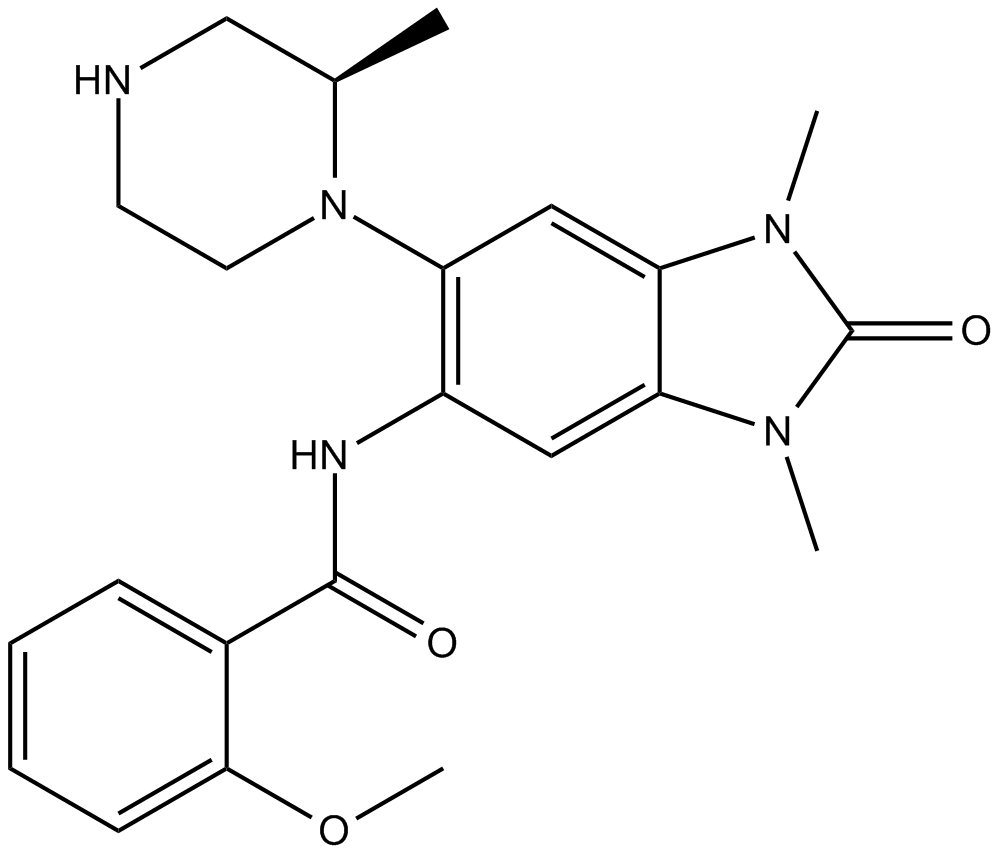
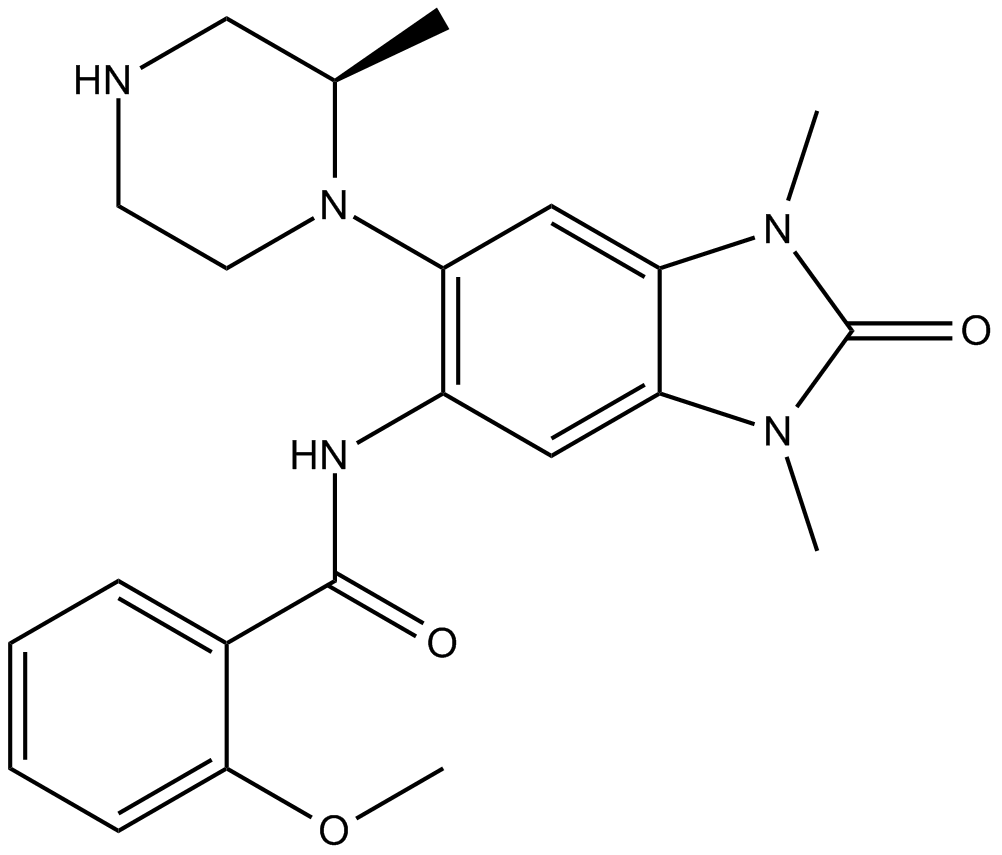
GSK6853
pIC50: 8.1
GSK6853 is a BRPF1 inhibitor.
The BRPF (Bromodomain and PHD Fingercontaining) protein family are key scaffolding proteins for assembly of MYST histone acetyltransferase complexes.
In vitro: In previous study, GSK6853 was tested in the BROMOscan panel of bromodomain binding assays and the results showed that consistent with the previous findings, it showed excellent BRPF1 potency (pKd 9.5) and greater than 1600-fold selectivity over all other bromodomains tested. In addition, screening GSK6853 against a panel of 48 unrelated assays revealed only off-target activities that were relatively weak compared to its high BRPF1 potency [1].
In vivo: To assess the suitability of GSK6853 for in vivo studies, its DMPK characteristics were studied in male CD1 mice by i.v., po, and i.p. administration. The results indicated that the i.p. route of administration would be suitable for dosing this molecule in further PK/PD models. Moreover, in order to be able to compare biochemical potency measurements to free blood concentrations, the fraction unbound in the CD1 mouse was also tested with a resulting value of 7.9% [1].
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Bamborough, P. ,Barnett, H.A.,Becher, I., et al. GSK6853, a chemical probe for inhibition of the BRPF1 bromodomain. ACS Med. Chem. Lett. 7(6), 552-557 (2016).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 409.5 |
| Cas No. | 1910124-24-1 |
| Formula | C22H27N5O3 |
| Solubility | ≤30mg/ml in ethanol;25mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | N-[2,3-dihydro-1,3-dimethyl-6-[(2R)-2-methyl-1-piperazinyl]-2-oxo-1H-benzimidazol-5-yl]-2-methoxy-benzamide |
| Canonical SMILES | C[C@H](CNCC1)N1c(c(NC(c(cccc1)c1OC)=O)c1)cc(N2C)c1N(C)C2=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure