Reparixin L-lysine salt
Reparixin L-lysine salt is an inhibitor of interleukin 8 receptor alpha (CXCR1) and beta (CXCR2) with IC50 value of 5.6 nM and 80 nM respectively [1].
CXCR1 and 2 belong to class A of 7-transmembrane G protein coupled receptor, and they share 78% amino acid identity. They are specific receptors for interleukin-8 which is a chemokine. They are mainly expressed in the neutrophils, where they mediate neutrophil migration to the site of inflammation. In addition, they are also involved in tumor aggressive growth and metastasis of human malignant melanoma.
Structural and biochemical study identified that Reparixin L-lysine salt had non-competitive allosteric interaction with CXCR1 and 2, by which blocking CXCR1 and CXR2 in an inactive conformation, and thus suppressed receptor-induced intracellular signaling cascade and cellular response [2]. When CXCR1 were expressed in L1.2 cells, cell migration induced by 10 nM CXC8 was significantly inhibited by reparixin L-lysine salt [1].
In mouse model, treatment of reparixin L-lysine salt after introduction of injury significantly (15 mg/ kg/ day for 7 days) suppressed secondary degeneration by reducing CXC8-dependent oligodendrocyte apoptosis, migration to the injury site of neutrophils and ED-1-positive cells. Additionally, the level of macrophage-inflammatory protein-2, tumor necrosis factor alpha, interleukin (IL)-6, and IL-1 beta was also reduced, and the proliferation of glial fibrillary acidic protein-positive cells was significantly reduced [2].
In mouse ischemia model, pre-treatment with reparixin reduced the motor deficits, myeloperoxidase activity and IL-1b level. Furthermore, ischemic injury was also attenuated [3].
References:
[1] Moriconi A et al. , Design of noncompetitive interleukin-8 inhibitors acting on CXCR1 and CXCR2. J Med Chem. 2007, 50(17): 3984-4002.
[2] Gorio A, Madaschi L, Zadra G, Reparixin, an inhibitor of CXCR2 function, attenuates inflammatory responses and promotes recovery of functionafter traumatic lesion to the spinal cord. J Pharmacol Exp Ther. 2007, 322(3): 973-981.
[3] Sousa L F, Coelho F M, Rodrigues D H, Blockade of CXCR1/2 chemokine receptors protects against brain damage in ischemic stroke in mice. Clinics (Sao Paulo). 2013, 68(3): 391-394.
- 1. Hyesol Lim, Minsoo Koh, et al. "Cancer-associated fibroblasts induce an aggressive phenotypic shift in non-malignant breast epithelial cells via interleukin-8 and S100A8." J Cell Physiol. 2021 Oct;236(10):7014-7032. PMID:33748944
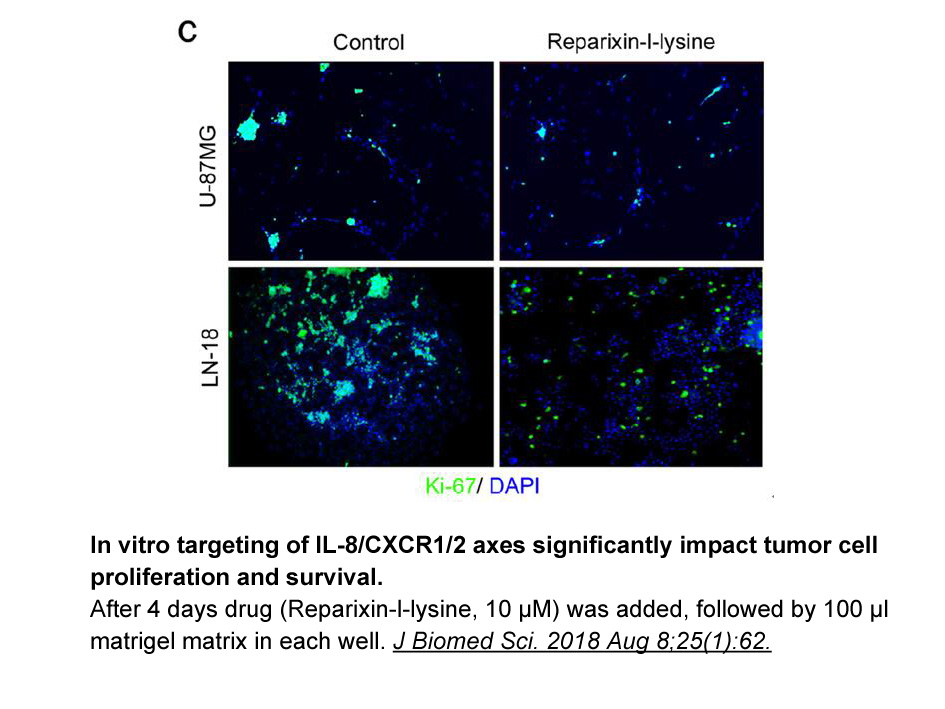
- 2. Sharma I, Singh A, et al. "IL-8/CXCR1/2 signalling promotes tumor cell proliferation, invasion and vascular mimicry in glioblastoma." J Biomed Sci. 2018 Aug 8;25(1):62. PMID:30086759
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 429.57 |
| Cas No. | 266359-93-7 |
| Formula | C20H35N3O5S |
| Synonyms | Repertaxin L-lysine salt |
| Solubility | insoluble in DMSO; ≥16.67 mg/mL in H2O with gentle warming; ≥66.67 mg/mL in EtOH |
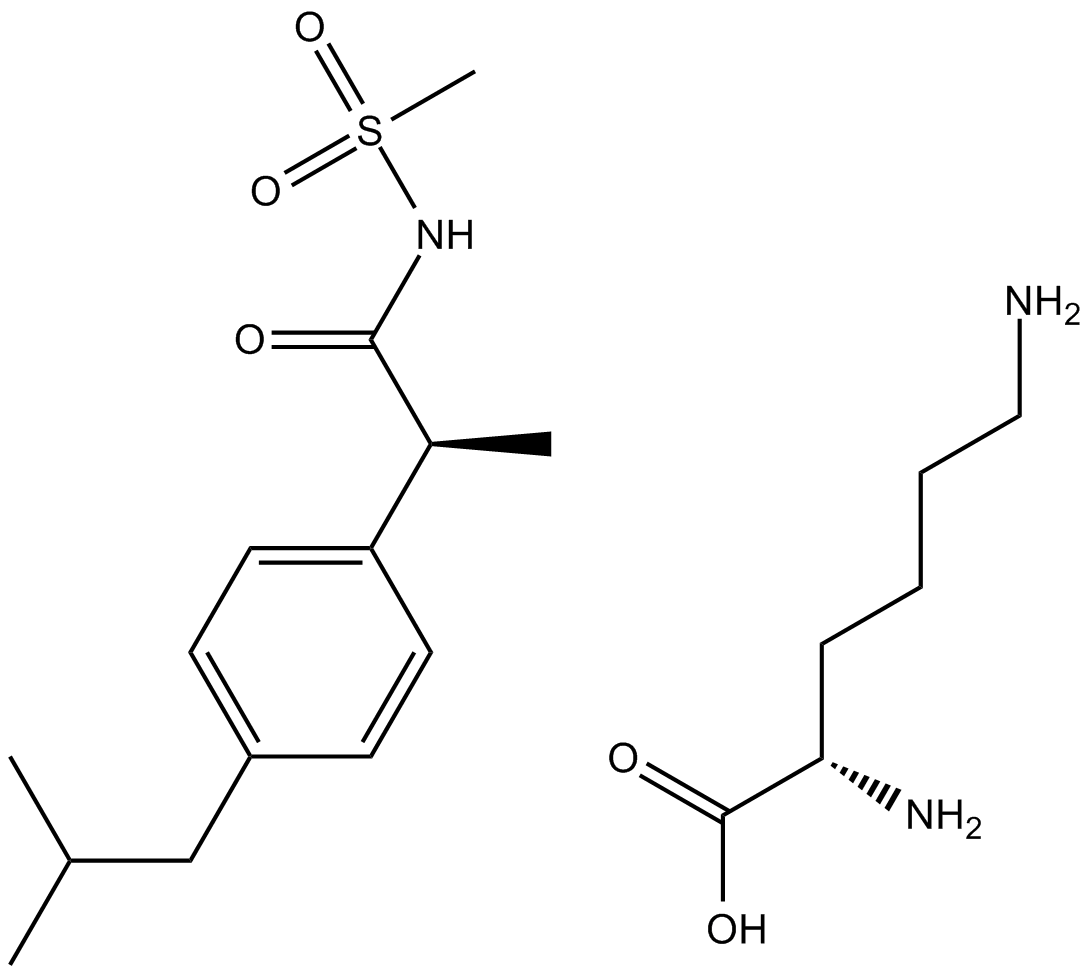
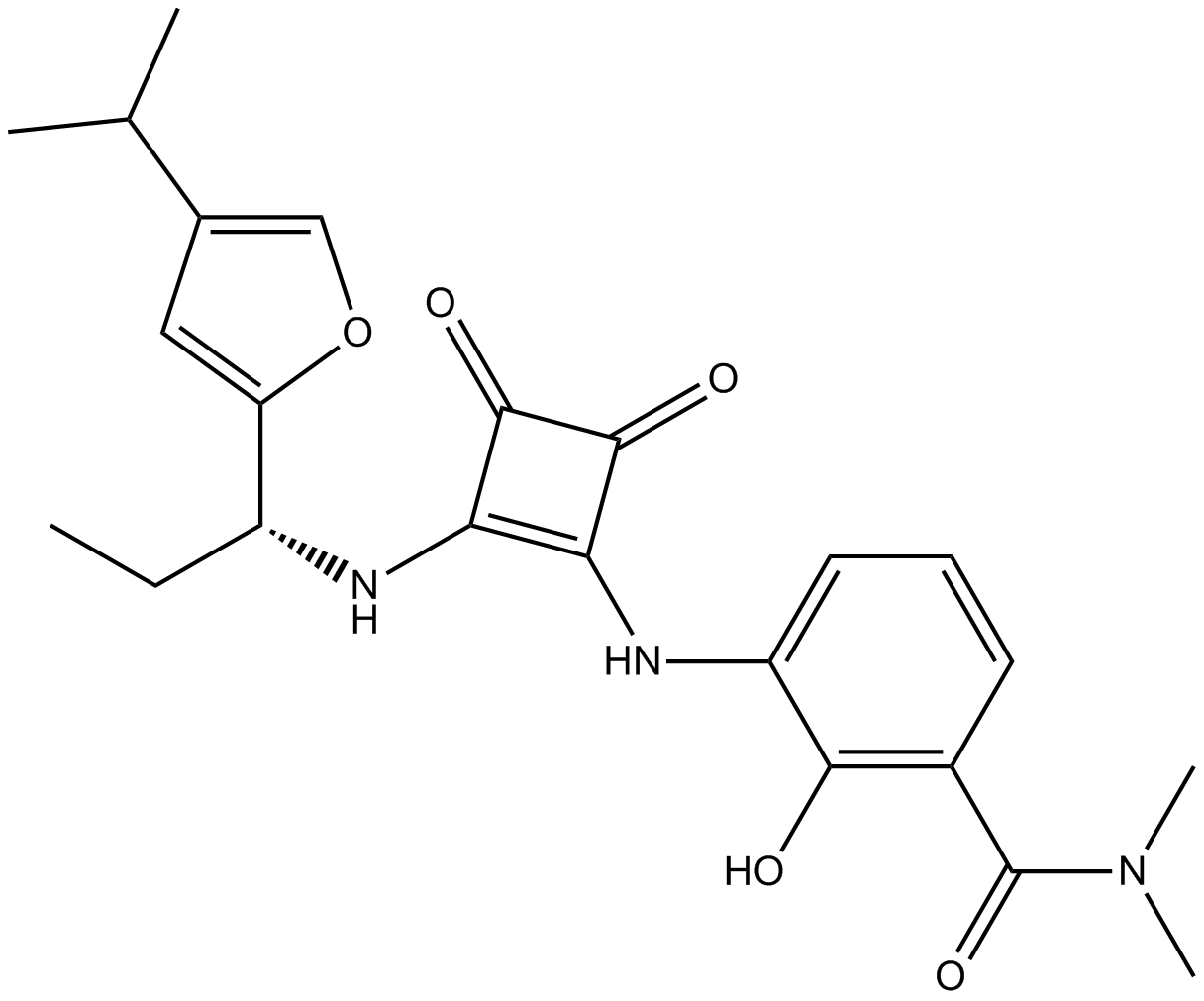
| Chemical Name | (2S)-2,6-diaminohexanoic acid;(2R)-2-[4-(2-methylpropyl)phenyl]-N-methylsulfonylpropanamide |
| Canonical SMILES | CC(C)Cc1ccc(C(C)C(NS(C)(=O)=O)=O)cc1.NCCCC[C@@H](C(O)=O)N |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Binding assays |
Isolated PMNs (107×mL) were resuspended in RPMI 1640 and incubated at 37℃ for 15 min in the presence of repertaxin (1 mM) or vehicle. After incubation cells were resuspended (2×107/mL) in binding medium (RPMI 1640 containing 10 mg/ml BSA, 20 mM HEPES, and 0.02% NaN3) in the presence of repertaxin or vehicle. Aliquots of 0.2 nM of [125I] CXCL8 and serial dilutions of unlabeled CXCL8 were added to 106 cells in 100 μL of binding medium and incubated at room temperature for 1 hr under gentle agitation. Unbound radioactivity was separated from cell-bound radioactivity by centrifugation through anoil gradient (80% silicon and 20% paraffin) on a microcentrifuge. Nonspecific binding was determined by a 200-fold molar excess of unlabeled CXCL8. Scatchard analysis was performed with the LIGAND program. |
| Cell experiment [1]: | |
|
Cell lines |
Human polymorphonuclear cells (PMN) and monocytes and rodent peritoneal PMN. |
|
Preparation method |
Soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reaction Conditions |
45 min (human PMN), 1 h (rodent PMN), or 2 h (monocytes). |
|
Applications |
Repertaxin inhibits human PMN migration induced by CXCL8 and CXCL1 with IC50 values of 1 nM and 400 nM respectively, which are mediated by CXCR1 and CXCR2, respectively. Repertaxin also inhibits rodent PMN chemotaxis induced by CXCL1 and CXCL2. |
| Animal experiment [1]: | |
|
Animal models |
Rat model of liver postischaemia RI. |
|
Dosage form |
3, 15, or 30 mg/kg; 15 min before reperfusion (i.v.) and 2 h after reperfusion (s.c.). |
|
Applications |
Repertaxin (15 mg/kg) inhibits PMN recruitment into reperfused livers by 90% and significantly reduces liver damage. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Bertini R, Allegretti M, Bizzarri C, et al. Noncompetitive allosteric inhibitors of the inflammatory chemokine receptors CXCR1 and CXCR2: prevention of reperfusion injury. Proc Natl Acad Sci U S A, 2004, 101(32): 11791-11796. |
|
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data