LY2857785
LY2857785 was identified as a type I reversible and competitive ATP kinase inhibitor against CDK9 and other transcription kinases CDK8 and CDK7.[1,2]
Cdk7 acts as a Cdk-activating kinase and regulate transcription. Cdk8 and Cdk9 regulate transcription via phosphorylation of the RNA polymerase II carboxyl terminal domain. The Cdk9-related pathway performs an important role in several biological processes, such as cell growth, proliferation, protection from apoptosis and differentiation. These kinases are reported dys-regulation in some cancers. [2]
Transcription activation requires phosphorylation of a carboxyl-terminal domain, by a variety of kinases including CDK7, CDK8, and CDK9. By inhibiting these kinases, LY2857785 has unique transcription inhibitor activity. LY2857785 dramatically inhibited XIAP protein level in MV-4-11 and other hematologic cancer cells. It also can significantly reduce RNAP II CTD phosphorylation and dramatically decreases MCL1 protein levels to result in apoptosis in a variety of leukemia and solid tumor cell lines. [1]
LY2857785 potently inhibits a carboxyl-terminal domain phosphorylation and exhibits antitumor efficacy in orthotopic models of leukemia. LY2857785 inhibits the growth of leukemia cells, including orthotopic leukemia preclinical models as well as in ex vivo acute myeloid leukemia and chronic lymphocytic leukemia patient tumor samples. LY2857785 may be used in treating patients with hematologic tumors, particularly AML and CLL. [1]
References:
[1] Yin T, Lallena MJ, Kreklau EL etal. , A novel CDK9 inhibitor shows potent antitumor efficacy in preclinical hematologic tumor models. Mol Cancer Ther. 2014 Jun;13(6):1442-56.
[2] Romano G1, Giordano A. Role of the cyclin-dependent kinase 9-related pathway in mammalian gene expression and human diseases. Cell Cycle. 2008 Dec;7(23):3664-8.
| Storage | Desiccate at -20°C |
| M.Wt | 448.6 |
| Cas No. | 1619903-54-6 |
| Formula | C26H36N6O |
| Solubility | insoluble in H2O; ≥13.87 mg/mL in EtOH with gentle warming; ≥2.63 mg/mL in DMSO with gentle warming and ultrasonic |
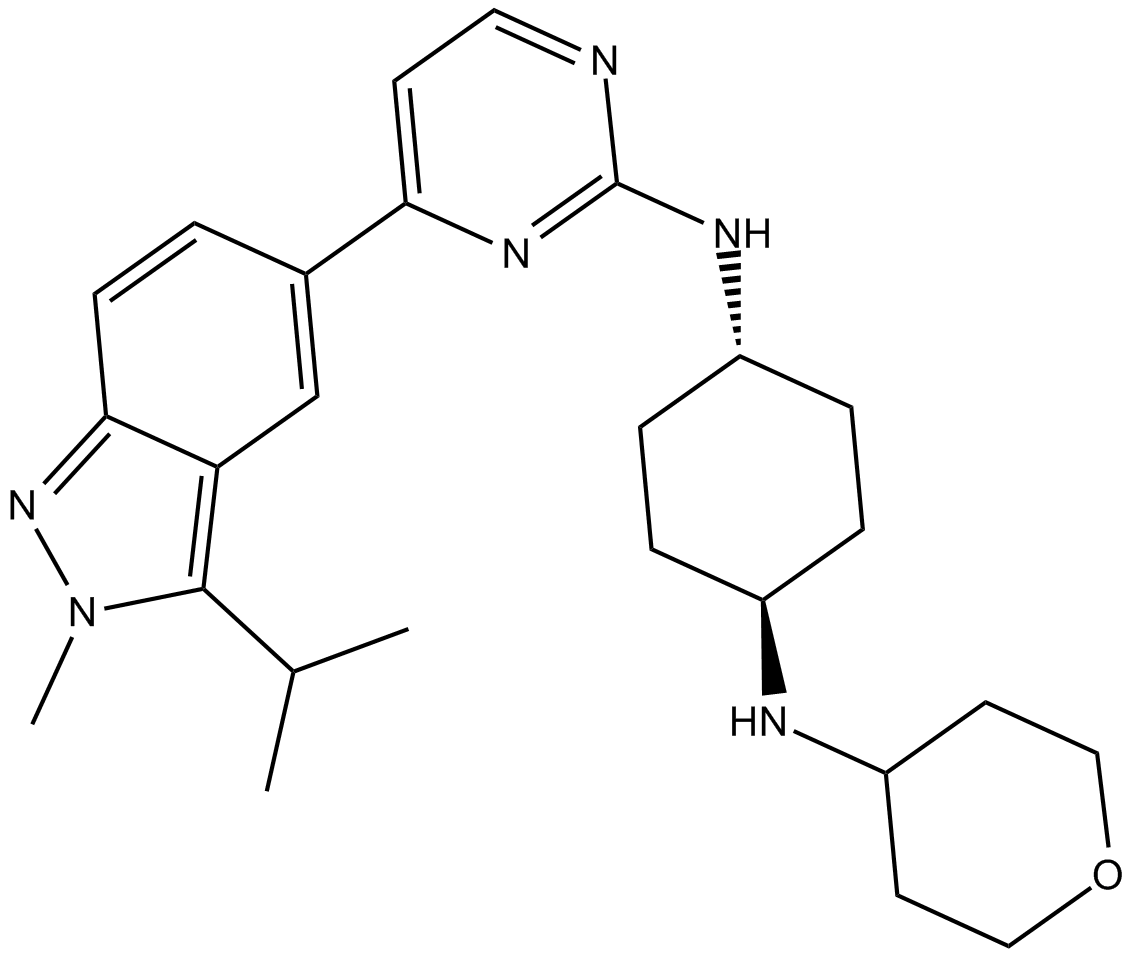
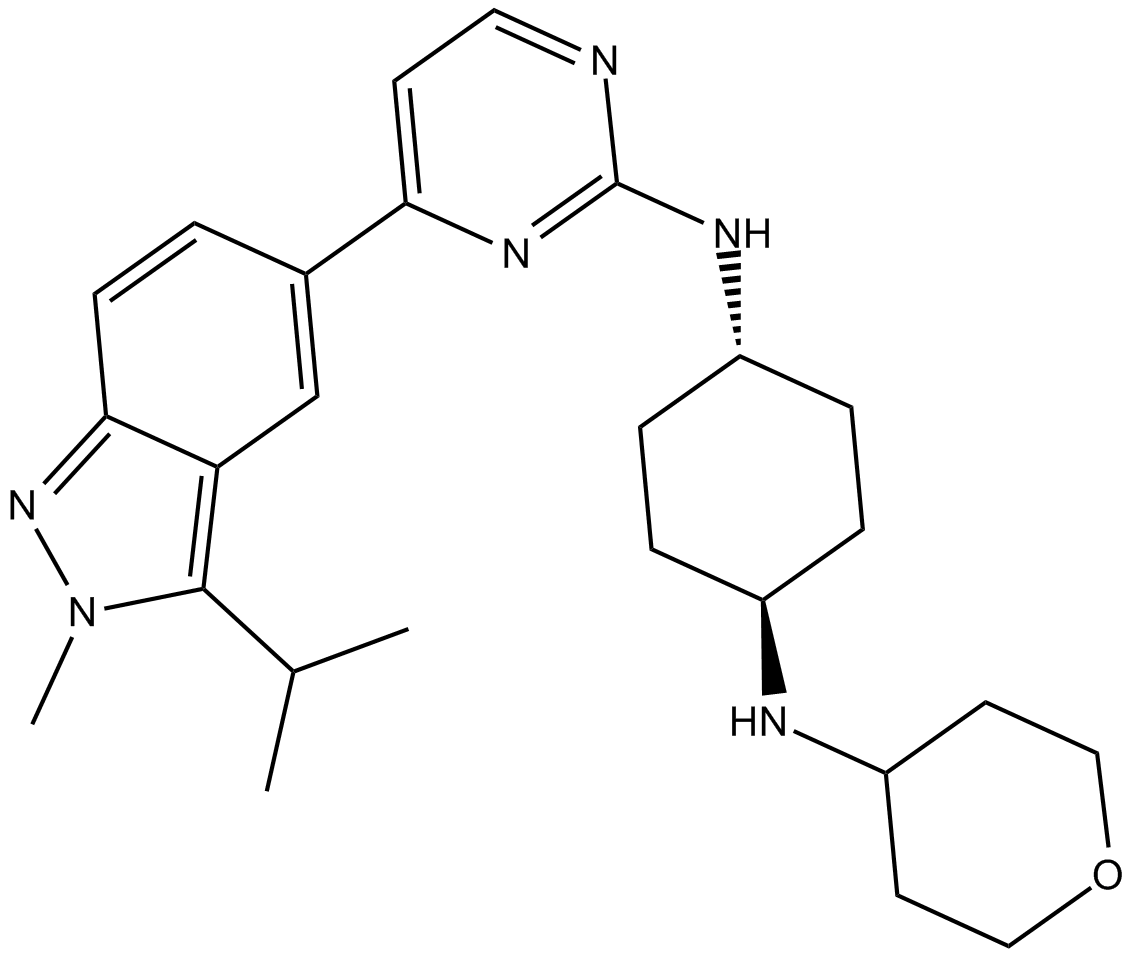
| Chemical Name | (1r,4r)-N1-(4-(3-isopropyl-2-methyl-2H-indazol-5-yl)pyrimidin-2-yl)-N4-(tetrahydro-2H-pyran-4-yl)cyclohexane-1,4-diamine |
| Canonical SMILES | CC(C)c1c(cc(cc2)-c3ccnc(N[C@H](CC4)CC[C@@H]4NC4CCOCC4)n3)c2n[n]1C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Binding assays |
CDK7 and CDK9 reaction mixtures contained 10 mmol/L Tris-HCl (pH 7.4), 10 mmol/L HEPES, 5 mmol/L DTT, 10 μmol/L ATP, 0.5 μCi 33p-ATP, 10 mmol/L MnCl2, 150 mmol/L NaCl, 0.01% Triton X-100, 2% dimethylsulfoxide (DMSO), 0.05 mmol/L CDK7/9ptide, and 2 nmol/L CDK7/Mat1/cyclin H (14-476M, Upstate), or 2 nmol/L CDK9/cyclin T1 (14-685M, Upstate), respectively. CDK8/cyclin C reaction is performed in HEPES 30 mmol/L, DTT 2 mmol/L, MgCl2 5 mmol/L, 0.015% Triton X-100, 5 μmol/L ATP, and 400 nmol/L of RBER-CHKStide containing 20 nmol/L of enzyme.Compound in DMSO was diluted serially 1:3 for dose response. Reactions were carried out in 96-well polystyrene plates. The reactions were incubated at room temperature for 60 minutes and followed by termination with 10% H3PO4 or 10% trichloroacetic acid (TCA). For the filter binding assay, reactions were transferred to 96-well filter plates and measured by Microbeta scintillation counter. |
| Cell experiment [1]: | |
|
Cell lines |
Human bone marrow myeloid progenitor cells |
|
Preparation method |
This compound is soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
Cells were treated with varying concentrations of compound for varying durations ranging from 4 to 24 hours. |
|
Applications |
LY2857785 inhibited hematologic tumor cell (human bone marrow myeloid progenitor cells) proliferation. |
| Animal experiment [1]: | |
|
Animal models |
Human cancer cells U87MG, MV-4-11, A375, and HCT116 xenograft rat models |
|
Dosage form |
4, 8, 18 mg/kg. i.v. every 3 days. |
|
Application |
LY2857785 demonstrates potent antitumor growth efficacy in preclinical tumor models (U87MG, MV-4-11, A375, and HCT116 xenograft rat models). |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Yin T, et al. A novel CDK9 inhibitor shows potent antitumor efficacy in preclinical hematologic tumor models. Mol Cancer Ther. 2014 Jun;13(6):1442-56. |
|
Quality Control & MSDS
- View current batch:
Chemical structure