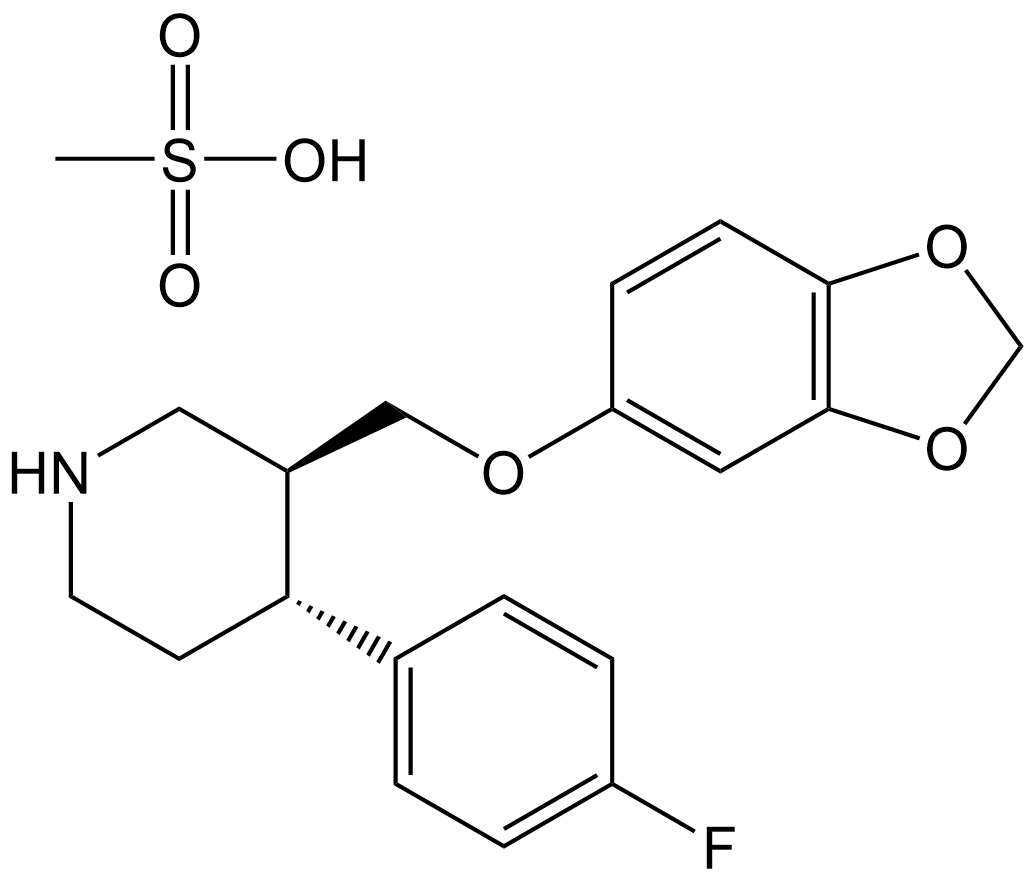
Paroxetine Mesylate
Paroxetine Mesylate (Paroxetine Mesylate, CAS No. 217797-14-3), as a selective 5 - hydroxytryptamine reuptake inhibitor (SSRI), has core biological functions spanning multiple areas including the treatment of psychiatric disorders, anti-tumor activity, cardiovascular regulation, and potential antiviral effects. It has broad targets and well-defined mechanisms: it exhibits extremely high binding affinity for the 5 - hydroxytryptamine transporter (SERT) (about 70.2±0.6 pM), blocks synaptic cleft 5 - hydroxytryptamine reuptake by inhibiting SERT, and can also inhibit the cytochrome P450 enzyme family (CYP2D6 Ki=0.065, CYP2B6 Ki=1.03μM), G protein-coupled receptor kinase 2 (GRK2, IC50=1.4μM). It can also target receptor tyrosine kinases (MET, ERBB3) and Ebola virus glycoprotein (GP, pKi≈3.19), and shows inhibitory activity against kinases such as KIT and JAK as well (IC50 mostly in the nanomolar to micromolar range).
In cell culture, its anti-colorectal cancer activity is significant. The IC50 values for HCT116 cells at 1 day and 2 days are 26.49μmol/L and 13.50μmol/L, respectively; for HT29 cells they are 14.22μmol/L and 7.01μmol/L, respectively. A concentration of 5μmol/L can inhibit 50% of colony formation, and 20μmol/L can induce apoptosis in HCT116 and HT29 cells (apoptosis rates of 5% and 13%, respectively), and inhibit 3D spheroid formation.
In animal experiments, common applications include the HT29 cell xenograft nude mouse colorectal cancer model, the epileptic baboon spontaneous generalized epilepsy model (used for SUDEP-related cardiac biomarker research), and canine aggression and stereotypy models. In the colorectal cancer model, intraperitoneal injection at 1mg/kg or 5mg/kg (3 times per week for 2 weeks) can significantly inhibit tumor growth. In the epilepsy model, no exploration of therapeutic concentrations was involved. Canine applications use routine veterinary clinical doses.
In clinical use, oral administration is used to treat psychiatric disorders such as major depressive disorder, obsessive-compulsive disorder, and social anxiety disorder. The conventional dose range is 20-60mg daily (controlled-release formulation or immediate-release tablets). At doses of 40mg/day and above, it can exert dual reuptake inhibition of 5 - hydroxytryptamine / norepinephrine. It can also be used off-label for the treatment of related psychiatric disorders in children and adolescents, and can be used for menopausal vasomotor symptoms, diabetic neuropathy, etc. Its metabolism is mainly mediated by hepatic CYP2D6. Steady-state concentrations are achieved after 4-14 days of repeated dosing, with no obvious accumulation. Bioavailability is affected by the hepatic first-pass effect but can be increased through repeated dosing.
References:
[1] Jang WJ, Jung SK, Vo TTL, Jeong CH. Anticancer activity of paroxetine in human colon cancer cells: Involvement of MET and ERBB3. J Cell Mol Med. 2019 Feb;23(2):1106-1115. doi: 10.1111/jcmm.14011. Epub 2018 Nov 13. PMID: 30421568; PMCID: PMC6349215.
[2] Szabó CÁ, Akopian M, González DA, de la Garza MA, Carless MA. Cardiac biomarkers associated with epilepsy in a captive baboon pedigree. Epilepsia. 2019 Nov;60(11):e110-e114. doi: 10.1111/epi.16359. Epub 2019 Oct 8. PMID: 31592545; PMCID: PMC7054851.
[3] Kowalska M, Nowaczyk J, Fijałkowski Ł, Nowaczyk A. Paroxetine-Overview of the Molecular Mechanisms of Action. Int J Mol Sci. 2021 Feb 7;22(4):1662. doi: 10.3390/ijms22041662. PMID: 33562229; PMCID: PMC7914979.
| Storage | Store at -20°C |
| M.Wt | 425.47 |
| Cas No. | 217797-14-3 |
| Formula | C20H24FNO6S |
| Synonyms | BRL29060 mesylate |
| Chemical Name | (3S,4R)-3-((benzo[d][1,3]dioxol-5-yloxy)methyl)-4-(4-fluorophenyl)piperidine methanesulfonate |
| Canonical SMILES | CS(O)(=O)=O.FC1=CC=C(C=C1)[C@@H]1CCNC[C@H]1COC1=CC2=C(OCO2)C=C1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |