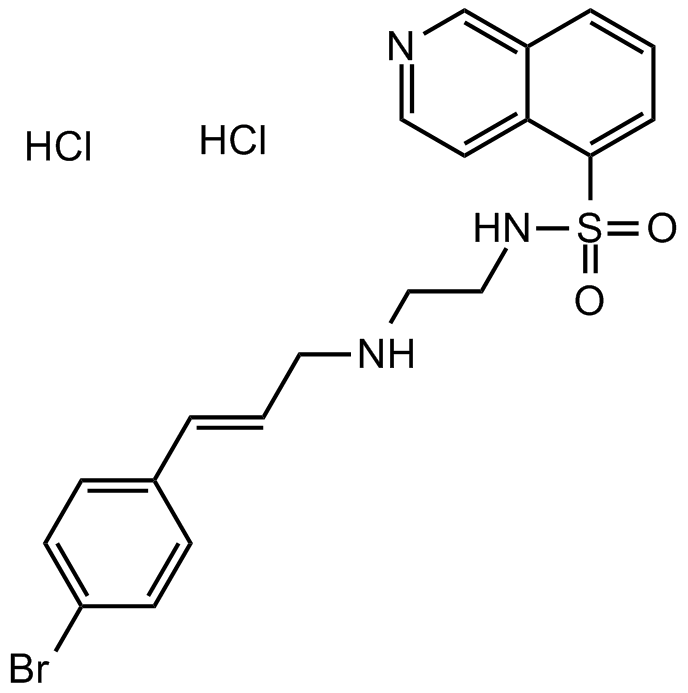
H 89 2HCl
H 89 2HCl is a potent PKA inhibitor. In a cell-free assay, the Ki of H 89 is 48 nM, 10-fold selective for PKA than PKG and 500-fold greater selectivity than PKC, MLCK, calmodulin kinase II and casein kinase I/II [1].
In vitro:In PC12D cells, pretreatment with H-89 dose-dependently inhibited the forskolin-induced protein phosphorylation, with no influence in intracellular cyclic AMP levels. In PC12D cells, H-89 significantly inhibited the forskolin-induced neurite outgrowth. In PC12D cells, pretreatment with H-89 (30 μM) strikingly inhibited cAMP-dependent histone IIb phosphorylation activity in cell lysates while showed no effects on other protein phosphorylation activity such as cGMP-dependent histone IIb phosphorylation activity [1]. H 89 was a potent and selective PKA inhibitor with Ki of 48 nM in a cell-free assay [2]. H89 also inhibited S6K1, MSK1, PKA, ROCKII, PKBα and MAPKAP-K1b kinases with IC50 of 80, 120, 135, 270, 2600 and 2800 nM, respectively [2]. In the hypotonic medium, 50 μM H89, a concentration commonly used to inhibit PKA, prevented the redistribution response. In normal medium, H89 (50 Μm) induced the redistribution of ERGIC 53 to the ER by 20 min [3].
References:
[1]. Chijiwa T, Mishima A, Hagiwara M, et al. Inhibition of forskolin-induced neurite outgrowth and protein phosphorylation by a newly synthesized selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-(p-bromocinnamylamino) ethyl]-5-isoquinolinesulfonamide (H-89), of PC12D pheochromocytoma cells[J]. Journal of Biological Chemistry, 1990, 265(9): 5267-5272.
[2]. Lochner A, Moolman J A. The many faces of H89: a review[J]. Cardiovascular drug reviews, 2006, 24(3‐4): 261-274.
[3]. Lee T H, Linstedt A D. Potential role for protein kinases in regulation of bidirectional endoplasmic reticulum-to-Golgi transport revealed by protein kinase inhibitor H89[J]. Molecular biology of the cell, 2000, 11(8): 2577-2590
.
- 1. Xinyue Liao, Zhaoke Luo, et al. "Trigeminal nerve root compression induced neuroinflammatory response promotes mechanical allodynia through the CGRP/SP-Piezo2 axis via Ca 2+ signaling." Cell Mol Biol Lett. 2025 Dec 3;31(1):3. PMID: 41340091
- 2. Dandan Lv, An Liu, et al. "Neuroligin 1 Regulates Autistic‐Like Repetitive Behavior through Modulating the Activity of Striatal D2 Receptor‐Expressing Medium Spiny Neurons." Adv Sci (Weinh). 2025 Feb;12(5):e2410728. PMID: 39661696
- 3. Chengjia You, Fangyuan Shen, et al. "O-GlcNAcylation mediates Wnt-stimulated bone formation by rewiring aerobic glycolysis." EMBO Rep. 2024 Sep 10. PMID: 39256595
- 4. Xiaodong Kong, Yuting Yang, et al. "GLP‑1 Receptor Agonist Inhibited the Activation of RIPK1 for Alleviation the Neuronal Death and Neuroinflammation in APP/PS1 Mice." International Journal of Peptide Research and Therapeutics. 31 March 2021
- 5. Liu DT, Hong WS, et al. "Upregulation of adamts9 by gonadotropin in preovulatory follicles of zebrafish." Mol Cell Endocrinol. 2019 Oct 2;499:110608 PMID: 31586455
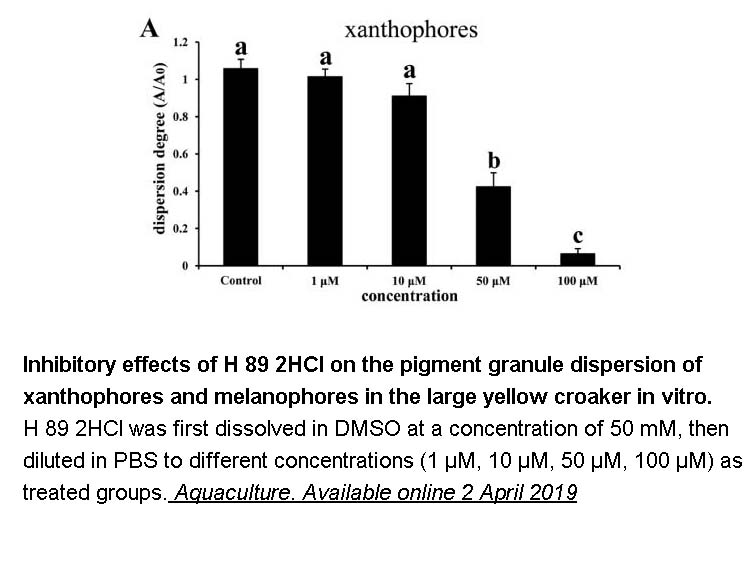
- 6. JianHanaWan, ShuHongac, et al. "The regulation of melanocyte-stimulating hormone on the pigment granule dispersion in the xanthophores and melanophores of the large yellow croaker (Larimichthys crocea)." Aquaculture. Available online 2 April 2019
- 7. MXinwei Feng, Junfeng Lu, et al. "Mycobacterium smegmatis Induces Neurite Outgrowth and Differentiation in an Autophagy-Independent Manner in PC12 and C17.2 Cells." Front. Cell. Infect. Microbiol., 19 June 2018
- 8. Matos, Steven Cordeiro, et al. "Peripheral Neuropathy Induces HCN Channel Dysfunction in Pyramidal Neurons of the Medial Prefrontal Cortex." The Journal of Neuroscience 35.38 (2015): 13244-13256 PMID: 26400952
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 519.28 |
| Cas No. | 130964-39-5 |
| Formula | C20H20BrN3O2S·2HCl |
| Solubility | ≥51.9 mg/mL in DMSO; insoluble in H2O; insoluble in EtOH |
| Chemical Name | (E)-N-(2-((3-(4-bromophenyl)allyl)amino)ethyl)isoquinoline-5-sulfonamide dihydrochloride |
| Canonical SMILES | O=S(C1=CC=CC2=C1C=CN=C2)(NCCNC/C=C/C3=CC=C(Br)C=C3)=O.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
PKA enzyme activity |
cAMP-dependent protein kinase activity is assayed in a reaction mixture containing, in a final volume of 0.2 mL, 50 mM Tris-HC1 (pH 7.0), 10 mM magnesium acetate, 2 mM EGTA, 1 μM cAMP or absence of cAMP, 3.3-20 μM [γ-32P]ATP (4 × 105 cpm), 0.5 μg of the enzyme, 100 μg of histone H2B, and each compound, as indicated. |
| Cell experiment [1]: | |
|
Cell lines |
PC12D cells |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
30 μM for 1h |
|
Applications |
H-89 (30 μM) significantly inhibited cAMP-dependent histone IIh phosphorylation activity and suppressed Forskolin-induced neurite outgrowth in PC12D cells. |
| Animal experiment [2]: | |
|
Animal models |
Rats model |
|
Dosage form |
20 or 200 mg/kg; s.c. twice daily for 2 days; |
|
Applications |
H89 caused distinct modifications of protein phosphorylation, with the most robust changes in phosphorylation were heterogeneous nuclear ribonucleoprotein (hnRNP), fructose-1,6-biphosphatase, NSFL1 cofactor p47, all which had potentially regulatory connections to cAMP/PKA. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: 1Chijiwa, T., Mishima, A., Hagiwara, M., Sano, M., Hayashi, K., Inoue, T., Naito, K., Toshioka, T. and Hidaka, H. (1990) Inhibition of forskolin-induced neurite outgrowth and protein phosphorylation by a newly synthesized selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-(p-bromocinnamylamino)ethyl]-5-isoquinolinesulfonamide (H-89), of PC12D pheochromocytoma cells. J Biol Chem. 265, 5267-5272 2Davis, M. A., Hinerfeld, D., Joseph, S., Hui, Y. H., Huang, N. H., Leszyk, J., Rutherford-Bethard, J. and Tam, S. W. (2006) Proteomic analysis of rat liver phosphoproteins after treatment with protein kinase inhibitor H89 (N-(2-[p-bromocinnamylamino-]ethyl)-5-isoquinolinesulfonamide). J Pharmacol Exp Ther. 318, 589-595 |
|
| Description | H 89 2HCl is a potent inhibitor of PKA with a Ki value of 48 nM. | |||||
| Targets | PKA | S6K1 | ||||
| IC50 | 48 nM(Ki) | 80 nM | ||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
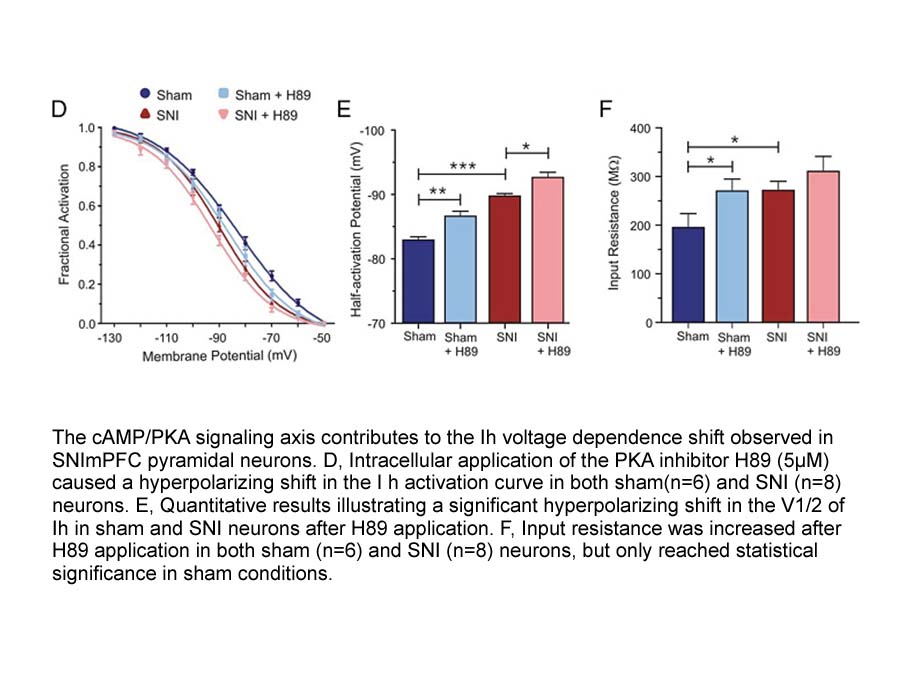
Related Biological Data
Related Biological Data