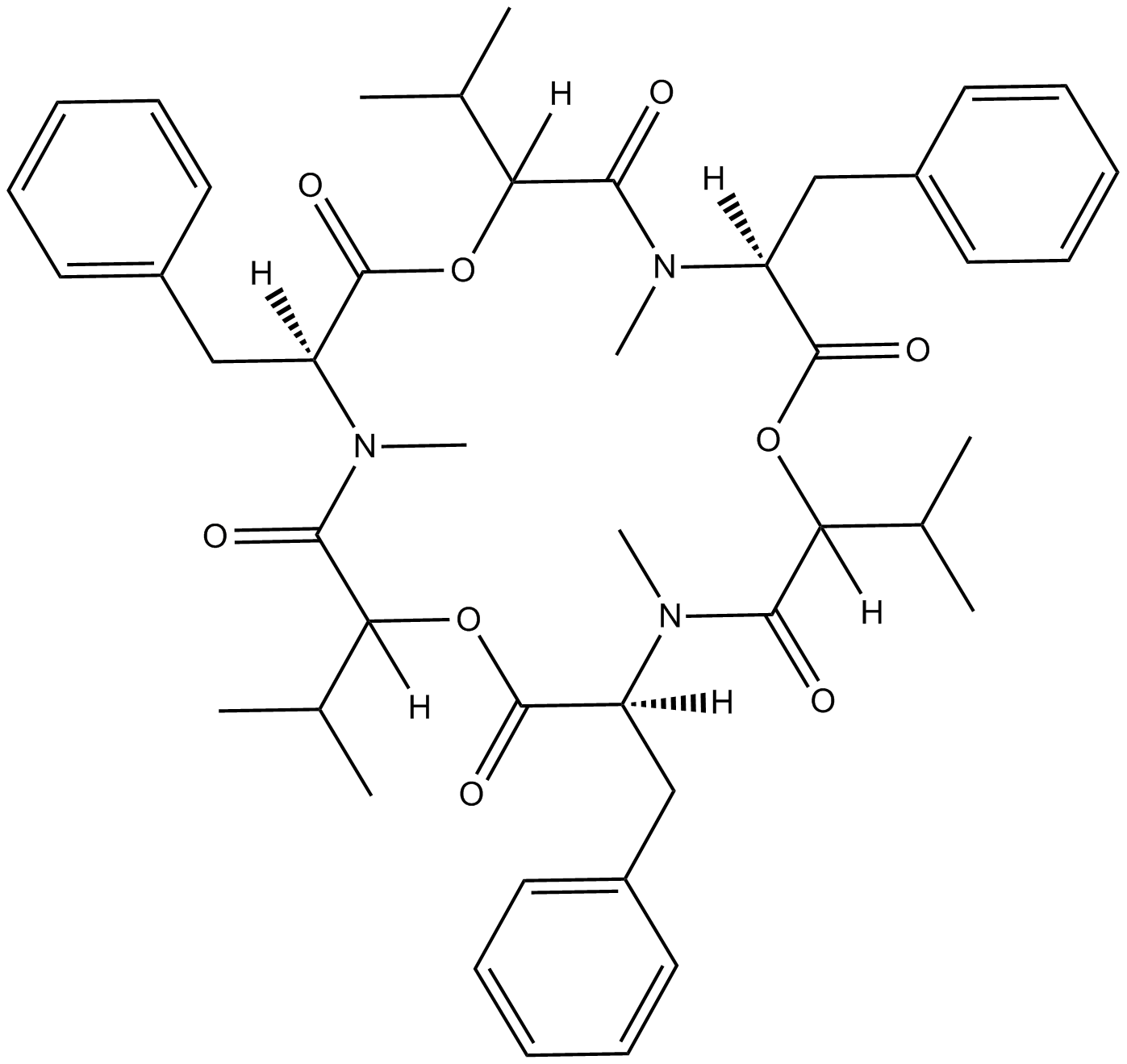
Beauvericin
IC50: 1.9 μM for antiviral activity; 1.7 μM for antimalarial activity
Beauvericin is a mycotoxin produced by various fungi including Fusarium spp. and Beaveria bassiana. As a cyclic hexadepsipeptide belonging to the enniatin antibiotic family, beauvericin contains three D-hydroxyisovaleryl and three N-methylphenylalanyl residues. Beauvericin was one of the active constituents of B. bassiana and was confirmed to have antibacterial and antifungal activities.
In vitro: Beauvericin has a strong antibacterial activity against human, animal and plant pathogenic bacteria, without selectivity between Gram-positive and Gram-negative bacteria. Unlike other antibiotics blocking the peptidoglycan biosynthesis, beauvericin does not target on the bacterial cell wall. Although it has broad-spectrum antibacterial activities, the antifungal activity of beauvericin is rarely reported as a single agent. Thus, the target of beauvericin is regarded to be different between bacteria and fungi and the activities of beauvericin need to be investigated against drug resistant bacteria [1].
In vivo: Previous study reported the the antifungal activity of beauvericin combined with miconazole or ketoconazole. Beauvericin at 0.5 mg/kg combined with ketoconazole at 0.5 mg/kg showed significant antifungal activity against Candida parapsilosis, which could cause high mortality rates quickly, particularly in neonates. In contrast, both beauvericin and ketoconazole alone have little to no inhibitory effect on C. parapsilosis [1].
Clinical trial: N/A
Reference:
[1] Wang Q, Xu L. Beauvericin, a bioactive compound produced by fungi: a short review. Molecules. 2012 Feb 24;17(3):2367-77.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 783.95 |
| Cas No. | 26048-05-5 |
| Formula | C45H57N3O9 |
| Solubility | Soluble in DMSO |
| Chemical Name | (3S,9S,15S)-3,9,15-tribenzyl-6,12,18-triisopropyl-4,10,16-trimethyl-1,7,13-trioxa-4,10,16-triazacyclooctadecane-2,5,8,11,14,17-hexaone |
| Canonical SMILES | CC(C(OC([C@](N(C(C(O1)([H])C(C)C)=O)C)([H])CC2=CC=CC=C2)=O)([H])C(N([C@@](C(OC(C(N([C@@](C1=O)([H])CC3=CC=CC=C3)C)=O)([H])C(C)C)=O)([H])CC4=CC=CC=C4)C)=O)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure