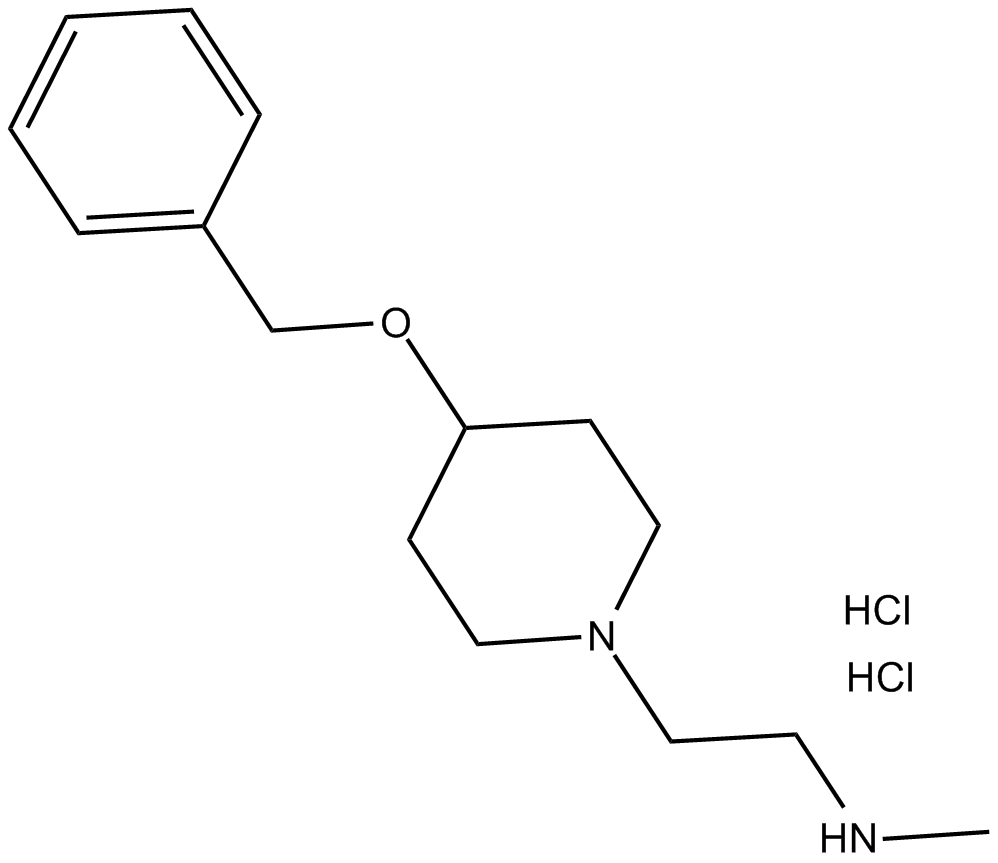
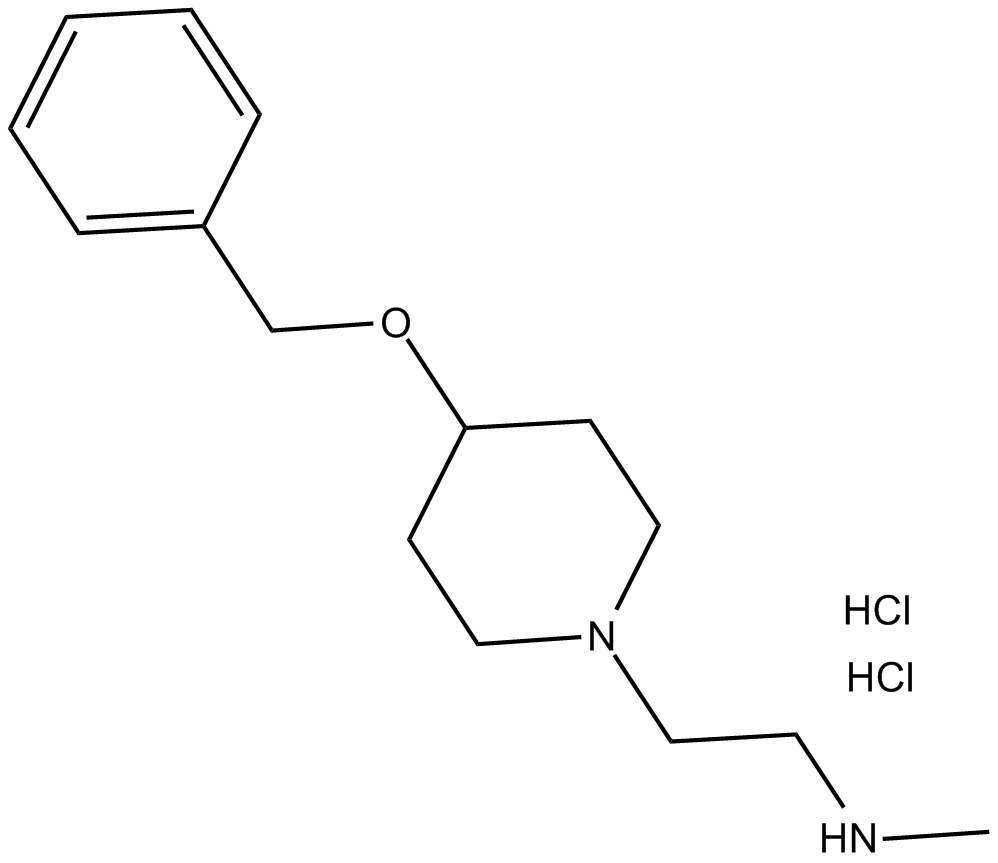
MS049 (hydrochloride)
IC50: 34 nM for PRMT4; 43 nM for PRMT6
MS049 is a dual PRMT4 and PRMT6 inhibitor.
PRMTs have been reported to have a key role in the regulation of the arginine methylation pattern and level of a plethora of different substrates, including both histones and non-histone proteins. Therefore, the dysregulation of PRMTs has been linked to various human diseases.
In vitro: Previous study evaluated selectivity of MS049 and its two close analogs against other PRMTs. It was found that none of these compounds showed inhibition against PRMT5 and PRMT7. In addition, all three compounds were more potent against PRMT4 and PRMT6 than other type I PRMTs. MS049 showed good selectivity over PRMT8 (>30-fold) and excellent selectivity over PRMT1 and PRMT3 (>300-fold). Moreover, MS049 could reduce the H3R2me2a mark in HEK293 cells in a concentration dependent manner. The effect of the 8 μM MS049 treatment matched with that of the catalytically inactive mutant. In addition, MS049 treatment was able to inhibit endogenous PRMT4 methyltransferase activity in a concentration dependent manner leading to reduced levels of cellular asymmetric arginine dimethylation of Med12 in HEK293 cells [1].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Shen Y1 et al. Discovery of a Potent, Selective, and Cell-Active Dual Inhibitor of Protein Arginine Methyltransferase 4 and Protein Arginine Methyltransferase 6. J Med Chem. 2016 Oct 13;59(19):9124-9139.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 321.3 |
| Formula | C15H24N2O·2HCl |
| Solubility | ≤30mg/ml in ethanol;30mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | N-methyl-4-(phenylmethoxy)-1-piperidineethanamine, dihydrochloride |
| Canonical SMILES | CNCCN(CC1)CCC1OCc1ccccc1.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure