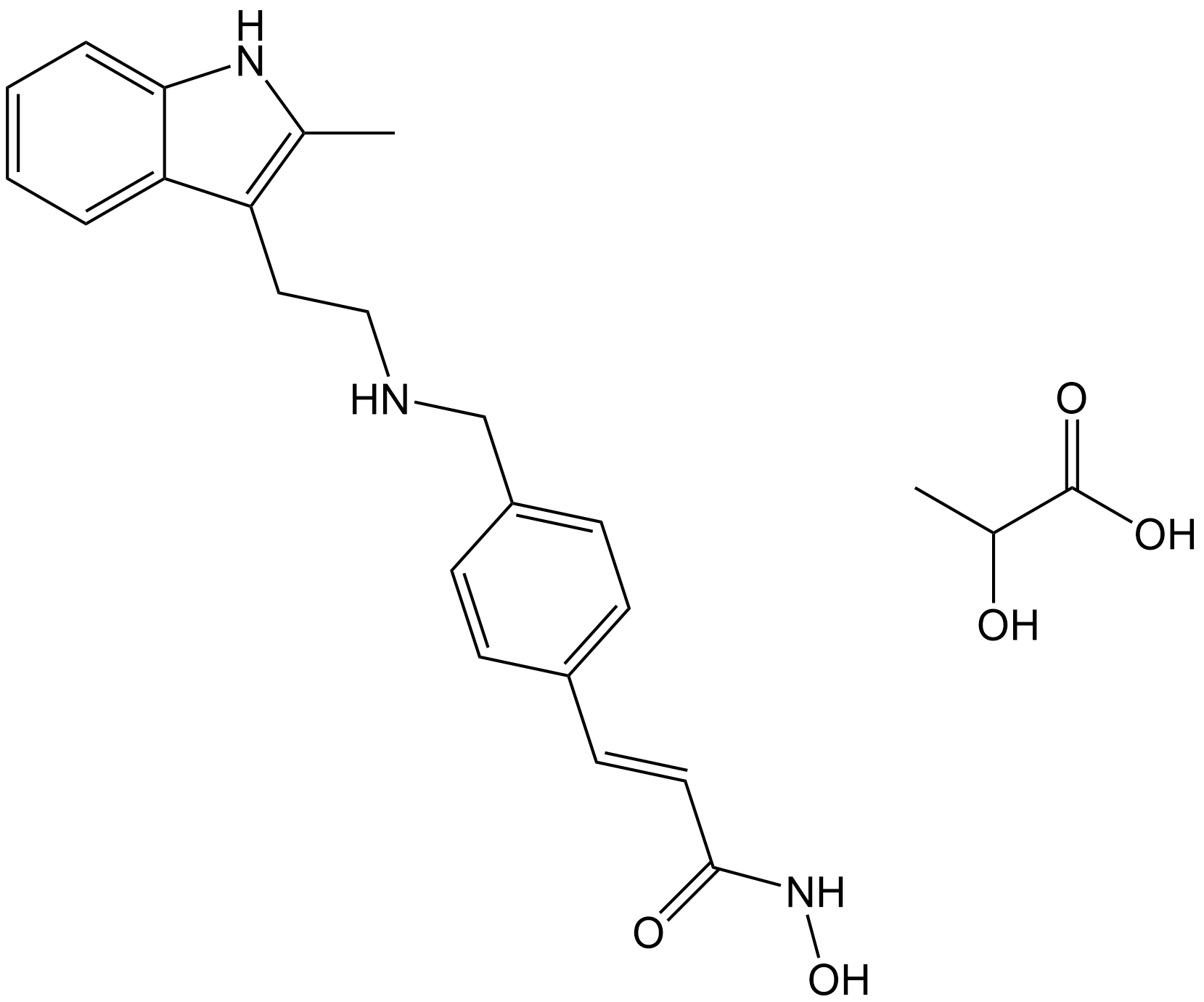
Panobinostat Lactate
Panobinostat Lactate (panobinostat, alias LBH589, CAS No. 960055-56-5) is a broad-spectrum histone deacetylase (HDAC) inhibitor. Its core activity is the potent inhibition of the enzymatic activity of nearly all HDAC family isoforms (IC50 values in the low nanomolar range). Inhibition of HDAC6 can lead to hyperacetylation of heat shock protein 90 (HSP90), thereby inhibiting its molecular chaperone function and promoting proteasomal degradation of the calcineurin catalytic subunit PPP3CA. It can also deplete histone H2B monoubiquitination at lysine 120 (H2BK120ub1) by downregulating expression of the RNF20/RNF40/WAC E3 ubiquitin ligase complex. In addition, it can inhibit the NF-κB signaling pathway and block osteoclast differentiation. When combined with the protein disulfide isomerase (PDI) inhibitor, it synergistically upregulates transcription of key effector factors in the endoplasmic reticulum stress pathway, including ATF3, DDIT3/CHOP, and DNAJB1, ultimately exerting therapeutic anti-proliferative and pro-apoptotic activity in hematologic malignancies.
In in vitro cell experiments, the single-agent EC50 values against multiple myeloma (MM) cell lines KMS11, OPM2, MM.1S BzR, and ANBL6 were 11.3 nM, 16.6 nM, 20.9 nM, and 12.0 nM, respectively. After combination with 1 μM PDI inhibitor LTI6426, the EC50 values decreased to 1.5 nM, 0.6 nM, 0.7 nM, and 1.9 nM, respectively, increasing tumor cell killing sensitivity by 6.3–30-fold, with no synergistic toxicity to normal human peripheral blood mononuclear cells (PBMCs); the single-agent EC50 was 9.6 nM. For MLL-rearranged acute lymphoblastic leukemia (ALL) cell lines SEM and KOPN8, treatment at 5 nM for 4 days significantly induced cell growth arrest and apoptosis. In primary infant ALL patient cells, 5–10 nM for 48 h induced apoptosis in a time- and dose-dependent manner, and this effective concentration range showed no obvious cytotoxicity to normal B cells, indicating a favorable therapeutic window.
In animal experiments, this drug has been used for therapeutic studies in malignant hematologic tumor models rather than for disease model induction. Commonly used models include NSG mouse xenograft models of MLL-rearranged ALL (from cell lines or primary patient cells), NSG mouse xenograft models of proteasome inhibitor–resistant MM, and NOD/SCID mouse subcutaneous xenograft models of MM. It has also been applied to mouse bone marrow–derived osteoclast differentiation models to evaluate its bone-protective effects. Regarding effective in vivo therapeutic doses, in ALL models, 5 mg/kg intraperitoneal injection (an intermittent regimen of 5 days on, 2 days off) significantly prolonged survival of tumor-bearing mice and reduced leukemia burden. In MM models, 4–20 mg/kg intraperitoneal injection (3 times per week or intermittent dosing within a 21-day cycle) inhibited tumor growth as a single agent; combination with LTI6426, bortezomib, or FK506 further improved in vivo antitumor efficacy. Tolerability was good at low doses, with no significant severe toxicity such as marked body weight loss.
In clinical use, this drug has been approved by the U.S. FDA and the European EMA for the treatment of relapsed/refractory multiple myeloma. The approved regimen is in combination with bortezomib and dexamethasone. A commonly used clinical oral dose is 20 mg, which can achieve nanomolar peak plasma concentrations in humans to exert therapeutic effects, with potential benefit particularly in bortezomib-resistant patients with high PPP3CA expression.
References:
[1] Imai Y, Ohta E, Takeda S, Sunamura S, Ishibashi M, Tamura H, Wang YH, Deguchi A, Tanaka J, Maru Y, Motoji T. Histone deacetylase inhibitor panobinostat induces calcineurin degradation in multiple myeloma. JCI Insight. 2016 Apr 21;1(5):e85061. doi: 10.1172/jci.insight.85061. PMID: 27699258; PMCID: PMC5033869.
[2] Garrido Castro P, van Roon EHJ, Pinhanços SS, Trentin L, Schneider P, Kerstjens M, Te Kronnie G, Heidenreich O, Pieters R, Stam RW. The HDAC inhibitor panobinostat (LBH589) exerts in vivo anti-leukaemic activity against MLL-rearranged acute lymphoblastic leukaemia and involves the RNF20/RNF40/WAC-H2B ubiquitination axis. Leukemia. 2018 Feb;32(2):323-331. doi: 10.1038/leu.2017.216. Epub 2017 Jul 10. PMID: 28690313.
[3] Robinson RM, Basar AP, Reyes L, Duncan RM, Li H, Dolloff NG. PDI inhibitor LTI6426 enhances panobinostat efficacy in preclinical models of multiple myeloma. Cancer Chemother Pharmacol. 2022 May;89(5):643-653. doi: 10.1007/s00280-022-04425-3. Epub 2022 Apr 5. PMID: 35381875; PMCID: PMC9054865.
| Storage | 4℃, sealed storage, away from moisture |
| M.Wt | 439.5 |
| Cas No. | 960055-56-5 |
| Formula | C21H23N3O2•C3H6O3 |
| Synonyms | LBH589 lactate; NVP-LBH589 lactate |
| Chemical Name | (E)-N-hydroxy-3-(4-(((2-(2-methyl-1H-indol-3-yl)ethyl)amino)methyl)phenyl)acrylamide 2-hydroxypropanoate |
| Canonical SMILES | C(CNCC1=CC=C(/C=C/C(NO)=O)C=C1)C=2C=3C(NC2C)=CC=CC3.C(C(O)=O)(C)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |