Sunitinib
Sunitinib (CAS 557795-19-4) is an orally bioavailable small molecule inhibitor targeting multiple receptor tyrosine kinases (RTKs), including VEGFR1-3, PDGFRα/β, c-kit, and RET. Research has shown sunitinib inhibits proliferation in nasopharyngeal carcinoma (NPC) cell lines HK1, CNE-1, CNE-2, HONE-1, and C666-1, with IC50 values ranging from approximately 2.06 to 7.57 μM. Additionally, it induces apoptosis and G0/G1 cell cycle arrest in NPC cell lines and significantly decreases microvessel density in CNE-2 xenograft tumor models in vivo. Thus, sunitinib is useful for investigating pathways involved in tumor angiogenesis and proliferation.
References:
[1]Hui EP1, Lui VW, Wong CS, Ma BB, Lau CP, Cheung CS, Ho K, Cheng SH, Ng MH, Chan AT. Preclinical evaluation of sunitinib as single agent or in combination with chemotherapy in nasopharyngeal carcinoma. Invest New Drugs. 2011 Dec;29(6):1123-31.
- 1. Zixuan Chen, Yu Zhang, et al. "Gingerenone A inhibits LDHA-mediated glycolysis and restores sunitinib sensitivity in renal cell carcinoma." Biochem Pharmacol. 2025 Nov 26:117563. PMID: 41314434
- 2. Zixuan Chen, Weiyuan Li, et al. "Chrysin enhances sunitinib sensitivity in renal cell carcinoma by inducing ferroptosis via targeting PI3K/Akt/GPX4 pathway." Toxicol Appl Pharmacol. 2025 Aug 21:117531. PMID: 40848919
- 3. Zixuan Chen, Xing Jia, et al. "A novel peptide TCL6148 induces ferroptosis via the GOT1/GPX4 pathway to enhance sunitinib sensitivity in renal cell carcinoma." Int J Biol Macromol. 2025 Jun:313:144242. PMID: 40379184
- 4. Zixuan Chen, Chengtao Han, et al. "2‐Undecanone induces ferroptosis via the STAT3/GPX4 pathway to enhance sensitivity of renal cell carcinoma to sunitinib." Biofactors. 2025 Mar-Apr;51(2):e70016. PMID: 40200786
- 5. Ângela Marques-Magalhães, Filipa Moreira-Silva, et al. "Combination of MLo-1508 with sunitinib for the experimental treatment of papillary renal cell carcinoma." Front Oncol. 2025 Mar 24;15:1399956. PMID: 40196736
- 6. Nicole A Muench, Heather M Schmitt, et al. "Preservation of Murine Whole Eyes With Supplemented UW Cold Storage Solution: Anatomical Considerations." Transl Vis Sci Technol. 2024 Nov 4;13(11):24. PMID: 39560629
- 7. Zixuan Chen, Xing Jia, et al. "AUY922 Improves Sensitivity to Sunitinib in Clear Cell Renal Cell Carcinoma Based on Network Pharmacology and in Vitro Experiments." Heliyon. 2024 Jul 18;10(14):e34834. PMID: 39149033
- 8. Zixuan Chen, Sheng Cheng, et al. "Syringin as a novel therapeutic agent for renal cell carcinoma by targeting EGFR/PI3K/Akt pathway and enhancing sunitinib efficacy." Journal of Functional Foods Volume 123, December 2024, 106556
- 9. Marieke Van Daele, Laura E Kilpatrick, et al. "Characterisation of tyrosine kinase inhibitor-receptor interactions at VEGFR2 using sunitinib-red and nanoBRET." Biochem Pharmacol. 2023 Aug;214:115672. PMID: 37406966
- 10. Jia-Qi Guo, Chang-Dong Wang, et al. "PDGF‐BB/PDGFRβ Promotes Epithelial‐Mesenchymal Transition by Affecting PI3K/AKT/mTOR‐Driven Aerobic Glycolysis in Wilms' tumor G401 Cells." Cell Biol Int. 2022 Feb 14. PMID: 35165984
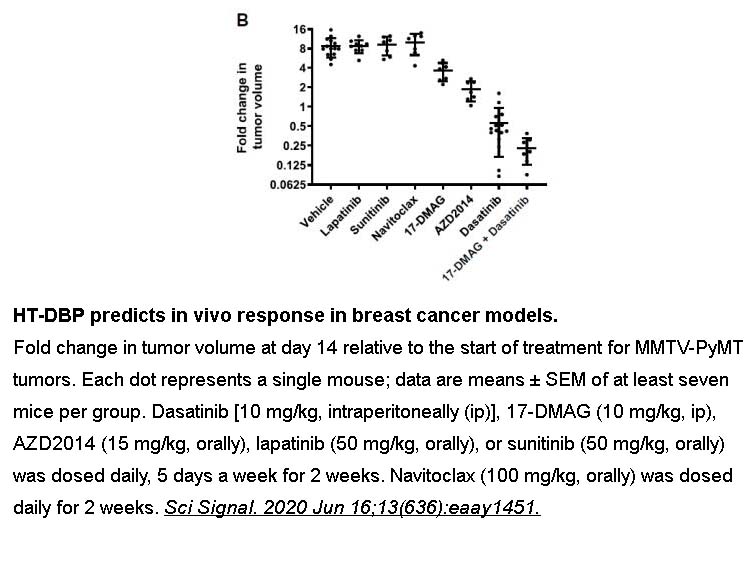
- 11. Bhola PD, Ahmed E, et al. "High-throughput dynamic BH3 profiling may quickly and accurately predict effective therapies in solid tumors." Sci Signal. 2020;13(636):eaay1451 PMID: 32546544
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 398.47 |
| Cas No. | 557795-19-4 |
| Formula | C22H27FN4O2 |
| Solubility | insoluble in H2O; ≥19.9 mg/mL in DMSO with gentle warming; ≥3.16 mg/mL in EtOH with gentle warming |
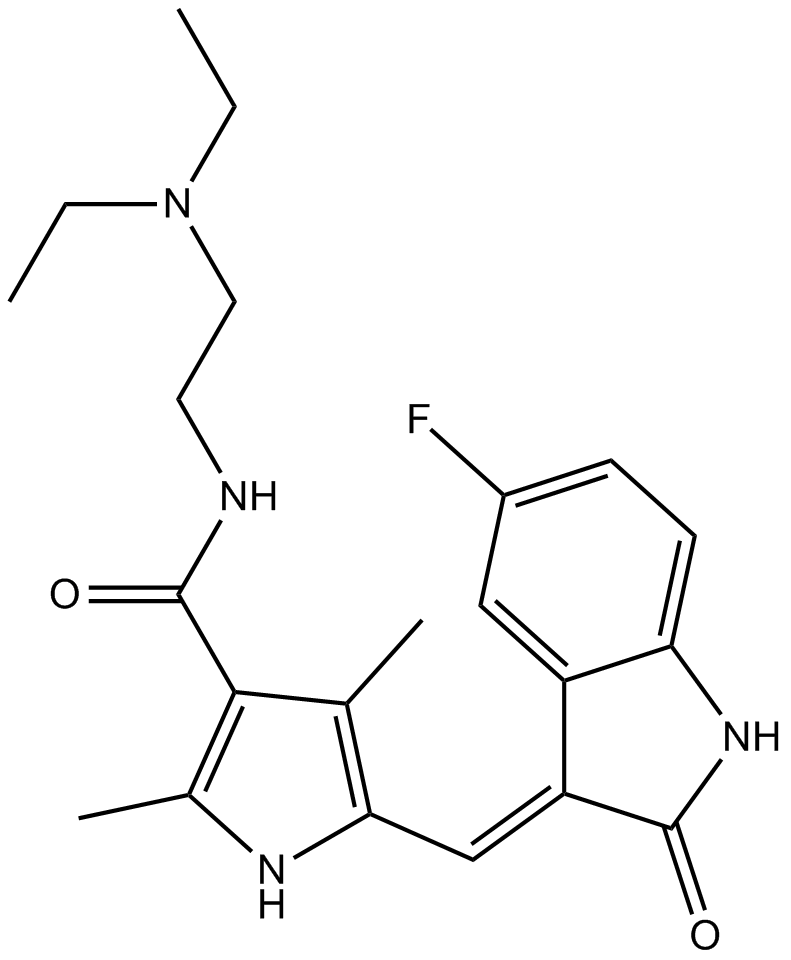
| Chemical Name | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fluoro-2-oxo-1H-indol-3-ylidene)methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide |
| Canonical SMILES | CCN(CC)CCNC(c1c(C)[nH]c(C=C(c(cc(cc2)F)c2N2)C2=O)c1C)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment: [1] | |
|
Cell lines |
Human 786-O and RCC4 cells, murine Renca cells |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while.Stock solution can be stored below -20°C for several months. |
|
Reaction Conditions |
10 μM, 24 hours |
|
Applications |
Sunitinib induced RCC tumor cell apoptosis in all three tumor cell lines. It also inhibited cell proliferation in a dose-dependent manner. For concentrations at which sunitinib caused effective tumor cell death; there were corresponding increases in cleaved PARP. Sunitinib treatment (24 h) of 786-O, RCC4 and Renca tumor cells reduced expression of several key anti-apoptotic and pro-proliferation genes, including Cyclin E, Cyclin D1 and Survivin. |
| Animal experiment: [1] | |
|
Animal models |
Female BALB/c mice injected with Renca cells |
|
Dosage form |
Oral administration, 40, 20, 10 mg/kg body weight, daily |
|
Applications |
Sunitinib induced tumor cell apoptosis in vivo as early as 1 day post treatment, which occurred in the presence of apparently intact tumor vessels. There appeared to be more apoptosis in the tumor on days 3 and 11 post treatment, with greater disruption of the tumor vasculature. Sunitinib treatment reduced Stat3 activity and induced tumor cell death as early as one day post treatment. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Xin H, Zhang C, Herrmann A, et al. Sunitinib inhibition of Stat3 induces renal cell carcinoma tumor cell apoptosis and reduces immunosuppressive cells. Cancer research, 2009, 69(6): 2506-2513. |
|
| Description | Sunitinib is an oral, multi-targeted and small-molecule inhibitor of receptor tyrosine kinase (RTK). | |||||
| Targets | VEGFR-1 | VEGFR-2 | VEGFR3 | PDGFRα | PDGFRβ | c-kit |
| IC50 | 4 nM | 69 nM | 39 nM | 1-10 nM | ||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data