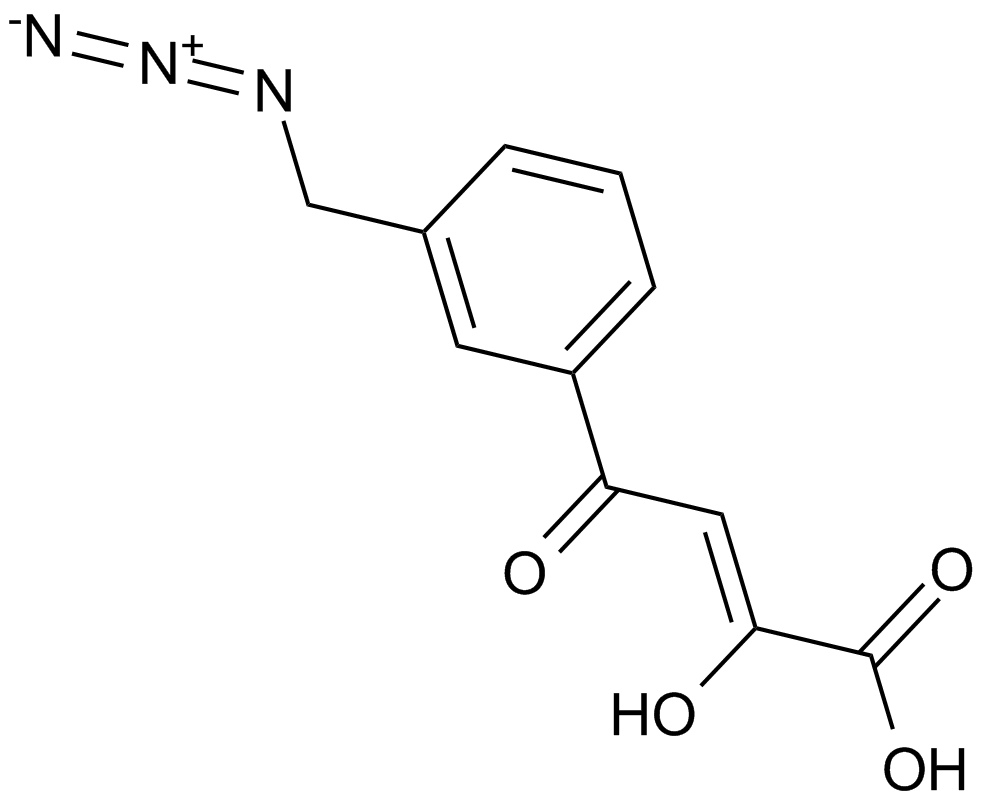
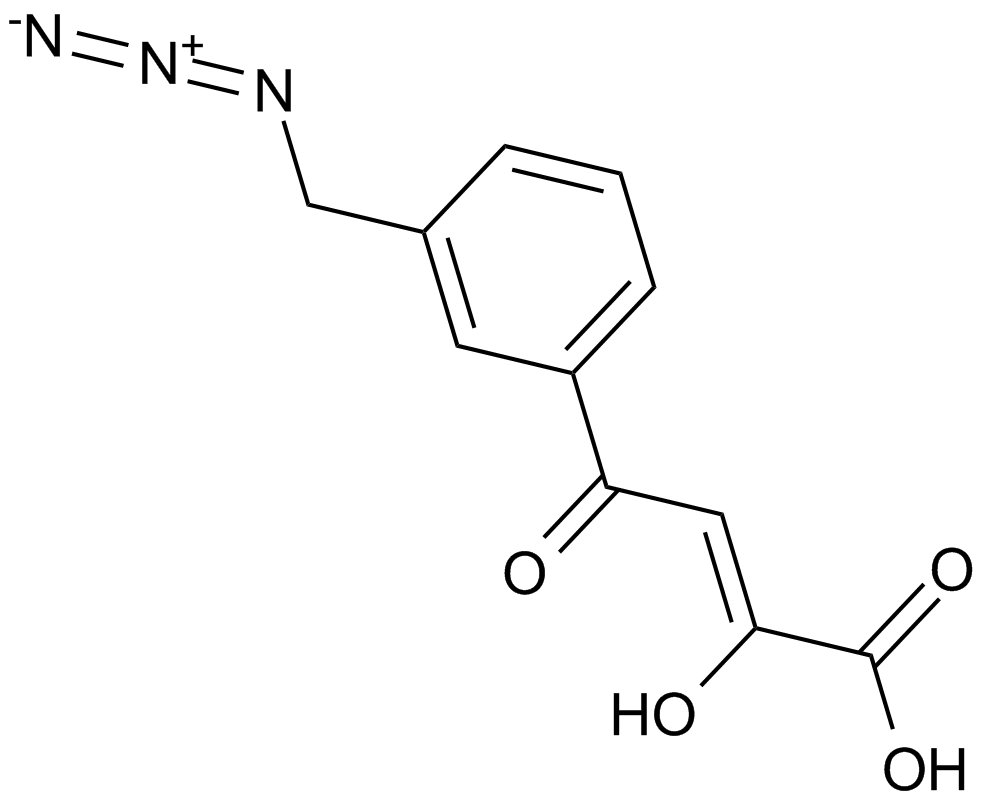
HIV-1 integrase inhibitor is useful for anti-HIV, with IC50 value of 0.33 µM,[1] which can target HIV-1 integrase and depress the activity in the treatment of HIV infection, AIDS, and other similar diseases characterized by integration of a retroviral genome into a host chromosome.
HIV integrase is a 32 kDa protein produced from the C-terminal portion of the Pol gene product, an enzyme produced by HIV that enables its genetic material to be integrated into the DNA of the infected cell [2]which are not to be confused with phage integrases and a key component in the retroviral pre-integration complex (PIC)[3]. HIV-1 integrase is composed of 3 structurally independent, functional domains: the N-terminal domain (NTD), catalytic core domain (CCD) and the C-terminal domain (CTD).The HIV-1 integration occurs through a multistep process that includes two catalytic reactions: 3’endonucleolytic processing of proviral DNA ends (termed 3’processing) and integration of 3’-processed viral DNA into cellular DNA (referred to as strand transfer)[4].
The human immunodeficiency virus (HIV) is the causative agent for the acquired immunodeficiency syndrome (AIDS)[5], then HIV integrase is an attractive target for new anti-HIV drugs. The drug design of HIV-1 integrase inhibitor include integrase strand transfer inhibitors (INSTIs),inhibition of the LEDGF/p75- integrase interaction and integrase binding inhibitors, but strand transfer inhibition is the most intuitively obvious and readily pursued to date.Mg2+ and Mn2+ are critical cofactors in the integration phase, so removing these cofactors (e.g. through chelation) causes functional impairment of integrase[6].Competitive inhibitors compete directly with viral DNA for binding to integrase in order to inhibit 3‘-end processing.[7] In doing this the inhibitors completely block the active site from binding to target DNA.INSTIs bind tightly and specifically to the IN that is associated with the ends of the DNA by chelating the divalent metal ions (Mg2+) which is coordinated by the catalytic triad, such as the DDE motif which is located in the CCD and is the active site of the enzyme[8].
Development of a successful INSTI treatment was accomplished when raltegravir was discovered by Merck Sharp & Dohme Limited.[9] S/GSK1349572 is an integrase inhibitor discovered by ViiV/Shinongi which was entering phase three in clinical trials in 2011. This new drug is promising and seems to be well tolerated and so far shows better results than both raltegravir and elvitegravir.[10]
References:
1.Loizidou EZ et al. Analysis of binding parameters of HIV-1 integrase inhibitors: correlates of drug inhibition and resistance. Bioorg Med Chem. 2009, 17(13):4806-18.
2.Cocohoba, J; Dong, BJ. "Raltegravir: the first HIV integrase inhibitor". Clinical therapeutics.2008, 30(10): 1747–65.
3.Mouscadet, JF; Delelis, O; Marcelin, AG; Tchertanov, L. "Resistance to HIV-1 integrase inhibitors: A structural perspective". Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy.2010, 13(4-5):139–50.
4.Fan, X; Zhang, FH.et al."Design of HIV-1 integrase inhibitors targeting the catalytic domain as well as its interaction with LEDGF/p75: a scaffold hopping approach using salicylate and catechol groups". Bioorganic & Medicinal Chemistry.2011,19 (16): 4935–52.
5.Pommier, Yves.et al. "Integrase inhibitors to treat HIV/Aids". Nature Reviews Drug Discovery.2005, 4 (3): 236–248.
6.Pendri, A.et al. "New first and second generation inhibitors of human immunodeficiency virus-1 integrase". Expert opinion on therapeutic patents. 2011,21 (8): 1173–89.
7.Chen, X; Tsiang, M, Yu, F, Hung, M, Jones, GS, Zeynalzadegan, A, Qi, X, Jin, H, Kim, CU, Swaminathan, S, Chen, JM. "Modeling, analysis, and validation of a novel HIV integrase structure provide insights into the binding modes of potent integrase inhibitors". Journal of Molecular Biology. 2008, 380 (3): 504–19.
8.Mouscadet, JF. et al."Resistance to HIV-1 integrase inhibitors: A structural perspective". Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy.2010,13(4-5):139–50.
9.McColl, DJ; Chen, X. "Strand transfer inhibitors of HIV-1 integrase: bringing IN a new era of antiretroviral therapy". Antiviral Research,. 2010,85 (1): 101–18.
10.Barnhart, Matthew,James Shelton."A better state of ART improving antiretroviral regimens to increase global access to HIV treatment". Journal of AIDS and HIV Research. 2011, 3 (4): 71–78.