CCG-100602
IC50: 9.8 μM for PC-3 prostate cancer cells
CCG-100602 is a Rho pathway inhibitor.
Rho, a member of the Ras superfamily of small GTP-binding proteins, plays a key role in various biological processes including microtubule dynamics, gene transcription, actin cytoskeleton organization, cell cycle progression, oncogenic transformation, as well as epithelial wound repair.
In vitro: CCG-100602 was developed as a CCG-1423 analog for improved selectivity, potency, and attenuated cytotoxicity. It was found that CCG-100602 was able to inhibit RhoA/C-mediated and SRF-driven luciferase expression in PC-3 prostate cancer cells. At 100 μM, CCG-100602 showed 72% inhibition of PC-3 cell invasion into a Matrigel model of metastasis, having an superior efficacy-toxicity profile to that of CCG-1423 [1].
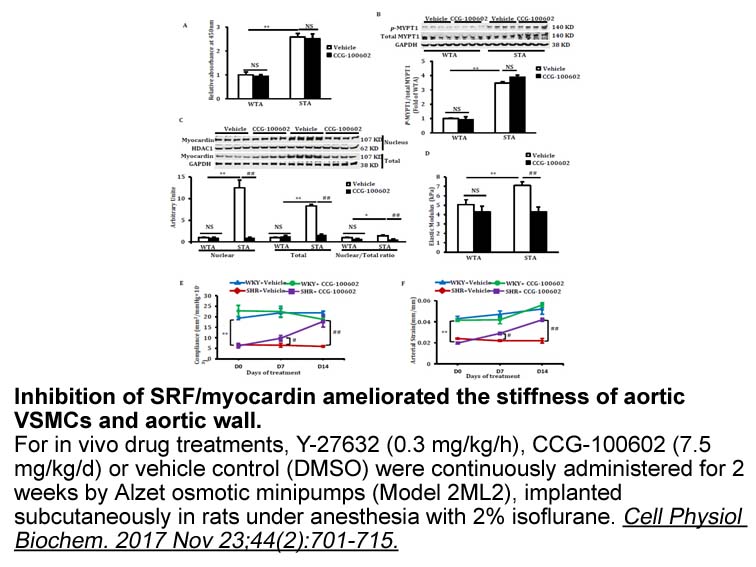
In vivo: To evaluate whether inhibition of SRF could protect podocytes from hyperglycaemia injury, daily ip administration of CCG-1423 was performed in DM rata. Results showed that CCG-1423 could ameliorate proteinuria dose-dependently. CCG-1423 at 0.02 mg/kg could significantly reduce the body weight, compared with the vehicle controls. In addition, the inhibition of SRF with CCG-1423 also significantly abrogated the reduction of synaptopodin expression and the induction of SRF,α-SMA, FSP-1 expression in renal cortex tissues [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Evelyn, C. R.,Bell, J.L.,Ryu, J.G., et al. Design, synthesis and prostate cancer cell-based studies of analogs of the Rho/MKL1 transcriptional pathway inhibitor, CCG-1423. Bioorganic & Medicinal Chemistry Letters 20, 665-672 (2010).
[2] Zhao L, Wang X, Sun L, Nie H, Liu X, Chen Z, Guan G. Critical role of serum response factor in podocyte epithelial-mesenchymal transition of diabetic nephropathy. Diab Vasc Dis Res. 2016 Jan;13(1):81-92.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 478.8 |
| Cas No. | 1207113-88-9 |
| Formula | C21H17ClF6N2O2 |
| Solubility | ≥152.4 mg/mL in DMSO; ≥26.9 mg/mL in EtOH; insoluble in H2O |
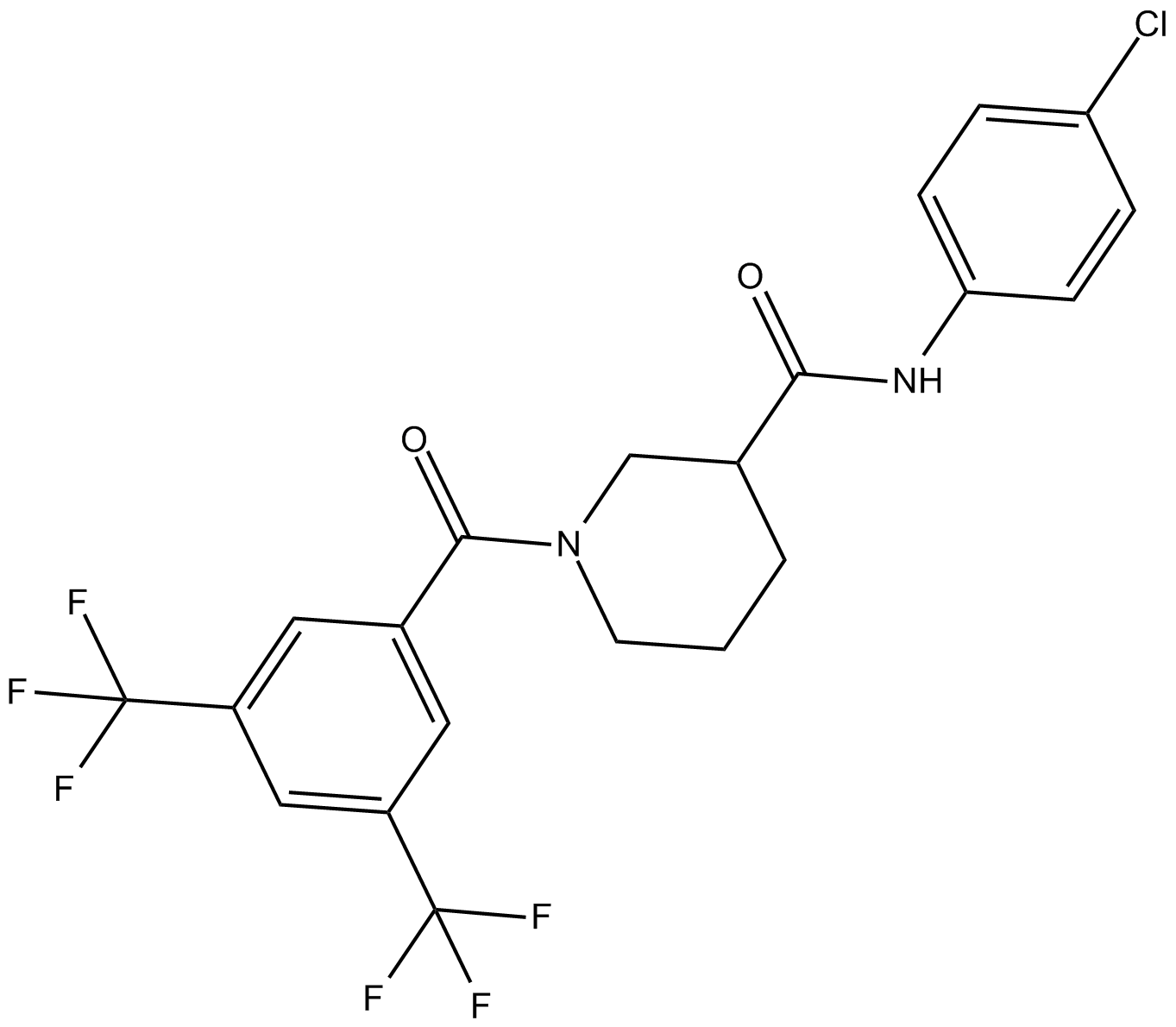
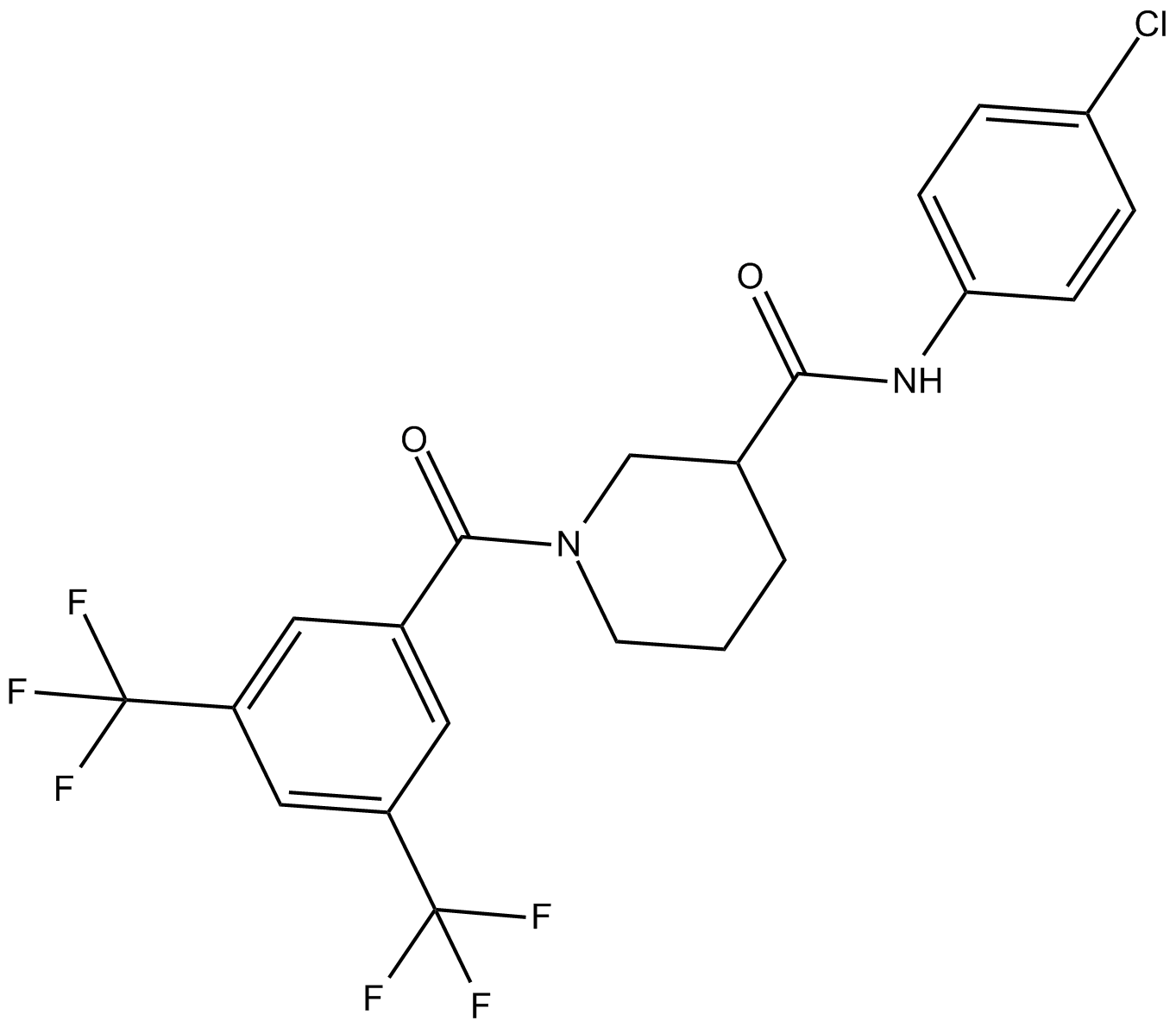
| Chemical Name | 1-[3,5-bis(trifluoromethyl)benzoyl]-N-(4-chlorophenyl)-3-piperidinecarboxamide |
| Canonical SMILES | O=C(C(CCC1)CN1C(c1cc(C(F)(F)F)cc(C(F)(F)F)c1)=O)Nc(cc1)ccc1Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data