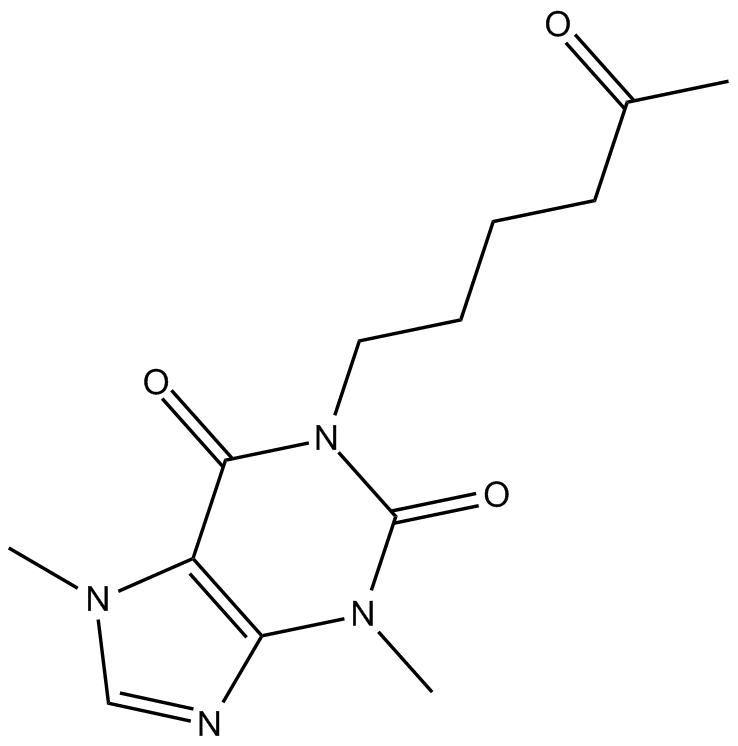
Pentoxifylline
Pentoxifylline (CAS No. 6493-05-6) is a methylxanthine derivative whose core biological functions include non-specifically inhibiting the activity of phosphodiesterase (PDE, especially PDE IV), increasing intracellular cAMP levels, thereby exerting anti-inflammatory, immunomodulatory, blood circulation-improving, and cell function-protecting effects. It can be used as an adjuvant therapy for diseases such as psoriasis, neonatal sepsis, male infertility, leishmaniasis, and HTLV-1-associated myelopathy (HAM/TSP); its target is phosphodiesterase (PDE). By inhibiting PDE activity, it reduces cAMP degradation, indirectly inhibits the activation of transcription factors such as NF-κB and NF-AT, downregulates the release of pro-inflammatory cytokines including TNF-α, IL-1β, IL-6, and IFN-γ, and simultaneously downregulates ICAM-1 expression in monocytes and the TLR4 signaling pathway. The IC₅₀ value for its inhibition of NO production in macrophages is 2.4-2.9 mM; it can be used for the treatment of animal disease models, with common models including imiquimod-induced psoriasis mouse models, LPS-stimulated inflammation models, SEB-induced macrophage activation models, and Leishmania infection models.
Common applications and concentrations: In cell culture, a concentration of 0.5-5 mM (incubation for 10-72 hours) is commonly used for PBMCs, RAW 264.7 macrophages, etc., in experiments such as inflammatory factor inhibition and sperm motility improvement; in animal experiments, mice/rats are given 400 mg/kg/day orally (divided into 3 doses), 14 mg/kg via intraperitoneal injection, and 5 mg/kg/h via intravenous infusion for neonatal sepsis models; in clinical practice, the conventional dose for adults is 400 mg orally three times a day. For psoriasis treatment, it can be formulated into liposomes with cyclosporine for transdermal administration, and the infusion dose for neonatal sepsis can be adjusted according to body weight; effective therapeutic concentrations: at the cellular level, 1-5 mM can significantly inhibit the secretion of pro-inflammatory factors and improve sperm motility; at the animal level, 400 mg/kg/day can improve psoriasis-induced skin damage and reduce inflammatory factor levels; in clinical practice, 400 mg three times a day can relieve symptoms of peripheral vascular diseases, shorten the healing time of mucosal lesions when used as an adjuvant therapy for leishmaniasis (with a cure rate of 90%-100% when combined with antimony agents), and in vitro incubation with 1-3.6 mM can enhance sperm survival rate and motility in the treatment of male infertility.
References:
[1] Neuner P, Klosner G, Pourmojib M, Knobler R, Schwarz T. Pentoxifylline in vivo and in vitro down-regulates the expression of the intercellular adhesion molecule-1 in monocytes. Immunology. 1997 Mar;90(3):435-9. doi: 10.1111/j.1365-2567.1997.00435.x. PMID: 9155652; PMCID: PMC1456607.
[2] Beshay E, Croze F, Prud'homme GJ. The phosphodiesterase inhibitors pentoxifylline and rolipram suppress macrophage activation and nitric oxide production in vitro and in vivo. Clin Immunol. 2001 Feb;98(2):272-9. doi: 10.1006/clim.2000.4964. PMID: 11161985.
[3] Ribeiro de Jesus A, Luna T, Pacheco de Almeida R, Machado PR, Carvalho EM. Pentoxifylline down modulate in vitro T cell responses and attenuate pathology in Leishmania and HTLV-I infections. Int Immunopharmacol. 2008 Oct;8(10):1344-53. doi: 10.1016/j.intimp.2008.03.020. Epub 2008 Apr 28. PMID: 18687297.
[4] Schüller SS, Wisgrill L, Herndl E, Spittler A, Förster-Waldl E, Sadeghi K, Kramer BW, Berger A. Pentoxifylline modulates LPS-induced hyperinflammation in monocytes of preterm infants in vitro. Pediatr Res. 2017 Aug;82(2):215-225. doi: 10.1038/pr.2017.41. Epub 2017 May 24. PMID: 28288151.
[5] Mahaldashtian M, Khalili MA, Nottola SA, Woodward B, Macchiarelli G, Miglietta S. Does in vitro application of pentoxifylline have beneficial effects in assisted male reproduction? Andrologia. 2021 Feb;53(1):e13722. doi: 10.1111/and.13722. Epub 2020 Oct 28. PMID: 33112447.
[6] Bhardwaj P, Tripathi P, Pandey S, Gupta R, Ramchandra Patil P. Cyclosporine and Pentoxifylline laden tailored niosomes for the effective management of psoriasis: In-vitro optimization, Ex-vivo and animal study. Int J Pharm. 2022 Oct 15;626:122143. doi: 10.1016/j.ijpharm.2022.122143. Epub 2022 Aug 28. PMID: 36037986.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 278.31 |
| Cas No. | 6493-05-6 |
| Formula | C13H18N4O3 |
| Solubility | ≥14 mg/mL in EtOH; ≥19.55 mg/mL in H2O; ≥27.91 mg/mL in DMSO |
| Chemical Name | 3,7-dimethyl-1-(5-oxohexyl)-1H-purine-2,6(3H,7H)-dione |
| Canonical SMILES | CC(CCCCN1C(N(C)C(N=CN2C)=C2C1=O)=O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure