S Tag Peptide
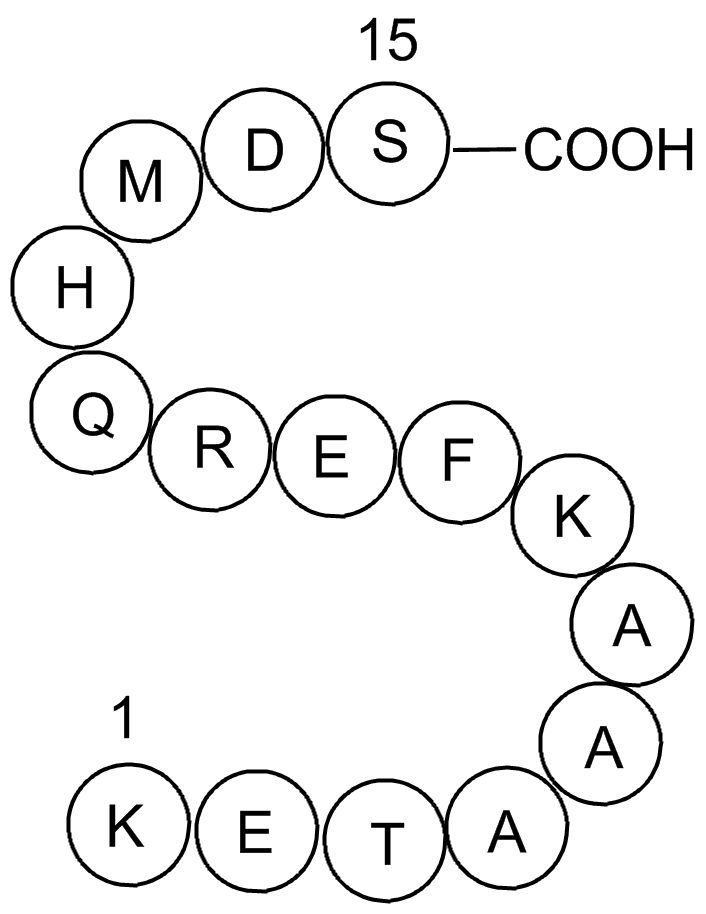
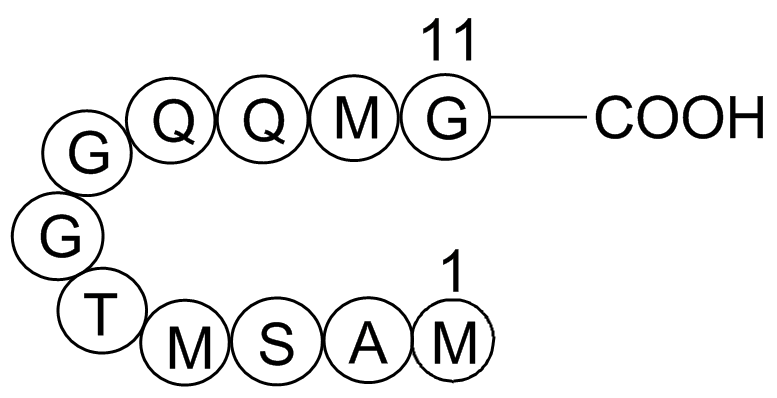
S Tag is the name of an oligopeptide derived from pancreatic ribonuclease A (RNase A). If RNase A is digested with subtilisin, a single peptide bond is cleaved, but the resulting two products remain weakly bound to each other and the protein, it is called ribonuclease S, remains active although each of the two products alone shows no enzymatic activity. The N-terminus of the original RNase A, also called S-peptide, consists of 20 amino acid residues, of which only the first 15 are required for ribonuclease activity. This 15 amino acids long peptide is called S15 or S-tag.
It is believed that the peptide with its abundance of charged and polar residues could improve solubility of proteins it is attached to. Moreover, the peptide alone is thought not to fold into a distinct structure. On DNA-level the S-tag can be attached to the N- or C-terminus of any protein. After gene expression, such a tagged protein can be detected by commercially available antibodies. [1]
References:
1. R.T. Raines et al., The S-Tag Fusion System for Protein Purification. Methods Enzymol. 326, 362-367 (2000)
- 1. Takushi Miyoshi, Qianli Zhang, et al. "Semi-automated single-molecule microscopy screening of fast-dissociating specific antibodies directly from hybridoma cultures." Cell Rep. 2021 Feb 2;34(5):108708. PMID: 33535030
- 2. Hawa Gyamfi. "Protein Engineering of Microbial Ferritins." University of Waterloo. 2019.
| Physical Appearance | A solid |
| Storage | Desiccate at -20°C |
| M.Wt | 1748.91 |
| Cas No. | 7429-70-1 |
| Formula | C73H117N23O25S |
| Synonyms | H-Lys-Glu-Thr-Ala-Ala-Ala-Lys-Phe-Glu-Arg-Gln-His-Met-Asp-Ser-OH |
| Solubility | ≥174.9 mg/mL in DMSO; insoluble in EtOH; ≥50 mg/mL in H2O |
| Chemical Name | S Tag |
| Canonical SMILES | CC(C(C(=O)NC(C)C(=O)NC(C)C(=O)NC(C)C(=O)NC(CCCCN)C(=O)NC(CC1=CC=CC=C1)C(=O)NC(CCC(=O)O)C(=O)NC(CCCNC(=N)N)C(=O)NC(CCC(=O)N)C(=O)NC(CC2=CN=CN2)C(=O)NC(CCSC)C(=O)NC(CC(=O)O)C(=O)NC(CO)C(=O)O)NC(=O)C(CCC(=O)O)NC(=O)C(CCCCN)N)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Description | S Tag Peptide is an oligopeptide derived from pancreatic ribonuclease A used for improving protein solubility. | |||||
| Targets | anti-S-Tag antibody | |||||
| IC50 | ||||||
Quality Control & MSDS
- View current batch:
Chemical structure