Search results for: 'rita nsc 653095'
-
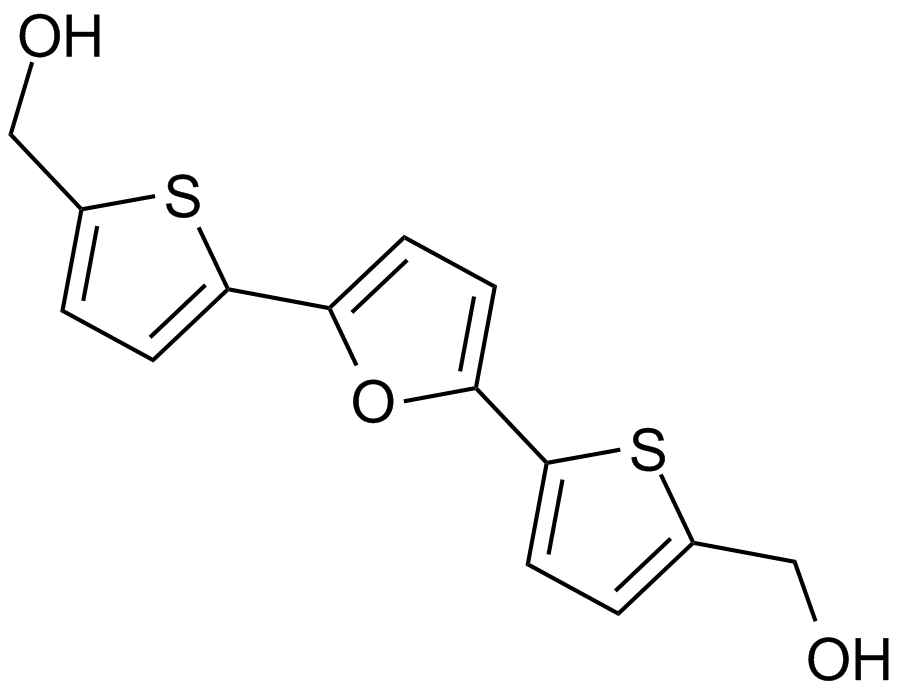 A4202 RITA (NSC 652287)Summary: Mdm2-p53 interaction and p53 ubiquitination blocking
A4202 RITA (NSC 652287)Summary: Mdm2-p53 interaction and p53 ubiquitination blocking -
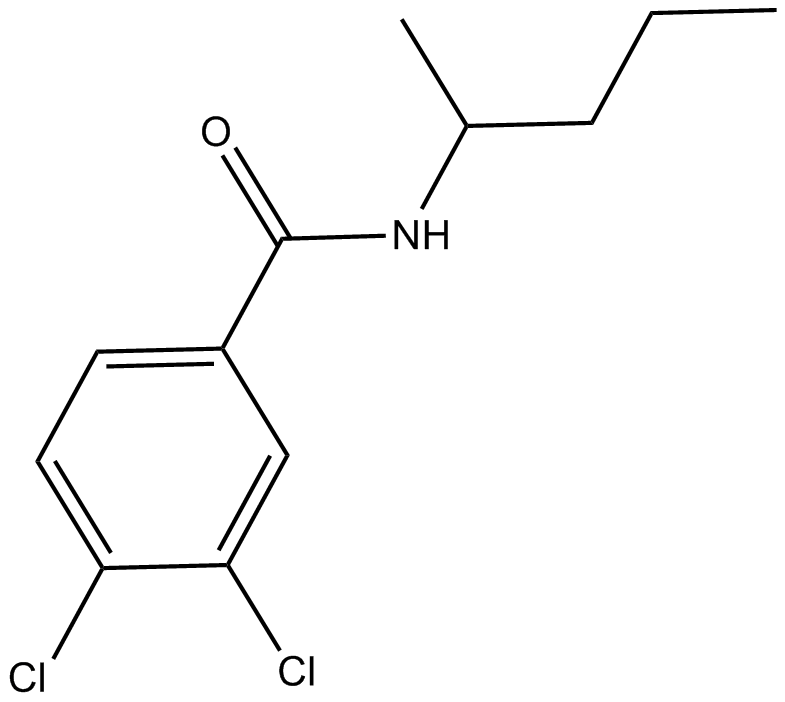 A4051 NSC 4050201 CitationTarget: MMPSummary: MT1-MMP inhibitor
A4051 NSC 4050201 CitationTarget: MMPSummary: MT1-MMP inhibitor -
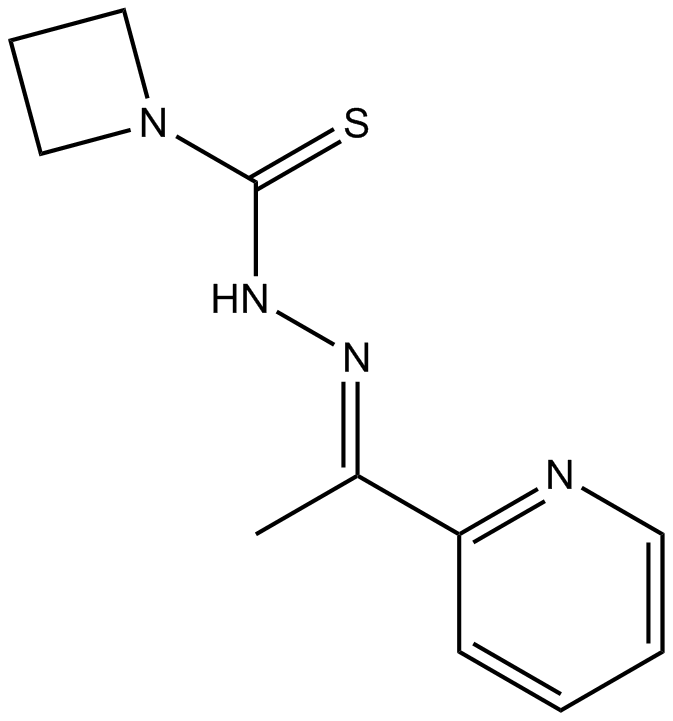 A4208 NSC 319726Target: p53Summary: Reactivator of mutant p53
A4208 NSC 319726Target: p53Summary: Reactivator of mutant p53 -
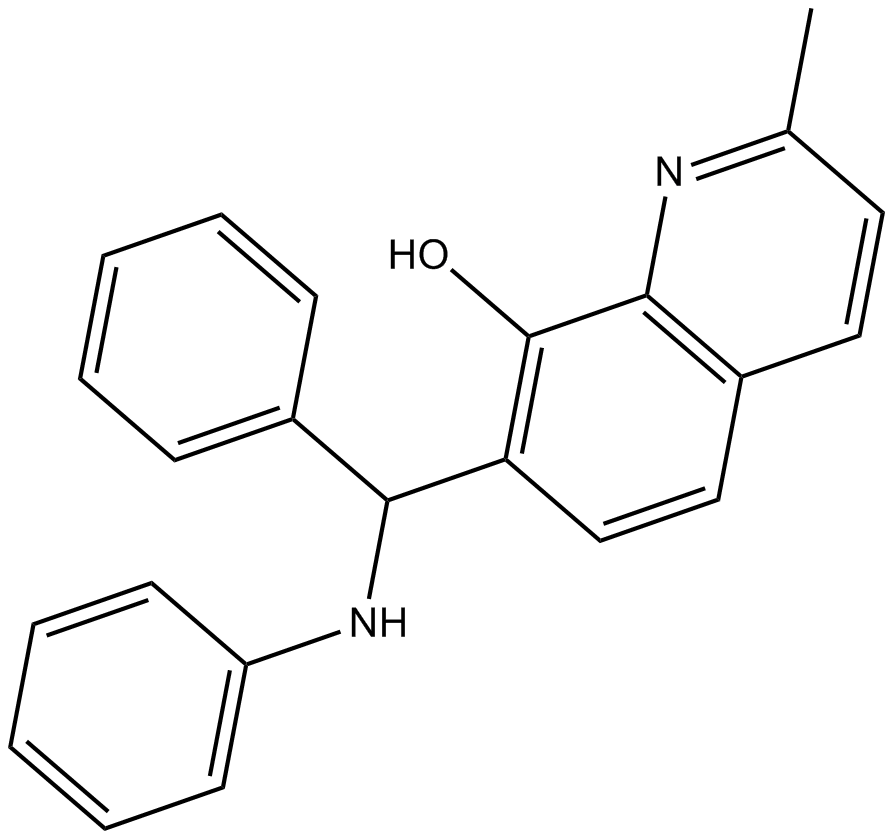 A4230 NSC 66811Summary: MDM2 inhibitor
A4230 NSC 66811Summary: MDM2 inhibitor -
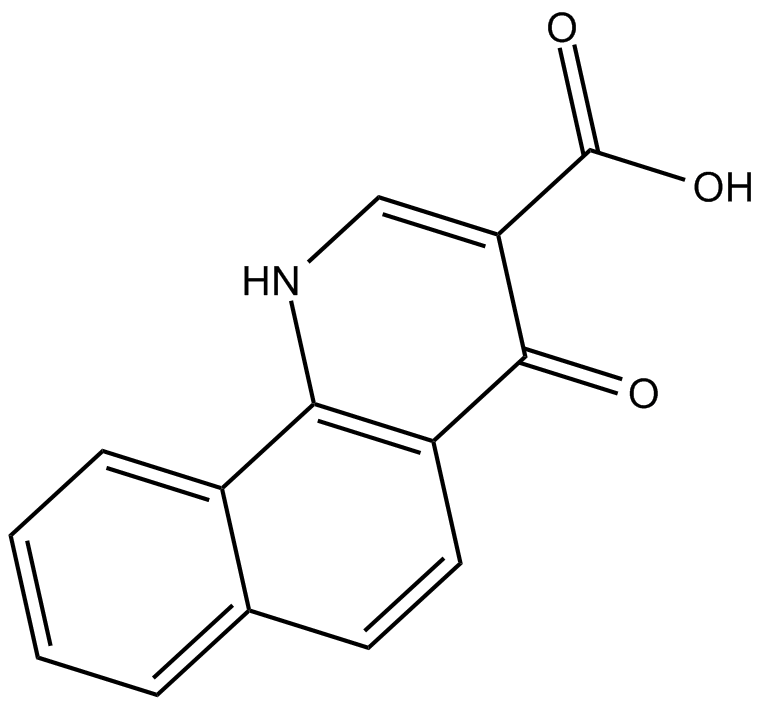 C4638 NSC 210902Summary: CK2 inhibitor
C4638 NSC 210902Summary: CK2 inhibitor -
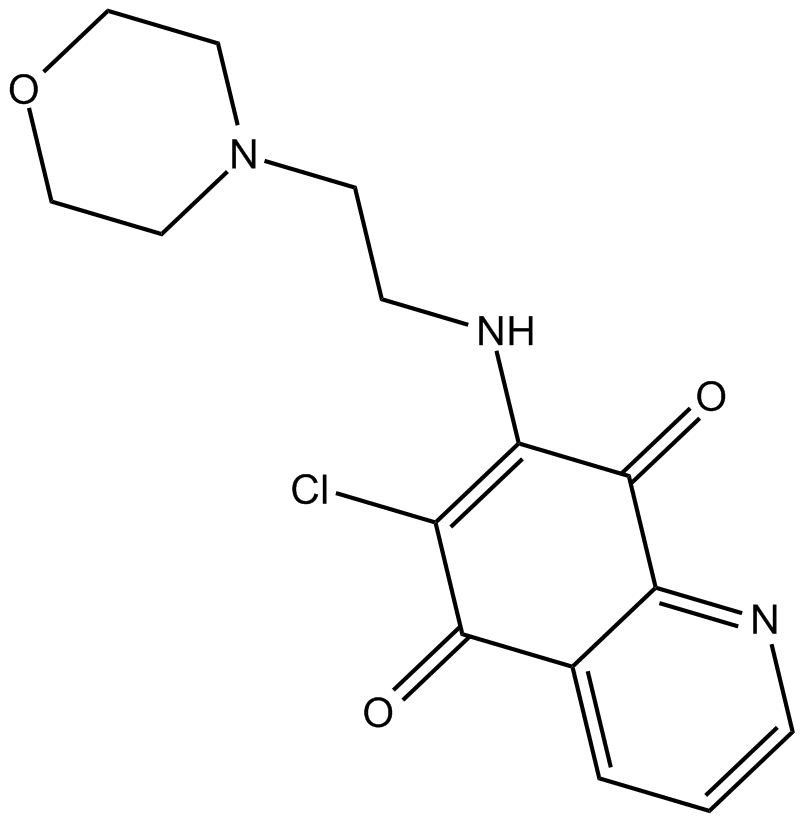 B6881 NSC 6632841 CitationSummary: Cdc25 dual specificity phosphatases inhibitor
B6881 NSC 6632841 CitationSummary: Cdc25 dual specificity phosphatases inhibitor -
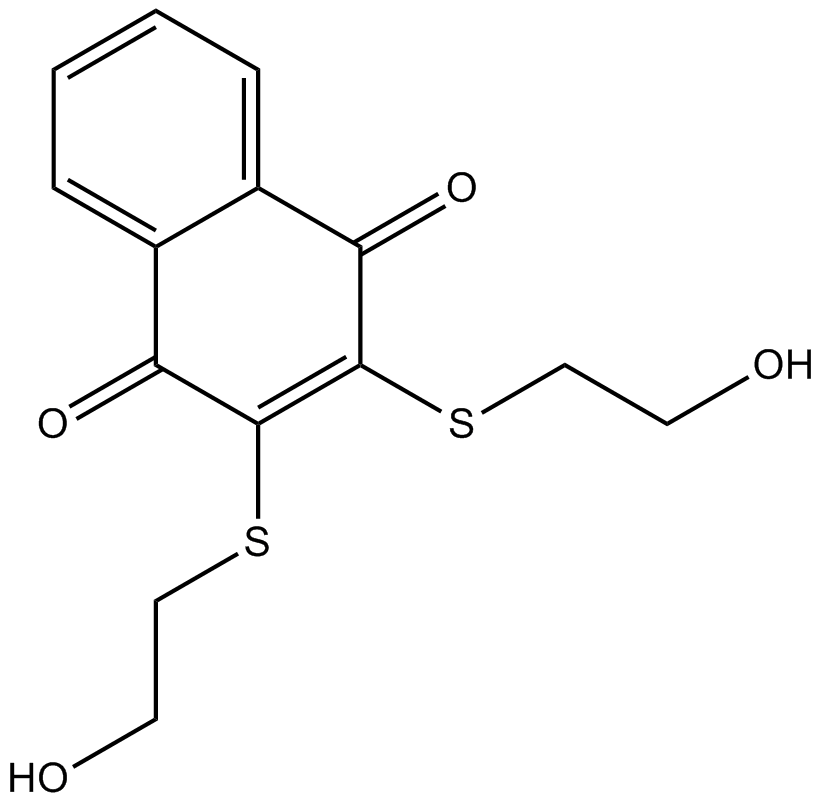 B6784 NSC 95397Summary: Cdc25 dual specificity phosphatases inhibitor
B6784 NSC 95397Summary: Cdc25 dual specificity phosphatases inhibitor -
 B6956 NSC 625987Summary: Cyclin-dependent kinase (cdk) 4 inhibitor
B6956 NSC 625987Summary: Cyclin-dependent kinase (cdk) 4 inhibitor -
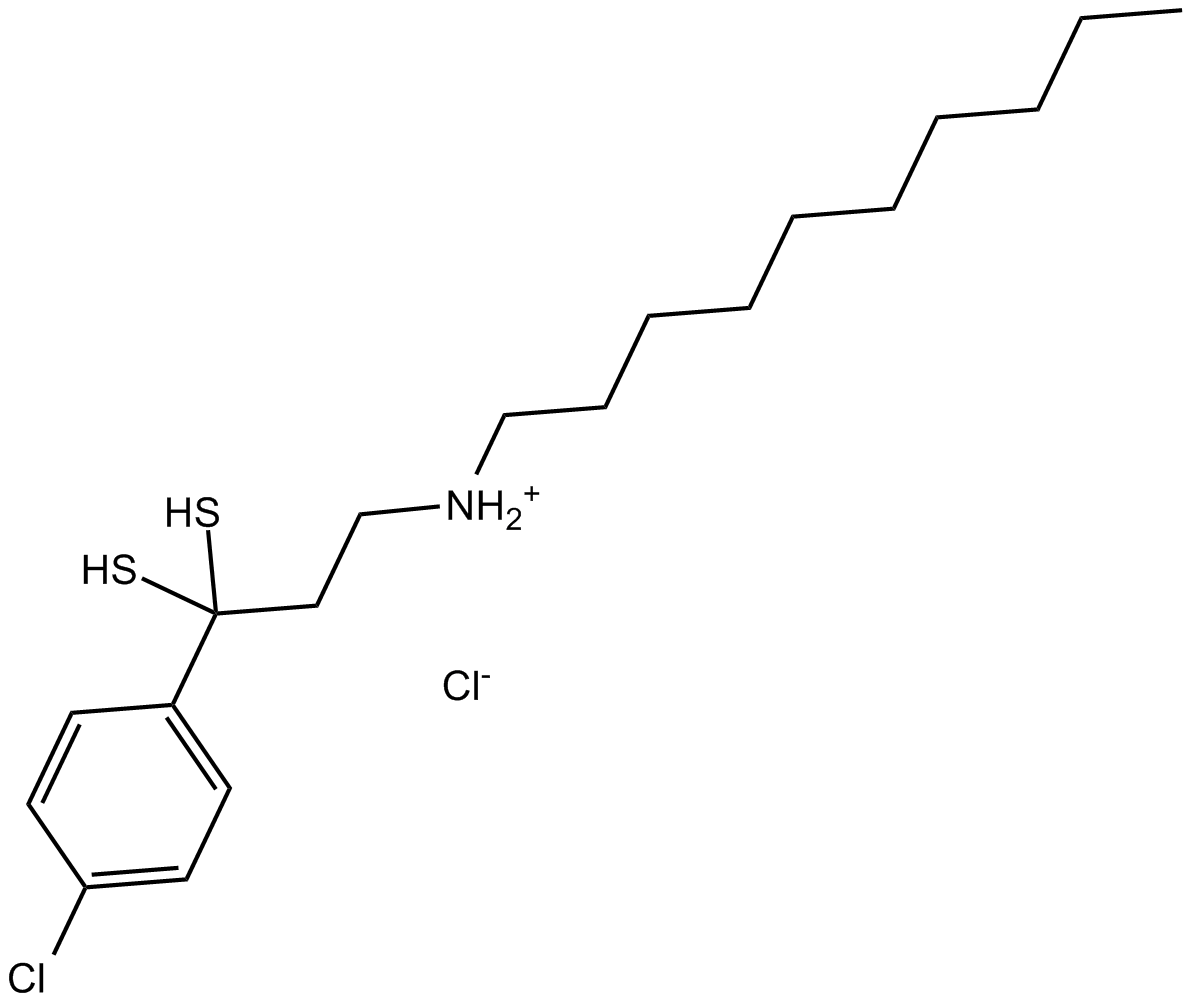 B7727 NSC 624206Summary: Ubiquitin-activating enzyme (E1) inhibitor
B7727 NSC 624206Summary: Ubiquitin-activating enzyme (E1) inhibitor -
 A4496 NSC 3852Target: Histone Deacetylases (HDACs)Summary: HDAC inhibitor
A4496 NSC 3852Target: Histone Deacetylases (HDACs)Summary: HDAC inhibitor


