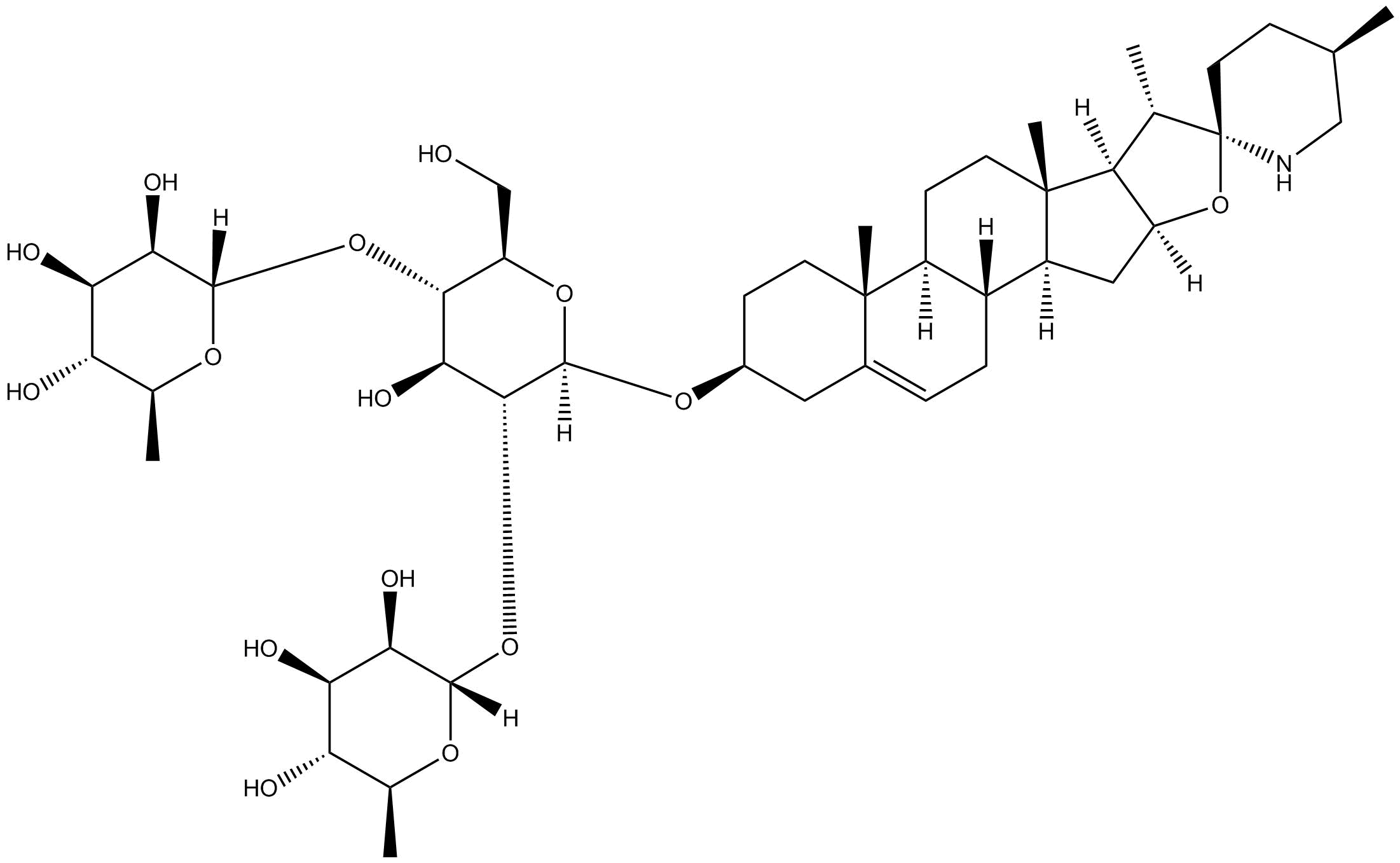
Solamargine
Solamargine, apoisonouschemical compound, occurs in plants of theSolanaceaefamily, such aspotatoes,tomatoes, andeggplants[1].Solamargine has been also isolated fromSolanum nigrumfungalendophyteAspergillus flavus[2].
In vitro: Solamargine significantly inhibited migration and invasion of HepG2 cells by down-regulation of MMP-2 and MMP-9 expression and activity. Solamargine reduced HepG2 cell viability in a concentration-dependent manner. In HepG2 cells, solamargine (7.5 μM) decreased cell viability by less than 20%.Solamargineat the highest dose decreased cell migration and invasion by more than 70% and 72%, respectively.Solamarginealso reduced expression and function of MMP-2 and MMP-9 proteins [3].In lung cancer cells, Solamargine inhibited proliferation and induced apoptosis through p38 MAPK-mediated suppression of phosphorylation and protein expression of Stat3, followed by inducing Stat3 downstream effector p21. In non-small-cell lung cancer (NSCLC) cells, solamargine inhibited growth and induced apoptosis of in a time- and dose-dependent manner.Solamargine also increased phosphorylation of p38 mitogen-activated protein kinase (p38 MAPK) in a time-dependent fashion [4].In SMMC-7721 cells, solamargine exerted potential anticancer activity through activation of caspase-3 and regulation of the cell cycle progression to induce apoptosis and inhibit hepatoma cells proliferation [5].In WM115 and WM239 cells, solamargine rapidly, selectively and effectively inhibited the growth of cells WM239 and WM115. Solamarginealso showed high efficacy against the vertical growth phase melanoma cells [6].
References:
[1]. Al Chami L, Méndez R, Chataing B, et al. Toxicological effects of α‐solamargine in experimental animals[J]. Phytotherapy Research, 2003, 17(3): 254-258.
[2]. El‐Hawary S S, Mohammed R, AbouZid S F, et al. Solamargine production by a fungal endophyte of Solanum nigrum[J]. Journal of applied microbiology, 2016.
[3]. Sani I K, Marashi S H, Kalalinia F. Solamargine inhibits migration and invasion of human hepatocellular carcinoma cells through down-regulation of matrix metalloproteinases 2 and 9 expression and activity[J]. Toxicology in Vitro, 2015, 29(5): 893-900.
[4]. Aggarwal B B, Sethi G, Ahn K S, et al. Targeting Signal‐Transducer‐and‐Activator‐of‐Transcription‐3 for Prevention and Therapy of Cancer[J]. Annals of the New York Academy of Sciences, 2006, 1091(1): 151-169.
[5]. Ding X, Zhu F S, Li M, et al. Induction of apoptosis in human hepatoma SMMC-7721 cells by solamargine from Solanum nigrumL[J]. Journal of ethnopharmacology, 2012, 139(2): 599-604.
[6]. Al Sinani S S, Eltayeb E A, Coomber B L, et al. Solamargine triggers cellular necrosis selectively in different types of human melanoma cancer cells through extrinsic lysosomal mitochondrial death pathway[J]. Cancer cell international, 2016, 16(1): 1.
| Storage | Store at -20°C |
| M.Wt | 868.07 |
| Cas No. | 20311-51-7 |
| Formula | C45H73NO15 |
| Solubility | ≥86.8 mg/mL in DMSO; insoluble in H2O; ≥36.6 mg/mL in EtOH |
| Chemical Name | (2S,2'S,3R,3'R,4R,4'R,5R,5'R,6S,6'S)-6,6'-(((2R,3S,4S,5R,6R)-4-hydroxy-2-(hydroxymethyl)-6-(((4S,5'R,6aR,6bS,8aS,8bR,9S,10R,11aS,12aS,12bS)-5',6a,8a,9-tetramethyl-1,3,4,5,6,6a,6b,7,8,8a,8b,9,11a,12,12a,12b-hexadecahydrospiro[naphtho[2',1':4,5]indeno[2,1-b |
| Canonical SMILES | C[C@H]1CN[C@@]2(O[C@]3(C[C@]4([C@@]5(CC=C6C[C@@H](O[C@]7([H])O[C@H](CO)[C@@H](O[C@@]8([H])[C@H](O)[C@H](O)[C@@H](O)[C@H](C)O8)[C@H](O)[C@H]7O[C@@]9([H])[C@H](O)[C@H](O)[C@@H](O)[C@H](C)O9)CC[C@@]6([C@]5(CC[C@@]4([C@]3([C@@H]2C)[H])C)[H])C)[H])[H])[H])CC1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure