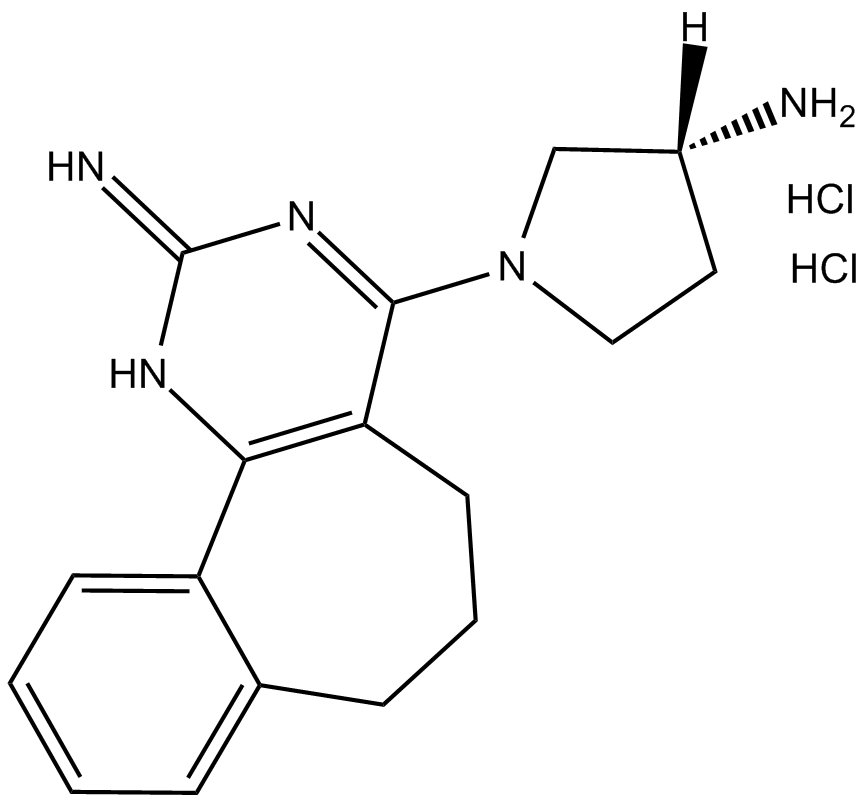
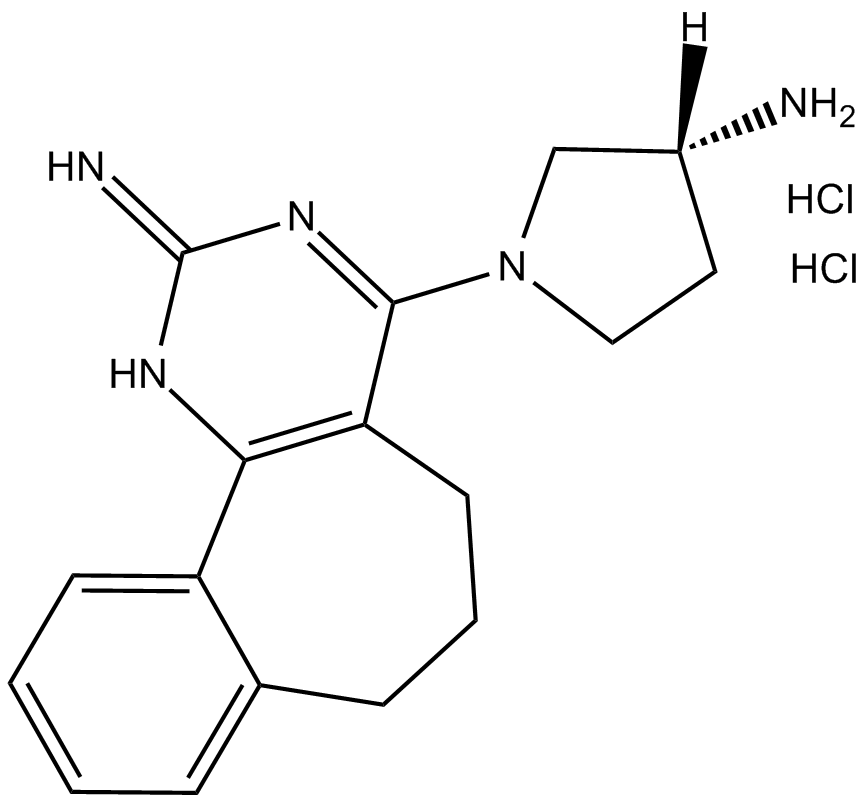
A 943931 dihydrochloride
A 943931, is an H4R (one of histamine receptor subtypes) antagonist [1] with high affinities to H4Rs of human (Ki = 5 nM), rat (Ki = 4 nM) and mouse (Kb = 6 nM) [2].
H4R is one of 4 known G-protein-coupled receptors (H1, H2, H3 and H4 receptors) of histamine for histamine to mediate its physiological functions [3].
HMC-1 cells incubated with A 943931 at a concentration of 300 nM for 20 min inhibited the increase in ALDH2 activity induced by H4R [4]. In microglia, A 943931 at a concentration of 10 μM partially abolish the release of TNF-α and IL-6 induced by histamine at a concentration of 0.1 μg/ml [5]. In bone marrow-derived mast cells, A 943931 inhibited the shape change induced by histamine (IC50 = 0.38 μM) [6].
Intraperitoneal administration of A 943931 at a dose of 33 μmol/kg potently inhibited itch induced by H4R agonist in mice [6]. In several preclinical models, H4R had been shown to be linked to inflammation [7]. A 943931 had excellent antagonistic activity both in vivo and in vitro across multiple species, displayed good oral bioavailability (90%) and excellent metabolic stability. This compound displays good efficacy in rat pain models and is a good anti-inflammatory agent in mice [8]. A 943931 has an in vivo oral bioavailability of 34% and a half-life of 2.6 h in rats [2]. A 943931 efficaciously reduced acute inflammatory pains induced by formalin in the flinch model and by carrageenan in mechanical and thermal hyperalgesia models in rats [9].
References:
[1]. Erich H. Schneider and Roland Seifert. The histamine H4-receptor and the central and peripheral nervous system: A critical analysis of the literature. Neuropharmacology, 2015, xxx:1-13.
[2]. Rogier A. Smits, Herman D. Lim, Tiffany van der Meer, et al. Ligand based design of novel histamine H4 receptor antagonists; fragment optimization and analysis of binding kinetics. Bioorg. Med. Chem. Lett., 2012, 22: 461-467.
[3]. Huaqing Liu, Robert J. Altenbach, Tracy L. Carr, et al. cis-4-(Piperazin-1-yl)-5,6,7a,8,9,10,11,11a-octahydrobenzofuro[2,3-h]quinazolin-2-amine (A-987306), A New Histamine H4R Antagonist that Blocks Pain Responses against Carrageenan-Induced Hyperalgesia. J. Med. Chem., 2008, 51:7094-7098.
[4]. Silvia Aldi, Ken-ichi Takano, Kengo Tomita, et al. Histamine H4-Receptors Inhibit Mast Cell Renin Release in Ischemia/Reperfusion via PKCε-Dependent Aldehyde Dehydrogenase Type-2 Activation. J. Pharmacol. Exp. Ther., 2014, 349(3):508-17.
[5]. Jin Zhu, Chen Qu, Xiang Lu, et al. Activation of Microglia by Histamine and Substance P. Cell Physiol. Biochem., 2014, 34(3):768-80.
[6]. Harald Engelhardt, Rogier A Smits, Rob Leurs, et al. A new generation of anti-histamines: Histamine H4 receptor antagonists on their way to the clinic. Curr. Opin. Drug Discov. Devel., 2009, 12(5):628-43.
[7]. Jeffery M Cowden, Fuqu Yu, Homayon Banie, et al. The histamine H4 receptor mediates inflammation and Th17 responses in preclinical models of arthritis. Ann. Rheum. Dis., 2014, 73:600-608.
[8]. Rob Leurs, Paul L Chazot, Fiona C Shenton, et al. Molecular and biochemical pharmacology of the histamine H4 receptor. British Journal of Pharmacology, 2009, 157: 14-23.
[9]. David Burns, Niu Shin, Ravi Jalluri, et al. Annual Reports in Medicinal Chemistry: H4 Receptor Antagonists and Their Potential Therapeutic Applications. Burlington: Academic Press, 2014.
| Physical Appearance | Off-white solid |
| Storage | Store at -20°C |
| M.Wt | 368.31 |
| Cas No. | 1227675-50-4 |
| Formula | C17H21N5·2HCl |
| Solubility | <36.83mg/ml in H2O; <36.83mg/ml in DMSO |
| Chemical Name | (R)-1-(2-imino-2,5,6,7-tetrahydro-1H-benzo[6,7]cyclohepta[1,2-d]pyrimidin-4-yl)pyrrolidin-3-amine dihydrochloride |
| SDF | Download SDF |
| Canonical SMILES | N[C@]1([H])CCN(C(C2=C(N3)C4=CC=CC=C4CCC2)=NC3=N)C1.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure