TCS 3035
Target: GPR35
IC50: N/A
TCS3035, a GPR35 agonist, shows significantly high potency at rat and human GPR35 orthologs with pEC50 values of 5.13 and 5.86, respectively [1]. G protein-coupled receptors (GPCRs) are the largest and most studied group of transmembrane polypeptides. GPR35 is a poorly characterized G protein-coupled receptor that plays an important role in immune-modulation, gastric function and the regulation of insulin secretion. GPR35 is predominantly expressed in the gastro-intestinal tract and immune tissues. The tryptophan metabolite kynurenic acid has been suggested to be the endogenous ligand for GPR35 [1].
In vitro: Mutation to alanine of the conserved arginine at position 3.36 in either FLAG-hGPR35-eYFP or FLAG-rGPR35-eYFP resulted in a complete loss of agonist function of TCS3035 [1]. In addition, TCS3035-induced internalization of GPR35 is correlated with TCS3035 potency in receptor-β-arrestin-2 interaction BRET assays. Mutation to alanine of tyrosine 3.32 in transmembrane domain III abolished β-arrestin-2 recruitment in response to TCS3035 [1].
In vivo: N/A
Reference:
1. Jenkins L, Alvarez-Curto E, Campbell K, de Munnik S, Canals M, Schlyer S, et al. Agonist activation of the G protein-coupled receptor GPR35 involves transmembrane domain III and is transduced via Galpha(1)(3) and beta-arrestin-2. Br J Pharmacol. 2011;162(3):733-48.
| Physical Appearance | Off White solid |
| Storage | Store at -20°C |
| M.Wt | 279.27 |
| Cas No. | 871085-49-3 |
| Formula | C12H9NO5S |
| Solubility | <27.93mg/ml in DMSO |
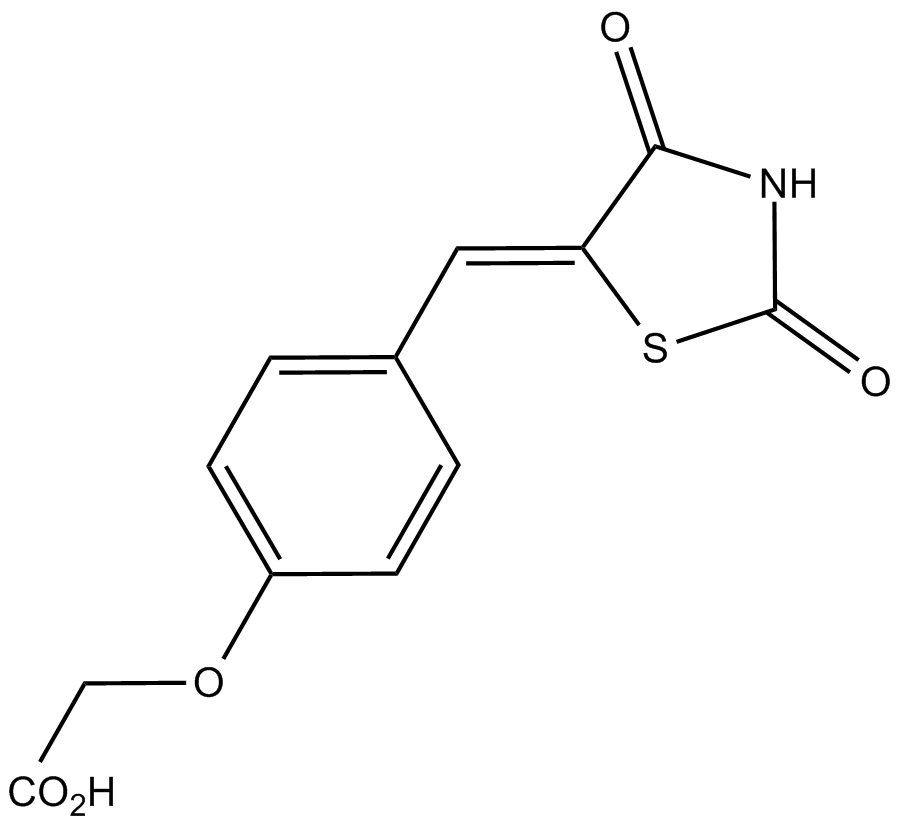
| Chemical Name | (Z)-2-(4-((2,4-dioxothiazolidin-5-ylidene)methyl)phenoxy)acetic acid |
| Canonical SMILES | OC(COc1ccc(/C=C(/C(N2)=O)\SC2=O)cc1)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure