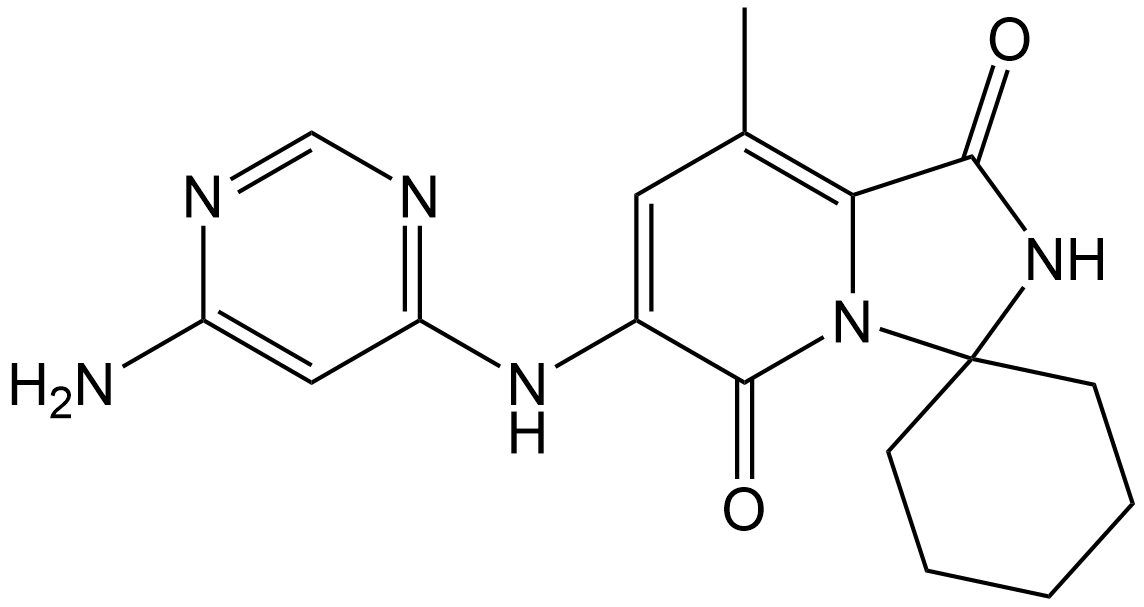
Tomivosertib
Tomivosertib (CAS No. 1849590-01-7) is a potent, highly selective, orally active MNK1 and MNK2 inhibitor. By directly inhibiting MNK1/2, it blocks phosphorylation activation of eukaryotic translation initiation factor 4E (eIF4E) at Ser209, and is involved in regulating the MNK-eIF4E signaling pathway, the AMPK-MNK-eIF4E metabolic pathway, and upstream RAS/RAF/MEK/ERK and p38 MAPK signaling pathways. The IC50 is 2.4 nM for MNK1 and 1 nM for MNK2.
Commonly used concentrations in cell culture are 25 nM (human dorsal root ganglion neurons), 0.1–10 μM (acute myeloid leukemia cells), 10–40 μM (glioblastoma cells and related endothelial cells), and 1 μM (mouse hepatocytes), mainly for evaluating eIF4E phosphorylation, cell proliferation / apoptosis, angiogenesis, spontaneous neuronal firing, and metabolism-related indicators.
Common dosing in animal studies is 10 mg/kg (pancreatic cancer mice, oral gavage) and 2 mg/kg (glioblastoma mice, oral), used to inhibit tumor growth, angiogenesis, and regulate ketogenesis.
References:
[1] Reich SH, Sprengeler PA, Chiang GG, Appleman JR, Chen J, Clarine J, Eam B, Ernst JT, Han Q, Goel VK, Han EZR, Huang V, Hung INJ, Jemison A, Jessen KA, Molter J, Murphy D, Neal M, Parker GS, Shaghafi M, Sperry S, Staunton J, Stumpf CR, Thompson PA, Tran C, Webber SE, Wegerski CJ, Zheng H, Webster KR. Structure-based Design of Pyridone-Aminal eFT508 Targeting Dysregulated Translation by Selective Mitogen-activated Protein Kinase Interacting Kinases 1 and 2 (MNK1/2) Inhibition. J Med Chem. 2018 Apr 26;61(8):3516-3540. doi: 10.1021/acs.jmedchem.7b01795. Epub 2018 Mar 29. PMID: 29526098.
[2] Suarez M, Blyth GT, Mina AA, Kosciuczuk EM, Dolniak B, Dinner S, Altman JK, Eklund EA, Saleiro D, Beauchamp EM, Platanias LC. Inhibitory effects of Tomivosertib in acute myeloid leukemia. Oncotarget. 2021 May 11;12(10):955-966. doi: 10.18632/oncotarget.27952. PMID: 34012509; PMCID: PMC8121614.
[3] Zhang Q, Zhao J, Xu T. Inhibition of eukaryotic initiation factor 4E by tomivosertib suppresses angiogenesis, growth, and survival of glioblastoma and enhances chemotherapy's efficacy. Fundam Clin Pharmacol. 2023 Aug;37(4):807-815. doi: 10.1111/fcp.12877. Epub 2023 Feb 7. PMID: 36691859.
[4] Yang H, Zingaro VA, Lincoff J, Tom H, Oikawa S, Oses-Prieto JA, Edmondson Q, Seiple I, Shah H, Kajimura S, Burlingame AL, Grabe M, Ruggero D. Remodelling of the translatome controls diet and its impact on tumorigenesis. Nature. 2024 Sep;633(8028):189-197. doi: 10.1038/s41586-024-07781-7. Epub 2024 Aug 14. PMID: 39143206; PMCID: PMC12147720.
[5] Li Y, Uhelski ML, North RY, Mwirigi JM, Tatsui CE, McDonough KE, Cata JP, Corrales G, Dussor G, Price TJ, Dougherty PM. Tomivosertib reduces ectopic activity in dorsal root ganglion neurons from patients with radiculopathy. Brain. 2024 Sep 3;147(9):2991-2997. doi: 10.1093/brain/awae178. PMID: 39046204; PMCID: PMC11370786.
| Storage | Store at -20°C |
| M.Wt | 340.39 |
| Cas No. | 1849590-01-7 |
| Formula | C17H20N6O2 |
| Chemical Name | 6'-((6-aminopyrimidin-4-yl)amino)-8'-methyl-2'H-spiro[cyclohexane-1,3'-imidazo[1,5-a]pyridine]-1',5'-dione |
| Canonical SMILES | O=C(C1=C(C)C=C(NC2=NC=NC(N)=C2)C(N13)=O)NC43CCCCC4 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |