Lambda Protein Phosphatase (RNase-free)
Lambda Protein Phosphatase (Lambda PP) is a Mn²⁺-dependent protein phosphatase that dephosphorylates phosphorylated serine, threonine, and tyrosine residues. It is the product of open reading frame ORF221 of lambda phage, encoding 221 amino acid residues. Lambda PP can be used for protein dephosphorylation to study the relationship between protein phosphorylation and its activity or structure, as well as to validate the specificity of phosphorylation site-specific antibodies, among other applications.
The concentration of this Lambda Protein Phosphatase product is 100 U/μL. In a 50 μL reaction system, 100 U of the enzyme can completely remove the phosphate groups from the equivalent of 5 μM mono-phosphorylated protein (i.e., 0.25 nmol) within 30 minutes. Based on this calculation, a total enzyme amount of 20 kU (20,000 U) can process approximately 50 nmol of mono-phosphorylated protein substrate, with the actual processing capacity depending on reaction conditions and substrate characteristics.
Product parameters:
|
Name |
Lambda Protein Phosphatase (RNase-free) |
|
Synonym |
λ-PPase, Lambda PP, Serine/threonine-protein phosphatase, Protein Phosphatase, Lambda |
|
Recognition/Cleavage Site |
No specific recognition sequence; dephosphorylates phosphoserine (pSer), phosphothreonine (pThr), and phosphotyrosine (pTyr) residues |
|
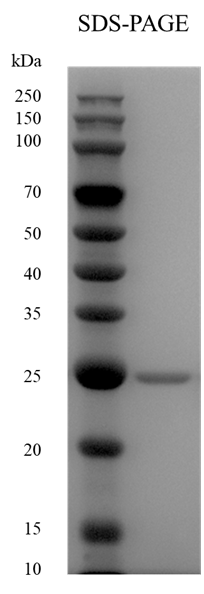
M.Wt |
~ 25 kDa |
|
Source |
E. coli |
|
Label |
Tag free |
|
Purity |
> 95%, determined by SDS-PAGE. |
|
Tev Protease Buffer |
50 mM HEPES (pH 7.5 @ 25°C), 100 mM NaCl, 2 mM DTT, 0.01% Brij 35, 0.1 mM EGTA, 0.1 mM MnCl2, 50% Glycerol. |
|
10×TEV Protease Reaction Buffer |
500 mM HEPES (pH 7.5 @ 25°C), 1 M NaCl,20 mM DTT, 0.1% Brij 35. |
|
Enzyme activity |
10 U/μL |
|
Unit Definition |
One unit (U) of activity represents the enzyme quantity needed to hydrolyze 1 nmol of p-nitrophenyl phosphate (50 mM) per minute in a 50 μL reaction system at 30°C. |
|
Optimal pH range |
pH 7.0 - 8.0 (optimal at pH 7.5) |
|
Reaction Temperature |
30°C |
|
Inactivation Condition |
Inactivate at 65°C for 1 hour in 50 mM EDTA. |
- 1. Wenzhen Gao, Li Zhu, et al. "BMAL1 regulates circadian rhythms via phase separation–mediated transcriptional hub formation." Signal Transduct Target Ther. 2026 May 1;11(1):160. PMID: 42062246
- 2. Qiaoxi Xia, Ronghui Zhong, et al. "PRMT5-mediated methylation of METTL3 promotes cisplatin resistance in ovarian cancer by facilitating DNA repair mechanisms." Cell Rep. 2025 Apr 22;44(4):115484. PMID: 40158218
- 3. Mengjun Zhang, Zhuang Gu, et al. "Phosphorylation-dependent charge blocks regulate the relaxation of nuclear speckle networks." Mol Cell. 2025 May 1;85(9):1760-1774.e7
| Components | 20 KU | 100 KU | 200 KU | Storage |
| Lambda PP (RNase-free) | 0.2 mL | 1 mL | 2 mL | -80°C |
| 10X Lambda PP Reaction Buffer | 1 mL | 5 x 1 mL | 10 x 1 mL | -80°C |
| 10X MnCl2 (10mM MnCl2) | 1 mL | 5 x 1 mL | 10 x 1 mL | -80°C |
Shipping: dry ice Shelf life: 2 years | ||||
Note: It is recommended to aliquot upon first use to avoid repeated freeze-thaw cycles: Store aliquots at -20°C for a validity period of 6 months.
Q: Is lambda protein phosphatase suitable for use in Phosbind assays (e.g., product code F4002)?
A: Yes. In Phosbind assays, lambda protein phosphatase can be used as a specific validation tool (e.g., as a positive control for in vitro kinase reactions) to confirm that the observed band shifts are indeed due to protein phosphorylation. By setting up a control group and treating the same sample with lambda protein phosphatase prior to electrophoresis, if the slow-migrating bands observed before treatment disappear or significantly decrease in signal after treatment, while fast-migrating bands increase, it demonstrates that the bands represent phosphorylated forms of the protein, thereby ruling out false positives caused by non-specific modifications or binding.
Q: What experimental applications is lambda protein phosphatase suitable for?
A: Validating protein phosphorylation modifications, verifying the specificity of phospho-specific antibodies, studying the relationship between protein phosphorylation and its activity and structure, and identifying phosphorylation peaks in Simple Western Charge Assays.
Q: What is the substrate specificity of lambda protein phosphatase?
A: The enzyme has dephosphorylation activity against phosphorylated serine (pSer), threonine (pThr), tyrosine (pTyr), and histidine (pHis) residues. It is a Mn²⁺-dependent dual-specificity phosphatase.
Q: Can lambda protein phosphatase be used on paraffin-embedded sections?
A: Not recommended.
Q: How should lambda protein phosphatase be stored? Does repeated freeze-thawing affect it?
A: Recommended storage temperature: –70°C (–80°C) for long-term storage; –20°C for short-term storage (e.g., up to 1 week). Avoid repeated freeze-thawing, as it leads to loss of enzyme activity. It is recommended to aliquot upon first use.
Q: What is the role of DTT in the reaction system?
A: DTT acts as a reducing agent to maintain the active conformation of the enzyme. The storage buffer typically contains 2 mM DTT, which is carried into the reaction system via the 1× reaction buffer.
Q: Which reagents inhibit or affect lambda protein phosphatase activity?
A: 10 mM sodium orthovanadate inhibits approximately 80–90% of activity; 50 mM EDTA inhibits approximately 95% of activity; 50 mM NaF combined with 10 mM sodium orthovanadate inhibits approximately 90% of activity.
Q: How is lambda protein phosphatase inactivated? How is the reaction terminated?
A: The enzyme can be inactivated by heating at 65°C for 1 hour in the presence of 50 mM EDTA. EDTA acts as a metal ion chelator, sequestering the Mn²⁺ required for enzyme activity, thereby inhibiting the enzyme.
Q: Which enzyme inhibitors are compatible with lambda protein phosphatase?
A: Compatible with PMSF, benzamidine, leupeptin, pepstatin A, aprotinin, and EDTA-free protease inhibitor cocktails. Not compatible with EDTA (chelates Mn²⁺), vanadate, sodium fluoride, or pyrophosphate.
Q: What causes incomplete cleavage or lack of dephosphorylation effect?
A: Possible reasons include: ① Insufficient enzyme amount: optimal enzyme concentration for different substrate proteins should be determined experimentally; ② Insufficient reaction time: incubation time can be extended as needed; ③ Lack of Mn²⁺: λ-PPase is Mn²⁺-dependent, and 1 mM MnCl₂ must be added to the reaction system; ④ Presence of inhibitors: 10 mM vanadate inhibits approximately 90% of activity, and 50 mM EDTA inhibits approximately 95% of activity; ⑤ Sample contains denaturants or high salt: certain reagents may interfere with enzyme activity.
Q: How can lambda protein phosphatase be removed using ultrafiltration tubes?
A: The molecular weight of lambda protein phosphatase is approximately 25 kDa. A 30 kDa ultrafiltration tube is not suitable for removing the enzyme (the molecular weight is lower than the cutoff, so the enzyme will pass into the filtrate). It is recommended to use a 10 kDa or smaller molecular weight cutoff ultrafiltration tube.