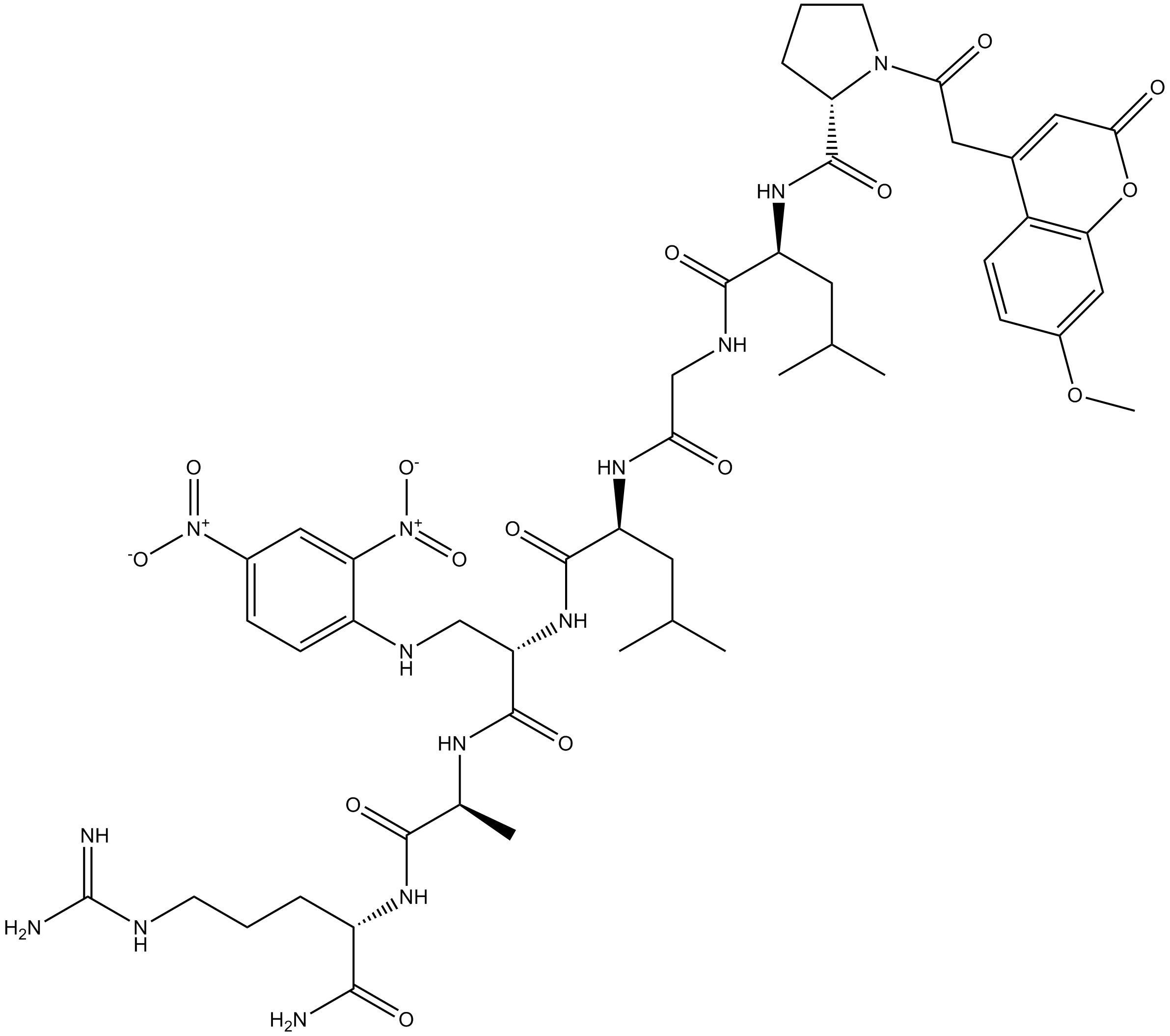
7-Methoxycoumarin-4-acetyl-P-L-G-L-β-(2,4-dinitrophenylamino)A-R amide
7-Methoxycoumarin-4-acetyl-P-L-G-L-beta-24-dinitrophenylamino A-R amide (CAS: 140430-53-1) is a fluorogenic peptide substrate targeting matrix metalloproteinases (MMPs). It is designed to enable the continuous measurement of MMP enzymatic activity, which is achieved by monitoring substrate cleavage between Gly and Leu residues. 7-Methoxycoumarin-4-acetyl-P-L-G-L-beta-24-dinitrophenylamino A-R amide exerts its utility through a fluorescence-based mechanism: cleavage by MMPs separates the fluorescent reporter group from the quencher, resulting in increased fluorescence signal. Based on these properties, 7-Methoxycoumarin-4-acetyl-P-L-G-L-beta-24-dinitrophenylamino A-R amide is employed in biochemical assays for the quantification of total or specific MMP activity in biological samples and enzyme preparations.
References:
1. Schectcr, I, and Beryer. A. (lYG7) Biochem. Biophyt;. Rcs, Commun. 27, 157-161
2. Nctzcl-Arnctt, s.. Mallya. SK., Nugasc. H.. Birkedal-Hansen, H. and Van Wurt. H.E. (1991) Anal. Biochcm. 195. 86-92.
3. Netzel-Arnctt, S., Fields. G., Birkedal-Hunscn. H. and Vai Wart. H.E. (1991) J. Biol. Chem. 266. 6747-6755
4. C.G.Knight, F. Willenbrock and G. Murphy. A novel coumarin-labelled peptide for sensitive continuous assays of the matrix metalloproteinases. FEDS. Volume 296, number 3, 263-266
| Storage | Store at -20°C |
| M.Wt | 1093.15 |
| Cas No. | 140430-53-1 |
| Formula | C49H68N14O15 |
| Solubility | ≥48.7 mg/mL in DMSO,insoluble in EtOH,insoluble in H2O |
| Chemical Name | (2R)-N-((6S,9S,12S,16R,22R)-1,12-diamino-6-carbamoyl-13-(2,4-dinitrophenyl)-1-imino-16-isobutyl-9,24-dimethyl-8,11,15,18,21-pentaoxo-2,7,10,14,17,20-hexaazapentacosan-22-yl)-1-(2-(7-methoxy-2-oxo-2H-chromen-4-yl)acetyl)pyrrolidine-2-carboxamide |
| Canonical SMILES | COC1=CC=C(C(CC(N2CCC[C@@H]2C(N[C@H](CC(C)C)C(NCC(N[C@H](CC(C)C)C(NC([C@H](C(N[C@@H](C(N[C@@H](C(N)=O)CCCNC(N)=N)=O)C)=O)N)C3=CC=C([N+]([O-])=O)C=C3[N+]([O-])=O)=O)=O)=O)=O)=O)=CC(O4)=O)C4=C1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure