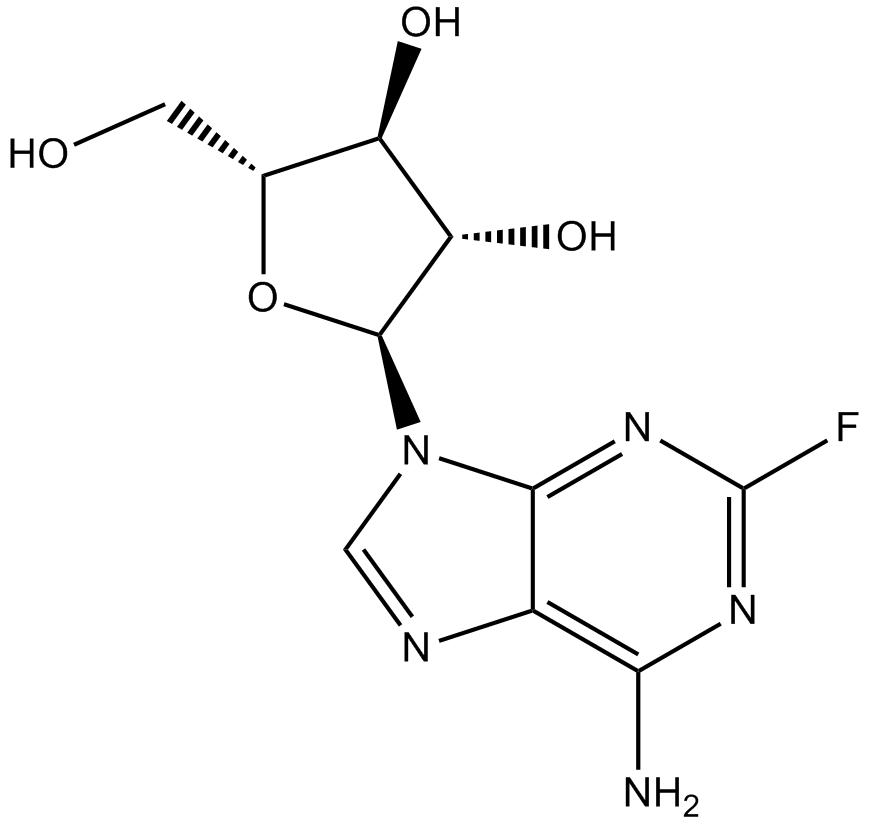
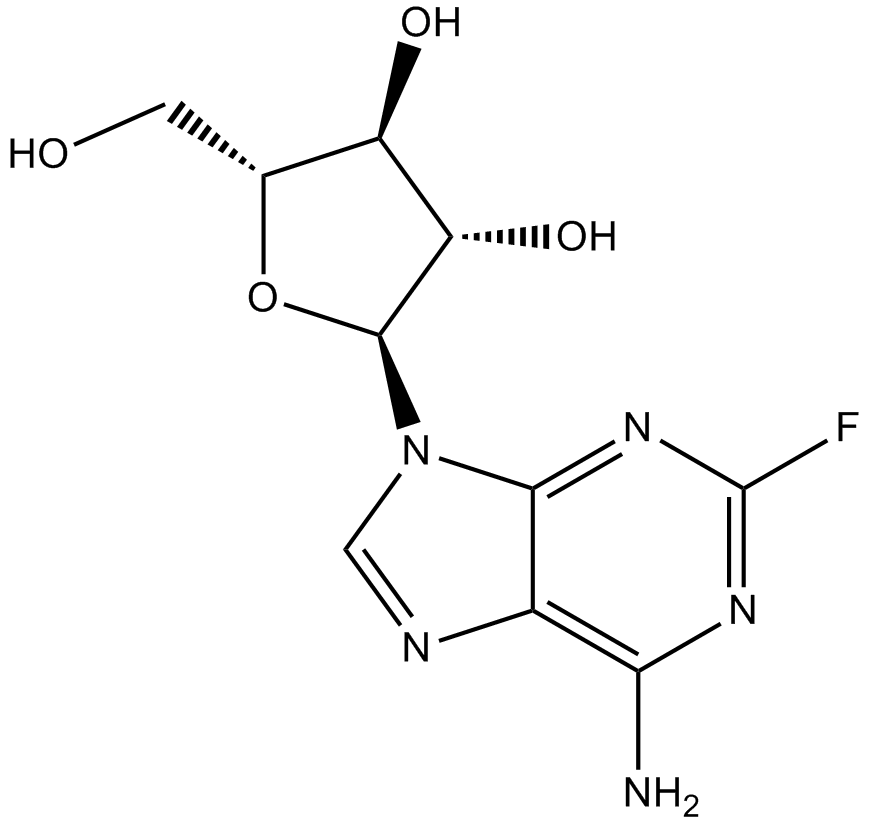
Fludarabine
Fludarabine (CAS 21679-14-1) is a purine analog prodrug that inhibits DNA synthesis. Upon cellular uptake, it undergoes phosphorylation to its active triphosphate form (F-ara-ATP), disrupting DNA replication through inhibition of critical enzymes such as DNA primase, DNA ligase I, ribonucleotide reductase, and DNA polymerases δ and ε. In human myeloma RPMI8226 cell assays, fludarabine suppressed cell growth, reduced Akt phosphorylation, and lowered anti-apoptotic proteins XIAP and Survivin. In vivo studies demonstrated marked tumor growth inhibition in RPMI8226 xenograft models. Fludarabine serves as a tool in oncology research, particularly leukemia and multiple myeloma-related studies.
- 1. Shira Sagie, Tomer Babu, Chen Weller. "Lymphodepleting chemotherapy potentiates neoantigen-directed T cell therapy by enhancing antigen presentation." Cell Rep Med. 2025 Dec 16;6(12):102506 PMID: 41406943
- 2. Madeline Dennis, Alyssa Hurley, et al. "Her2 amplification, Rel-A, and Bach1 can influence APOBEC3A expression in breast cancer cells." PLoS Genet. 2024 May 28;20(5):e1011293 PMID: 38805570
- 3. Haoshen Feng, Menglu Li, et al. "Cigarette smoke extracts induce apoptosis in Raw264. 7 cells via endoplasmic reticulum stress and the intracellular Ca2+/P38/STAT1 pathway." Toxicol In Vitro. 2021 Dec;77:105249 PMID: 34560245
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 285.23 |
| Cas No. | 21679-14-1 |
| Formula | C10H12FN5O4 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥9.25 mg/mL in DMSO |
| Chemical Name | (2R,3S,4S,5R)-2-(6-amino-2-fluoropurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol |
| Canonical SMILES | Nc1c2nc[n]([C@@H]([C@H]3O)O[C@H](CO)[C@H]3O)c2nc(F)n1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
RPMI 8226 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 9.3 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
1, 2 or 4 μg/mL; 6, 12 or 24 hrs |
|
Applications |
Fludarabine potently inhibited the proliferation of RPMI 8226 cells in dose- and time-dependent manners, with an IC50 value of 1.54 μg/mL. Fludarabine arrested RPMI 8226 cells in the G1 phase of cell cycle and triggered apoptosis. The immunoblotting results showed that Fludarabine time-dependently induced cleavage of caspase-8, -9, -3 and -7, followed by PARP cleavage. In addition, Fludarabine time-dependently up-regulated Bax expression, without affecting Bak expression. |
| Animal experiment [1]: | |
|
Animal models |
SCID mice bearing RPMI 8226 cells |
|
Dosage form |
40 mg/kg; i.p. |
|
Applications |
Tumors of mice treated with PBS grew rapidly, to approximately 10 folds of their initial volume in the 25th day, whereas, tumors in the Fludarabine treatment group increased less than 5 folds. In SCID mice bearing RPMI 8226 cells, treatment with 40 mg/kg Fludarabine for 10 days resulted in a significant increase in the number of apoptotic cells. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Meng H, Yang C, Ni W, et al. Antitumor activity of fludarabine against human multiple myeloma in vitro and in vivo. Eur J Haematol, 2007, 79(6): 486-493. | |
Quality Control & MSDS
- View current batch:
Chemical structure
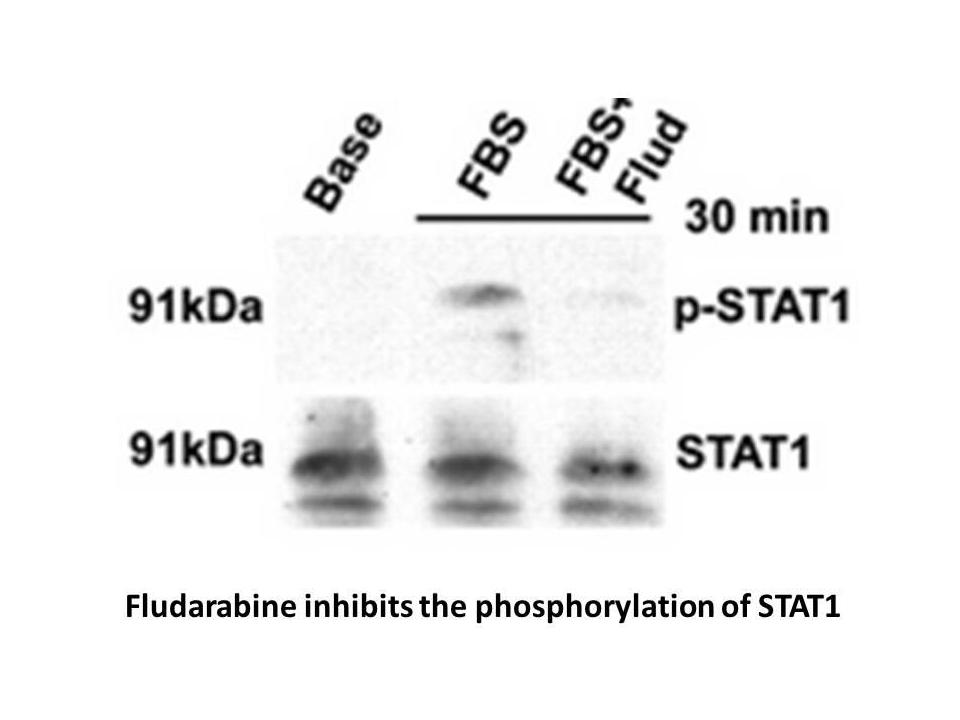
Related Biological Data
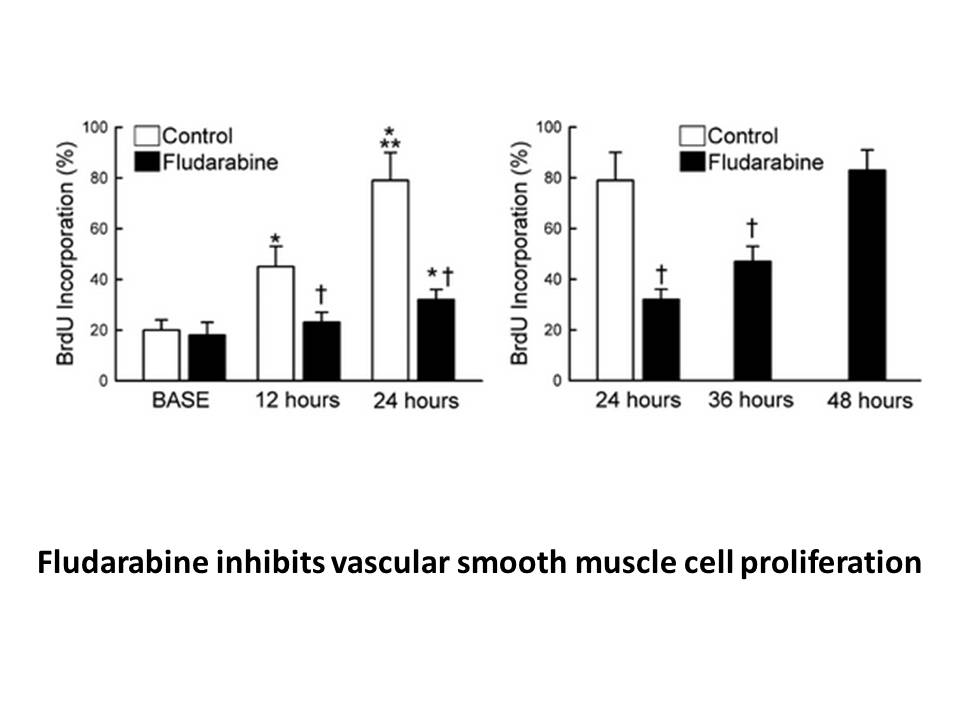
Related Biological Data