TEV Protease
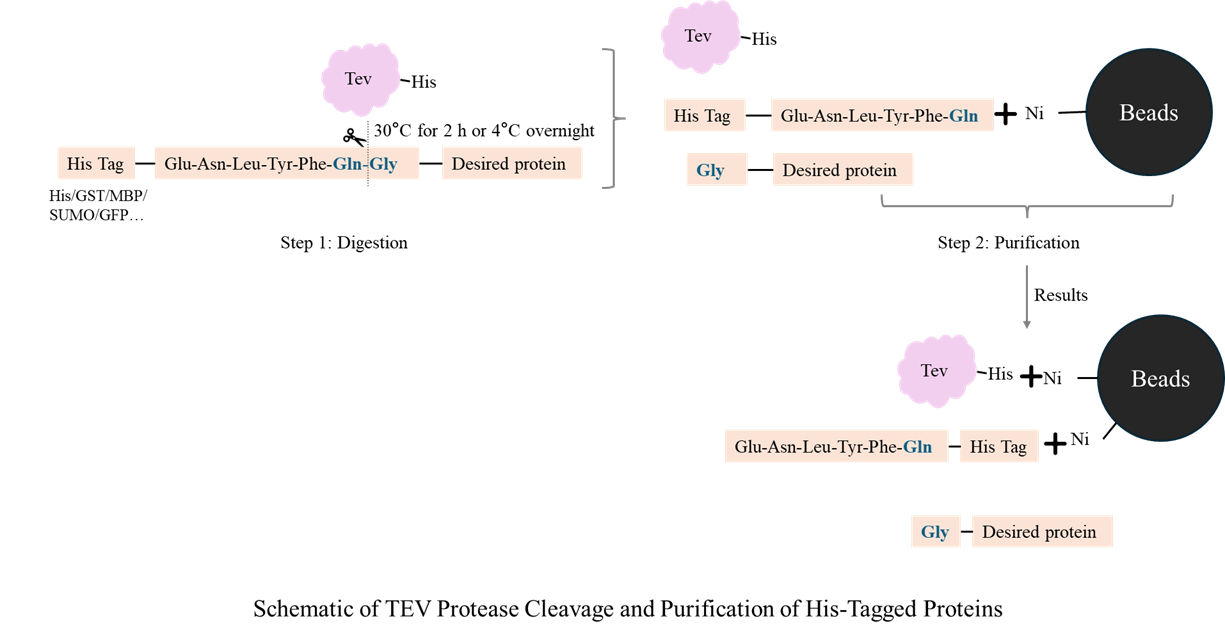
TEV protease is a cysteine protease used to cleave fusion tags from recombinant proteins.It is expressed using an E. coli expression system. It specifically recognizes the amino acid sequence ENLYFQ▼S, but tolerates amino acids G, A, M, C, or H at the P1' position. TEV protease maintains activity in the pH range of 6.0–9.0, with reduced or absent enzyme activity at pH ≤ 5.0, and is often used in affinity purification steps to remove His, GST, or other fusion tags. To maximize protein activity and structure, cleavage reactions are typically performed overnight at 4°C.
|
Name |
TEV Protease |
|
Synonym |
TEVp, rTEV |
|
Recognition/Cleavage Site |
Glu-Asn-Leu-Tyr-Phe-Gln-▼Gly (Gly= Gly/Ala/Met/Cys/His) |
|
Tag |
Tags such as His, GST, MBP, SUMO, and GFP |
|
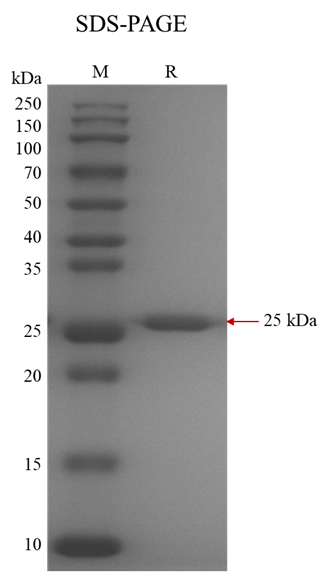
M.Wt |
~ 25 kDa |
|
Source |
E. coli |
|
Label |
N-terminal His Tag |
|
Purity |
> 95%, determined by SDS-PAGE. |
|
Tev Protease Buffer |
50 mM Tris-HCl, 1 mM EDTA, 250 mM NaCl, 50% (v/v) glycerol, 0.1% (w/v) Triton X-100, pH 7.5 @25°C. |
|
10×TEV Protease Reaction Buffer |
500 mM Tris-HCl, 5 mM EDTA, pH 8.0 @25°C. |
|
Enzyme activity |
10 U/μL |
|
Unit Definition |
One unit (U) of activity represents the enzyme quantity needed to cleave ≥85% of 3 μg control substrate after 1 h at 30°C and pH 8.0. |
|
Optimal pH range |
pH 6.0 - 9.0 (optimal at pH 7.0) |
|
Reaction Temperature |
4°C - 30°C |
|
Inactivation Condition |
65°C, 20 min |
|
Components |
1000 U |
5000 U |
10000 U |
Storage |
|
TEV Protease |
0.1 mL |
0.5 mL |
1 mL |
-80°C |
|
10X TEV Protease Reaction Buffer |
1 mL |
5 x 1 mL |
10 x 1 mL |
-80°C |
|
0.1 M DTT |
0.5 mL |
2.5 mL |
5 mL |
-80°C |
|
Shipping: dry ice Shelf life: 2 years |
||||
Note: It is recommended to aliquot upon first use to avoid repeated freeze-thaw cycles: store aliquots at -20°C for a validity period of 6 months.
1. What types of fusion protein tags can TEV protease be used for?
A: It can be used for fusion proteins containing tags such as His, GST, MBP, SUMO, and GFP.
2. What is the recognition sequence?
A: Glu-Asn-Leu-Tyr-Phe-Gln-Gly, with cleavage occurring between Gln and Gly. However, the amino acid at the P1' position (Gly) can also be Gly, Ala, Met, Cys, or His.
3. What are the reaction conditions and inactivation conditions for TEV protease?
A: Reaction conditions: Active over a broad temperature range (4°C–30°C, with an optimum of 30°C) and pH range (6.0–8.5).
Inactivation conditions: The enzyme can be inactivated by heating at 65°C for 20 minutes. To maximize protein activity and preserve structural integrity, cleavage is recommended to be performed overnight at 4°C.
4. What factors affect TEV protease cleavage activity?
A: Cleavage activity may be affected if the fusion protein sample contains denaturants (>2 M urea or >0.5 M guanidine hydrochloride), histidine residue analogs (>50 mM imidazole), certain metal ions (≥5 mM Zn²⁺, ≥1 mM Cu²⁺, and ≥10 mM Co²⁺), or abnormal pH (e.g., pH <6 or >9). It is generally recommended to dialyze the fusion protein before TEV protease cleavage. Additionally, TEV cleavage systems are compatible with the following protease inhibitors (e.g., aprotinin, benzamidine, leupeptin, pepstatin, and PMSF), as well as certain salts (e.g., up to 2 M NaCl, with optimal activity at ≤0.2 M NaCl; 10 mM MgSO₄/MnCl₂/CaCl₂) and EDTA (up to 100 mM).
5. Is TEV protease compatible with protease inhibitor cocktails?
A: Yes, it is compatible with a series of inhibitor cocktail products, such as the inhibitor cocktail for tagged proteins (K1010) and universal inhibitor cocktails (K1007 and K4002).
6. What reaction buffers are compatible with TEV protease?
A: The recommended cleavage reaction buffer is 50 mM Tris-HCl, 0.5 mM EDTA, 1 mM DTT, pH 7.5. The enzyme is also active in pH 6–9 buffers such as HEPES, potassium phosphate, and sodium acetate.
7. What is the role of DTT in the cleavage system?
A: The catalytic activity of TEV protease depends entirely on the cysteine residue (Cys151) at its active site, which must maintain a reduced sulfhydryl group (-SH) to form a functional catalytic dyad with histidine (His46) for substrate cleavage. DTT, a strong reducing agent, is essential for maintaining this reduced state. TEV protease reactions typically require a final concentration of 1 mM DTT. If column purification after digestion precludes the use of DTT, successful digestion may still be achieved without DTT, but DTT is still recommended and can be removed by dialysis prior to column purification.
8. How is TEV protease removed from the reaction after tag cleavage?
A: TEV protease contains a His tag at its N-terminus. After fusion protein cleavage, TEV protease can be removed from the reaction by immobilized metal affinity chromatography. However, since the reaction system contains components such as EDTA and DTT, dialysis is recommended before applying to the Ni column.
9. What should be done if the protein yield after cleavage is low as assessed by SDS-PAGE?
A: Optimization can be performed by considering factors such as the fusion protein loading amount, buffer composition, inhibitor composition, cleavage system conditions, and cleavage reaction time (e.g., overnight or longer at 4°C, up to three days for some proteins).
10. What other tag cleavage products are available for fusion proteins, and what are the advantages of TEV protease?
A: In addition to TEV protease, we also recommend PreScission Protease (PSP) (K1101), which recognizes the sequence Leu-Glu-Val-Leu-Phe-Gln-▼Gly-Pro, and SUMO Protease (K1103), which cleaves after the Gly-Gly at the C-terminus of SUMO. TEV protease offers higher substrate sequence specificity and is active over a broad temperature range (4–30°C) and a wide pH range (6.0–8.5), meeting the requirements for various reaction conditions.