HJC 0350
HJC 0350 is a potent and selective antagonist of EPAC2 with IC50 value of 0.3 μM [1].
cAMP/cAMP regulated guanine nucleotide exchange factor (EPAC/cAMP-GEF) is a guanine nucleotide exchange factor for the small GTPases Rap1 and Rap2 in response to intracellular cAMP. EPAC2 is mainly expressed in the central nervous system, pancreas and adrenal gland [1].
HJC 0350 is a potent and selective EPAC2 antagonist. HJC 0350 competed with 8-NBD-cAMP in binding recombinant fusion protein EPAC2 with IC50 value of 0.3 μM and exhibited 133-fold more potent than cAMP, which competed with 8-NBD-cAMP in binding EPAC2 with IC50 value of 40 μM. In the presence of 25 μM cAMP, HJC 0350 (25 μM) inhibited EPAC2 GEF activity but had no effect on EPAC1-mediated Rap1-GDP exchange activity and cAMP-mediated PKA activation, which suggested that HJC 0350 was EPAC2-specific antagonist. In HEK293 cells expressing EPAC1- or EPAC2-based fluorescence resonance energy transfer (FRET) sensor (EPAC2-FL or EPAC1-FL), HJC 0350 (10 μM) completely inhibited the 007-AM (a membrane permeable EPAC selective cAMP analogue) induced decrease of FRET in HEK293/EPAC2-FL cells but had no effect on HEK293/EPAC1-FL cells [1].
Reference:
[1]. Chen H, Tsalkova T, Chepurny OG, et al. Identification and characterization of small molecules as potent and specific EPAC2 antagonists. J Med Chem, 2013, 56(3): 952-962.
| Storage | Store at -20°C |
| M.Wt | 277.38 |
| Cas No. | 885434-70-8 |
| Formula | C15H19NO2S |
| Solubility | insoluble in H2O; ≥10.84 mg/mL in EtOH with ultrasonic; ≥13.85 mg/mL in DMSO |
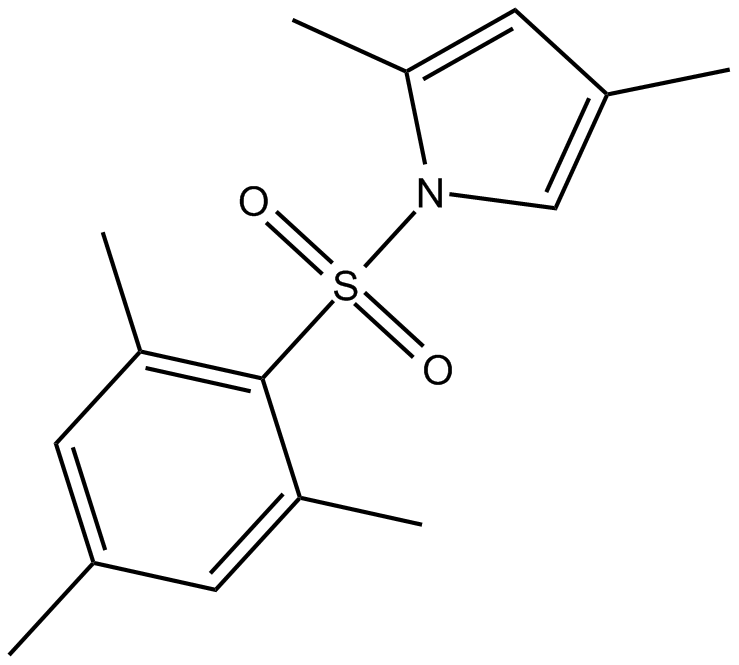
| Chemical Name | 1-(mesitylsulfonyl)-2,4-dimethyl-1H-pyrrole |
| Canonical SMILES | Cc(cc1C)c[n]1S(c1c(C)cc(C)cc1C)(=O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure