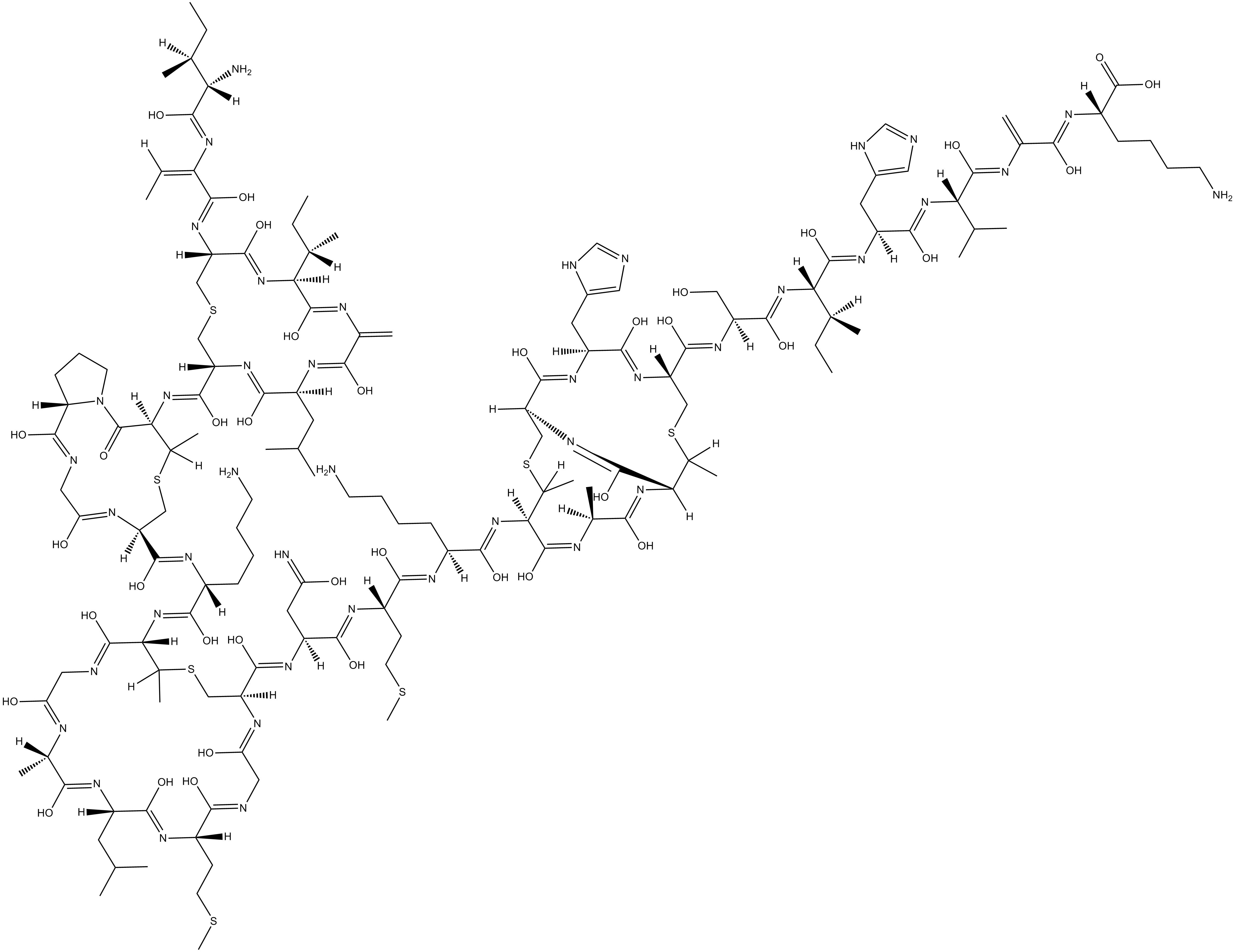
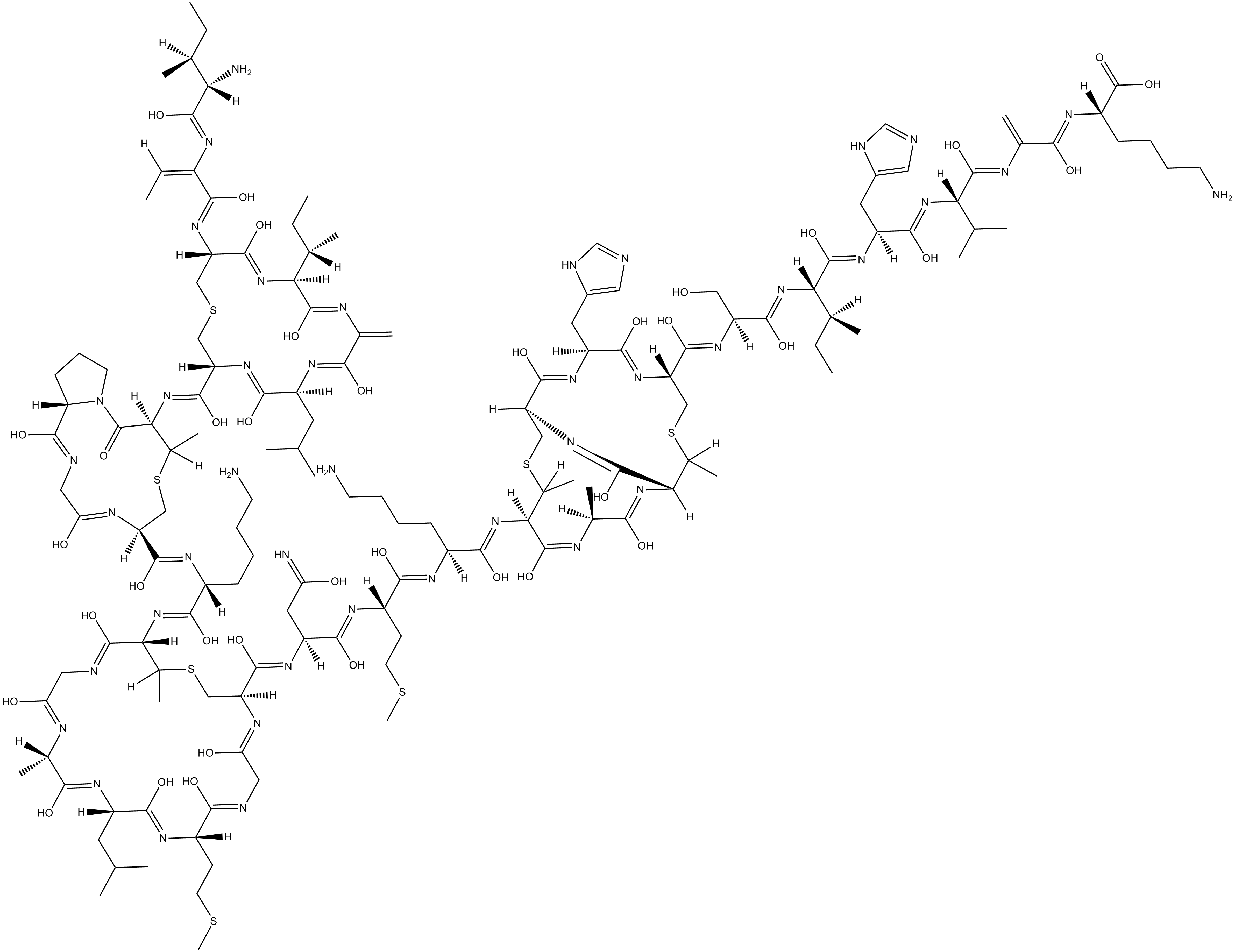
Nisin
Nisin is an antibacterial peptide.
Nisin is produced by the lactic acid bacterium L. lactis using uncommon amino acids, including lanthionine, and is a member of the class of antibiotics referred to as lantibiotics.
In vitro: Previous study showed that the sperm motility could be completely inhibited with nisin The minimum effective concentration of nisin required to immobilize sperm in vitro within 20 s was found to be 50 μg in rat, 200 μg in rabbit and 300-400 μg in monkey and human. Such inhibitory effect on sperm motility was found to be dose- and time-dependent [1].
In vivo: Intravaginal administration of nisin before mating during proestrus-estrous transition phase caused complete arrest of sperm motility and blockage of conception. Subacute toxicity studies in rats showed that, repetitive intravaginal application of nisin at the dose of 200 microg for 14 days caused no abnormalities either in the length of estrous cycle or in the morphology of vaginal epithelial cells. Moreover, no histopathological abnormalities in vaginal tissue or any change in blood and serum biochemical profiles were seen. In addition, no adverse effects were found on subsequent reproductive performance, development of pups and neonate survival [1].
Clinical trial: Up to now, nisin is still in the preclinical development stage.
Reference:
[1] Aranha C, Gupta S, Reddy KV. Contraceptive efficacy of antimicrobial peptide Nisin: in vitro and in vivo studies. Contraception. 2004 Apr;69(4):333-8.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 3354.1 |
| Cas No. | 1414-45-5 |
| Formula | C143H230N42O37S7 |
| Synonyms | Ambicin N|Nisaplin® |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥5.63 mg/mL in DMSO with gentle warming and ultrasonic |
| Chemical Name | L-isoleucyl-(Z)-2,3-didehydro-2-aminobutanoyl-D-cysteinyl-L- isoleucyl-2,3-didehydroalanyl-L-leucyl-L-cysteinyl-threo-3- mercapto-D-2-aminobutanoyl-L-prolylglycyl-L-cysteinyl-L-lysyl- threo-3-mercapto-D-2-aminobutanoylglycyl-L-alanyl-L-leucyl-L- methionyl |
| Canonical SMILES | O=C(N1[C@@]([H])(CCC1)C(NC2)=O)[C@@H](NC([C@@H](NC([C@H](CC(C)C)NC(C(N3)=C)=O)=O)CSC[C@@H](NC(/C(NC([C@H]([C@H](CC)C)N)=O)=C/C)=O)C(N[C@]([H])(C3=O)[C@H](CC)C)=O)=O)[C@H](C)SC[C@@H](C(N[C@H](C(N[C@@H]([C@H](C)SC[C@@H](C(N[C@H](C(N[C@H](C(N[C@H](C(N[C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
human HNSCC cell lines |
|
Preparation method |
The solubility of this compound in DMSO is ≤ 0.25mg/ml. General tips for obtaining a higher concentration: Please warm the tube at 37 ° C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20 ° C for several months. |
|
Reacting condition |
5, 10, 20, 40 and 80 μg/ml |
|
Applications |
Treatment of three different HNSCC cell lines with higher concentrations of nisin (5, 10, 20, 40, and 80 μg/mL) for 24 hours resulted in increased levels of DNA fragmentation or apoptosis. |
| Animal experiment [1]: | |
|
Animal models |
Oral cancer mouse model |
|
Dosage form |
200 mg/kg, intragastrical administration, once a day for three weeks |
|
Application |
Compared with the control group, the tumor volume of mice treated with nisin decreased statistically. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Joo NE, Ritchie K, Kamarajan P, Miao D, Kapila YL. Nisin, an apoptogenic bacteriocin and food preservative, attenuates HNSCC tumorigenesis via CHAC1. Cancer Med. 2012 Dec;1(3):295-305. doi: 10.1002/cam4.35. Epub 2012 Oct 2. PubMed PMID: 23342279; PubMed Central PMCID: PMC3544465 |
|
Chemical structure