Search results for: 'research area proteases aminopeptidase'
-
 MA3695 Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal AntibodySummary: Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal Antibody
MA3695 Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal AntibodySummary: Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal Antibody -
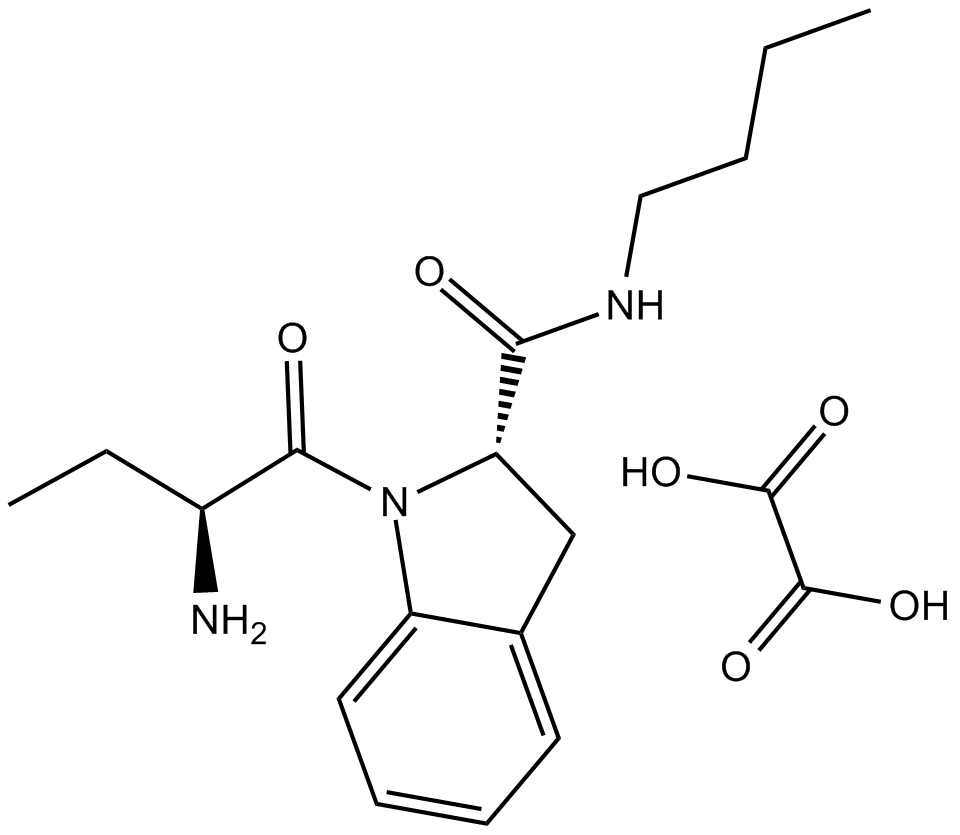 B6689 Butabindide oxalateSummary: CCK-inactivating serine protease (tripeptidyl peptidase II) inhibitor
B6689 Butabindide oxalateSummary: CCK-inactivating serine protease (tripeptidyl peptidase II) inhibitor -
 BA7971 M8891Summary: M8891 is an orally active, reversible and blood-brain barrier-crossing inhibitor of methionine aminopeptidase 2.
BA7971 M8891Summary: M8891 is an orally active, reversible and blood-brain barrier-crossing inhibitor of methionine aminopeptidase 2. -
 P1578 Recombinant Aeromonas AminopeptidaseSummary: Recombinant Aeromonas Aminopeptidase (E.coli, Tag Free, Lyophilized)
P1578 Recombinant Aeromonas AminopeptidaseSummary: Recombinant Aeromonas Aminopeptidase (E.coli, Tag Free, Lyophilized) -
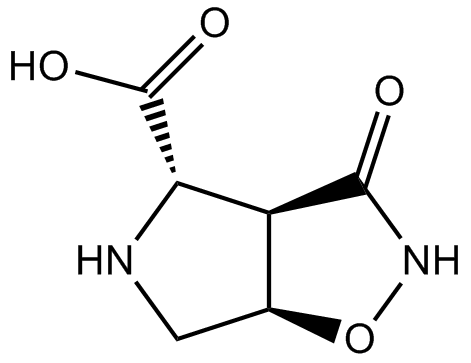 B6982 (±)-HIP-ASummary: excitatory amino acid transporter (EAAT) blocker
B6982 (±)-HIP-ASummary: excitatory amino acid transporter (EAAT) blocker -
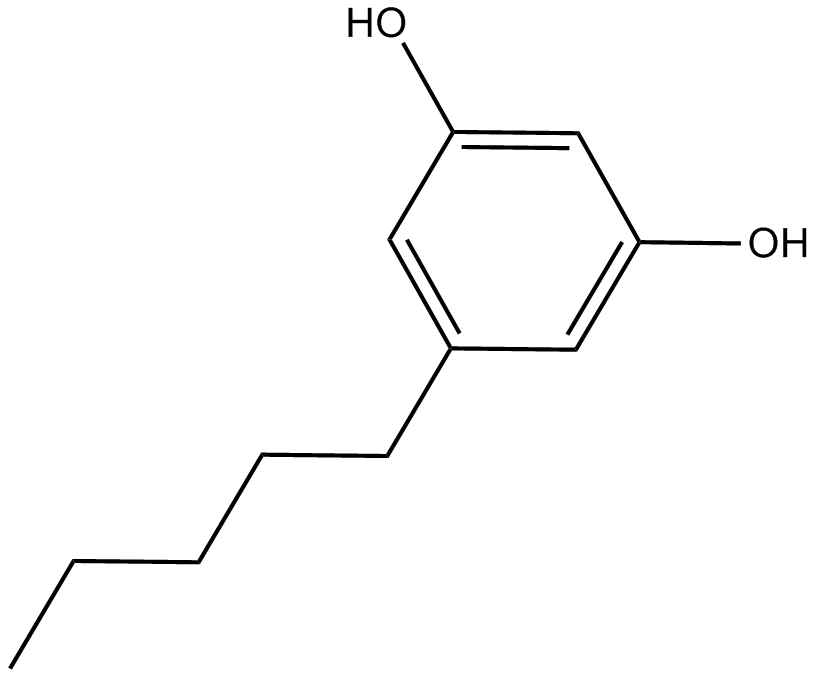 C7059 Olivetol
C7059 Olivetol -
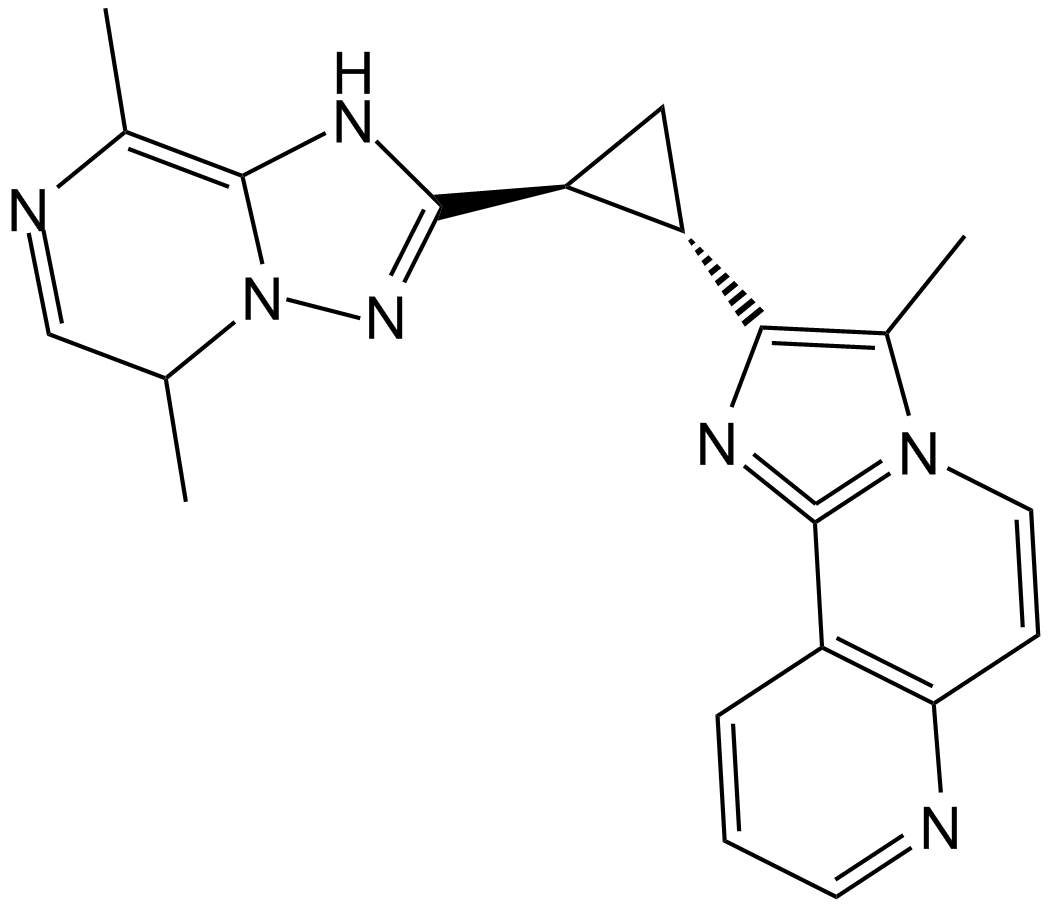 B5891 SEP-0372814
B5891 SEP-0372814 -
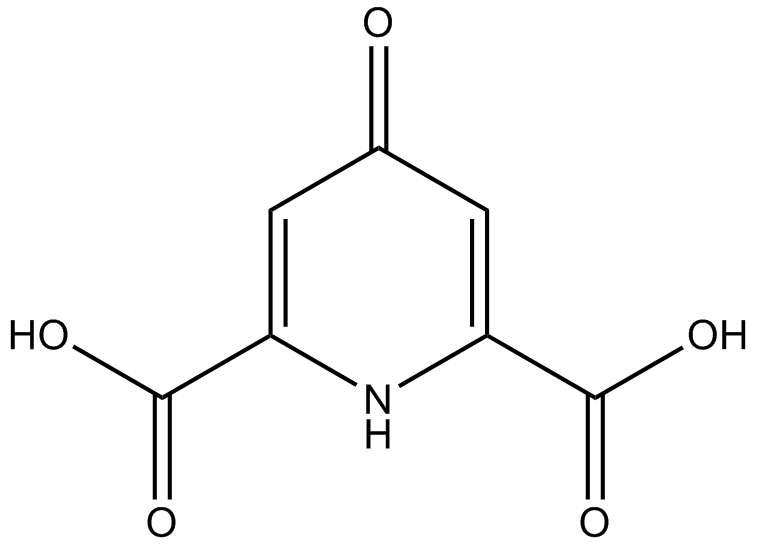 C7196 Chelidamic acid
C7196 Chelidamic acid -
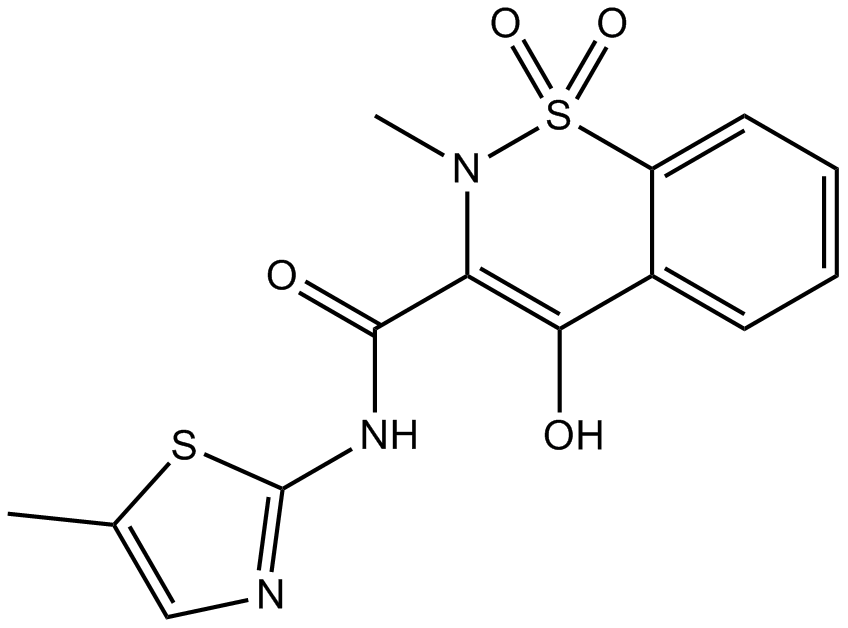 A8466 Meloxicam (Mobic)Target: COXSummary: Nonsteroidal anti-inflammatory drug
A8466 Meloxicam (Mobic)Target: COXSummary: Nonsteroidal anti-inflammatory drug -
 BA1307 TolfenpyradSummary: Tolfenpyrad is an insecticide.
BA1307 TolfenpyradSummary: Tolfenpyrad is an insecticide.


