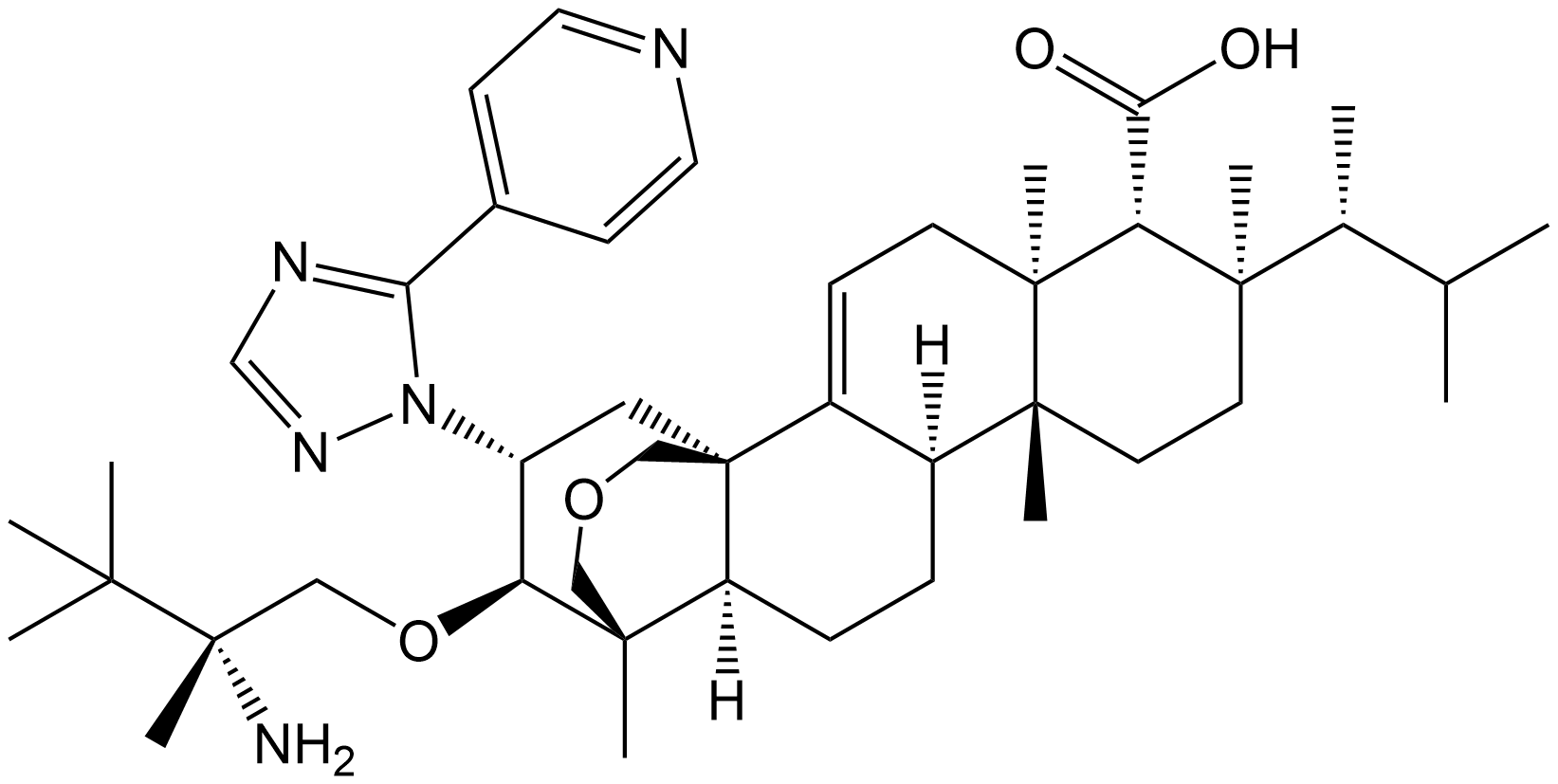
Ibrexafungerp
Ibrexafungerp (CAS: 1207753-03-4) is the first novel triterpenoid oral antifungal drug. Its core target is 1,3-β-D - glucan synthase in the fungal cell wall. By non-competitively inhibiting the enzymatic activity of this target, it blocks the synthesis of key components of the fungal cell wall to exert fungicidal effects. It shares the same target as echinocandins but differs in binding site, with only limited cross-resistance. In in vitro susceptibility testing, CLSI M27-A4 or EUCAST 7.3.2 broth microdilution is commonly used; the typical drug concentration range in the test system is 0.03~2mg/ml, incubated at 35℃ for 24h to read MIC values. The endpoint criteria are 80% fungal growth inhibition for the CLSI method and 50% fungal growth inhibition for the EUCAST method. For fluconazole-susceptible and -resistant Candida albicans, MIC90 is 0.03μg/ml under pH7.0 conditions, and MIC90 is 0.06μg/ml under the vaginal physiological acidic environment pH4.5 in VVC patients, indicating that antifungal activity is not negatively affected by low pH. Meanwhile, MIC90 values under pH4.5 for Candida glabrata, Candida krusei, Candida parapsilosis, and Candida tropicalis are 0.5mg/ml, 0.25mg/ml, 0.25mg/ml, and 0.25mg/ml, respectively. Against 192 echinocandin-resistant Candida isolates confirmed by genotype and phenotype, MIC50/90 is 0.5/2mg/L for Candida albicans and 2/>4mg/L for Candida glabrata, and activity is stronger against strains with central mutations in the FKS gene hotspot region (Candida albicans S645 mutant MIC50/90 is 0.25/1mg/L), while activity decreases significantly against strains with mutations at the start of the FKS gene hotspot region (Candida glabrata F659 mutant MIC50/90>4/>4mg/L). For 54 fluconazole-resistant Candida auris isolates, the MIC range is 0.25~2mg/ml, with MIC50 and MIC90 both 1mg/ml, and a geometric mean MIC of 0.764mg/ml; for 2 echinocandin-resistant Candida auris isolates, MIC is 0.5mg/ml for both.
In animal studies, this drug is mainly used to evaluate therapeutic efficacy in various Candida infection disease models. Common modeling types include an invasive candidiasis model of Candida auris in neutropenic mice, a guinea pig cutaneous candidiasis infection model, and a preclinical rat vaginal candidiasis model, all used to verify in vivo therapeutic effects. In the mouse invasive candidiasis model, oral dosing was initiated with a 24h delay after infection; dosing at 20, 30, and 40mg/kg twice daily for 7 consecutive days showed that 30mg/kg and 40mg/kg were the core effective therapeutic doses, significantly improving 21-day survival, and dose-dependently reducing renal fungal burden. The 40mg/kg dose group reduced renal fungal burden by more than 2.5log10 CFU/g compared with pre-treatment. At the clinical application level, the drug has been approved by the FDA for the treatment of vulvovaginal candidiasis (VVC) and for reducing the incidence of recurrent VVC. It can stably maintain anti-Candida activity in the acidic physiological environment of pH 3.8~4.5 in the vagina of VVC patients, meeting the treatment needs of local vaginal infections. Meanwhile, treatment and prevention of invasive candidiasis are in phase Ⅱ/Ⅲ clinical trials; oral administration can achieve clinically attainable drug exposure in humans. Clinical cases have confirmed that oral administration can successfully treat Candida auris bloodstream infection, and it also shows potential clinical therapeutic value for invasive infections caused by echinocandin-resistant Candida.
References:
[1] Wiederhold NP, Najvar LK, Olivo M, Morris KN, Patterson HP, Catano G, Patterson TF. Ibrexafungerp Demonstrates In Vitro Activity against Fluconazole-Resistant Candida auris and In Vivo Efficacy with Delayed Initiation of Therapy in an Experimental Model of Invasive Candidiasis. Antimicrob Agents Chemother. 2021 May 18;65(6):e02694-20. doi: 10.1128/AAC.02694-20. PMID: 33753333; PMCID: PMC8315906.
[2] Sobel JD, Borroto-Esoda K, Azie N, Angulo D. In Vitro pH Activity of Ibrexafungerp against Fluconazole-Susceptible and -Resistant Candida Isolates from Women with Vulvovaginal Candidiasis. Antimicrob Agents Chemother. 2021 Jul 16;65(8):e0056221. doi: 10.1128/AAC.00562-21. Epub 2021 Jul 16. PMID: 34001513; PMCID: PMC8284466.
[3] Aldejohann AM, Menner C, Thielemann N, Martin R, Walther G, Kurzai O. In vitro activity of ibrexafungerp against clinically relevant echinocandin-resistant Candida strains. Antimicrob Agents Chemother. 2024 Feb 7;68(2):e0132423. doi: 10.1128/aac.01324-23. Epub 2024 Jan 11. PMID: 38206004; PMCID: PMC10848775.
| Storage | Store at -20°C |
| M.Wt | 730.03 |
| Cas No. | 1207753-03-4 |
| Formula | C44H67N5O4 |
| Synonyms | MK 3118; SCY-078 |
| Canonical SMILES | [H][C@]12CC[C@@]3([H])[C@@]4(C)COC[C@@]3(C[C@H]([C@@H]4OC[C@](C)(N)C(C)(C)C)N3N=CN=C3C3=CC=NC=C3)C1=CC[C@@]1(C)[C@H](C(O)=O)[C@](C)(CC[C@]21C)[C@H](C)C(C)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |