Search results for: 'pyridoxal isonicotinoyl hydrazone'
-
 BA7471 PyridoxalSummary: Pyridoxal is one of the main forms of vitamin B6.
BA7471 PyridoxalSummary: Pyridoxal is one of the main forms of vitamin B6. -
 B7911 Pyridoxal hydrochlorideSummary: An endogenous metabolite
B7911 Pyridoxal hydrochlorideSummary: An endogenous metabolite -
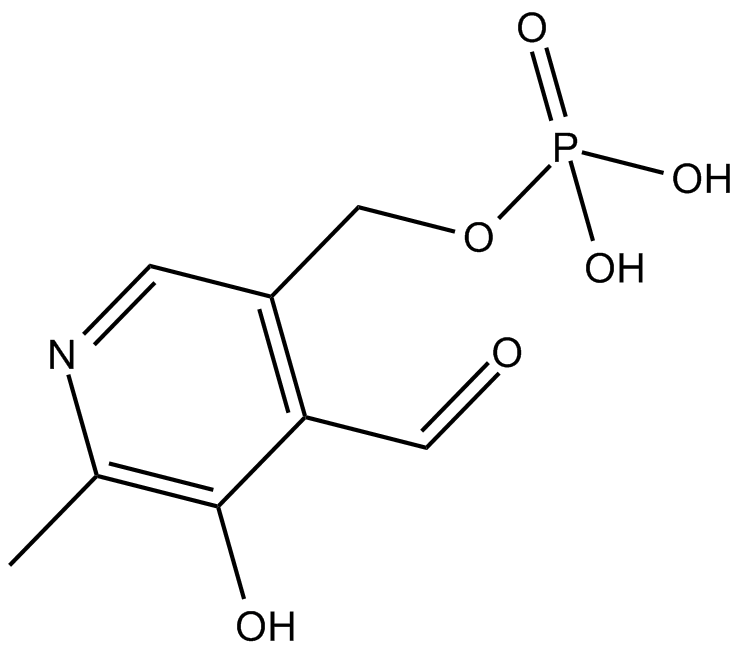 B7910 Pyridoxal 5 phosphateSummary: Active form of vitamin B6 serving as a coenzyme for synthesis of amino acids, neurotransmitters (serotonin, norepinephrine), sphingolipids, aminolevulinic acid.
B7910 Pyridoxal 5 phosphateSummary: Active form of vitamin B6 serving as a coenzyme for synthesis of amino acids, neurotransmitters (serotonin, norepinephrine), sphingolipids, aminolevulinic acid. -
 C8681 4-(4-Acetyl-phenoxy)-butyric acidSummary: A core structural unit of the acid-sensitive hydrazone linker in ADCs.
C8681 4-(4-Acetyl-phenoxy)-butyric acidSummary: A core structural unit of the acid-sensitive hydrazone linker in ADCs.


