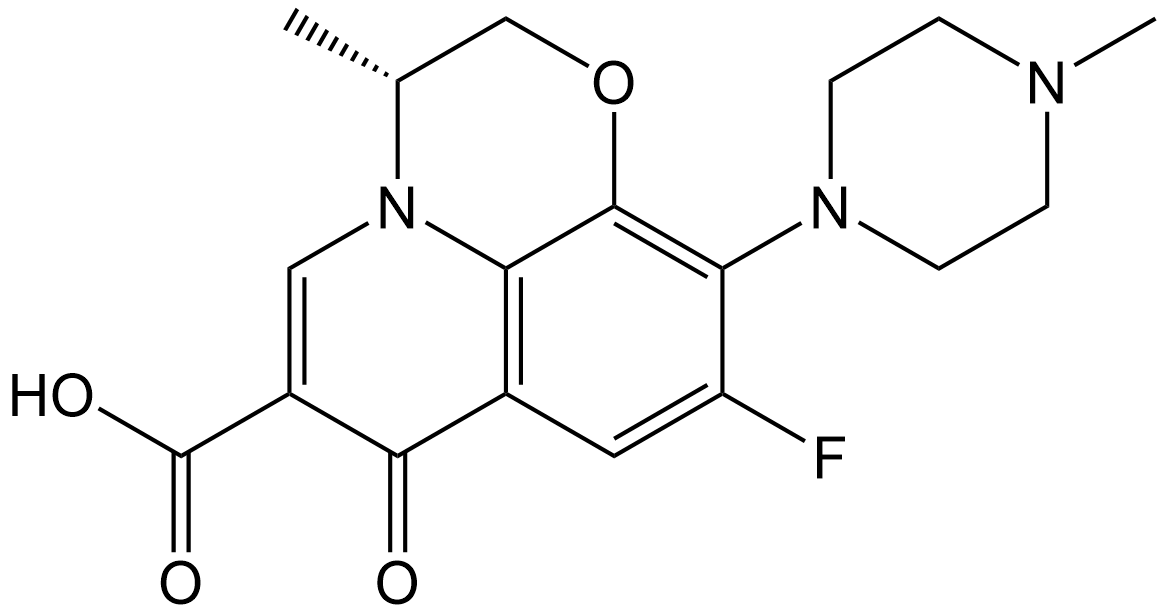
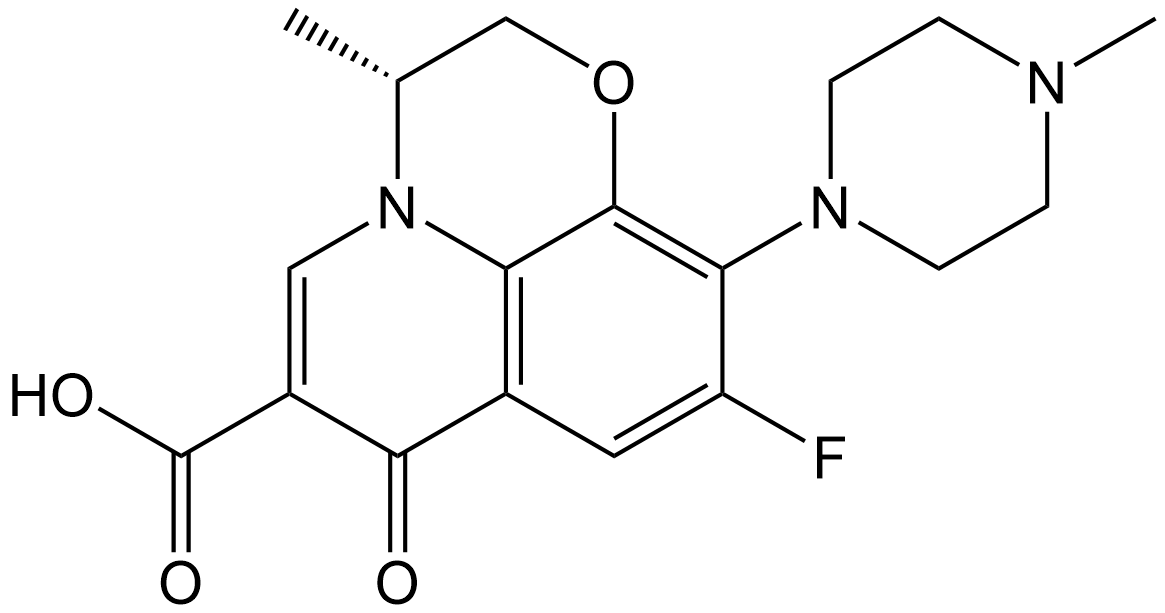
(+)-Ofloxacin
(R)-(+)-ofloxacin (CAS No. 100986-86-5) is the R-enantiomer of the fluoroquinolone antibacterial drug ofloxacin. Its core biological function is to form a complex by binding bacterial DNA and DNA topoisomerase II (DNA gyrase), thereby inhibiting DNA strand annealing and bacterial replication; however, its antibacterial activity is only 1/8~1/128 that of S-(-)-ofloxacin (CAS No. 82419-36-1, Cat. No.: A5511), with no standalone therapeutic value, and it is often used together with the racemate. Its binding to DNA polynucleotides is selective: it only binds to poly [d (G-C)₂], with binding constants Ksv of 430 M⁻¹ and KBH of 1599 M⁻¹, and shows no significant interaction with poly [d (A-T)₂] or poly [d (I-C)₂]. Its antibacterial MIC values are significantly higher than those of S-(-)-ofloxacin (the latter has an MIC₉₀ of 0.07 mg/L against Escherichia coli). Common application concentrations: 1 μM in vitro for binding-characteristic studies; in animal experiments, the rat intestinal perfusion concentration is 0.346 mM (0.125 mg/ml, racemate component). After oral administration of the racemate at 20 mg/kg in red sea bream, the peak concentration (Cmax) of R-(+)-ofloxacin is 8.13 ± 0.22 μg/mL and the elimination half-life (t₁/₂) is 12.33 ± 0.45 h. After clinical intravenous infusion of 400 mg racemate (administered 1 hour preoperatively), the Cmax of R-(+)-ofloxacin is 2.68 ± 1.58 mg/L, AUC₀-∞ is 20.50 ± 2.06 mg・h/L, and clearance is 0.16 ± 0.04 L/h/Kg; it is mainly used for antibacterial prophylaxis in biliary surgery (relying on S-(-)-ofloxacin in the racemate to play the core role). Animal modeling is commonly seen in rat intestinal absorption models and red sea bream pharmacokinetic models (both analyze separated enantiomers after racemate dosing). Its in vivo exposure (AUC₀-∞ of 80.46 ± 1.67 μg・h/mL in red sea bream) is higher than that of S-(-)-ofloxacin, but due to weak activity, it has no effective standalone therapeutic concentration and needs to rely on S-(-)-ofloxacin to maintain antibacterial effects.
References:
[1] Rabbaa L, Dautrey S, Colas-Linhart N, Carbon C, Farinotti R. Absorption of ofloxacin isomers in the rat small intestine. Antimicrob Agents Chemother. 1997 Oct;41(10):2274-7. doi: 10.1128/AAC.41.10.2274. PMID: 9333061; PMCID: PMC164106.
[2] Gascón AR, Campo E, Hernández RM, Calvo B, Errasti J, Pedraz Muñoz JL. Pharmacokinetics of ofloxacin enantiomers after intravenous administration for antibiotic prophylaxis in biliary surgery. J Clin Pharmacol. 2000 Aug;40(8):869-74. doi: 10.1177/00912700022009602. PMID: 10934671.
[3] Hwangbo HJ, Yun BH, Cha JS, Kwon DY, Kim SK. Enantioselective binding of S- and R-ofloxacin to various synthetic polynucleotides. Eur J Pharm Sci. 2003 Feb;18(2):197-203. doi: 10.1016/s0928-0987(02)00279-8. PMID: 12594014.
[4] Nie J, Wang YG, Gao XF, OuYang XK, Yang LY, Yu D, Wu WJ, Xu HP. Pharmacokinetic study of ofloxacin enantiomers in Pagrosomus major by chiral HPLC. Biomed Chromatogr. 2016 Mar;30(3):426-31. doi: 10.1002/bmc.3565. Epub 2015 Aug 27. PMID: 26179954.
| Storage | Store at 4°C |
| M.Wt | 361.37 |
| Cas No. | 100986-86-5 |
| Formula | C18H20FN3O4 |
| Synonyms | Dextrofloxacin; R-Ofloxacin; D-Ofloxacin |
| Solubility | insoluble in DMSO; insoluble in EtOH; ≥25.35 mg/mL in H2O with ultrasonic |
| Chemical Name | (R)-9-fluoro-3-methyl-10-(4-methylpiperazin-1-yl)-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid |
| Canonical SMILES | O=C1C=2C3=C(C(=C(F)C2)N4CCN(C)CC4)OC[C@@H](C)N3C=C1C(O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure