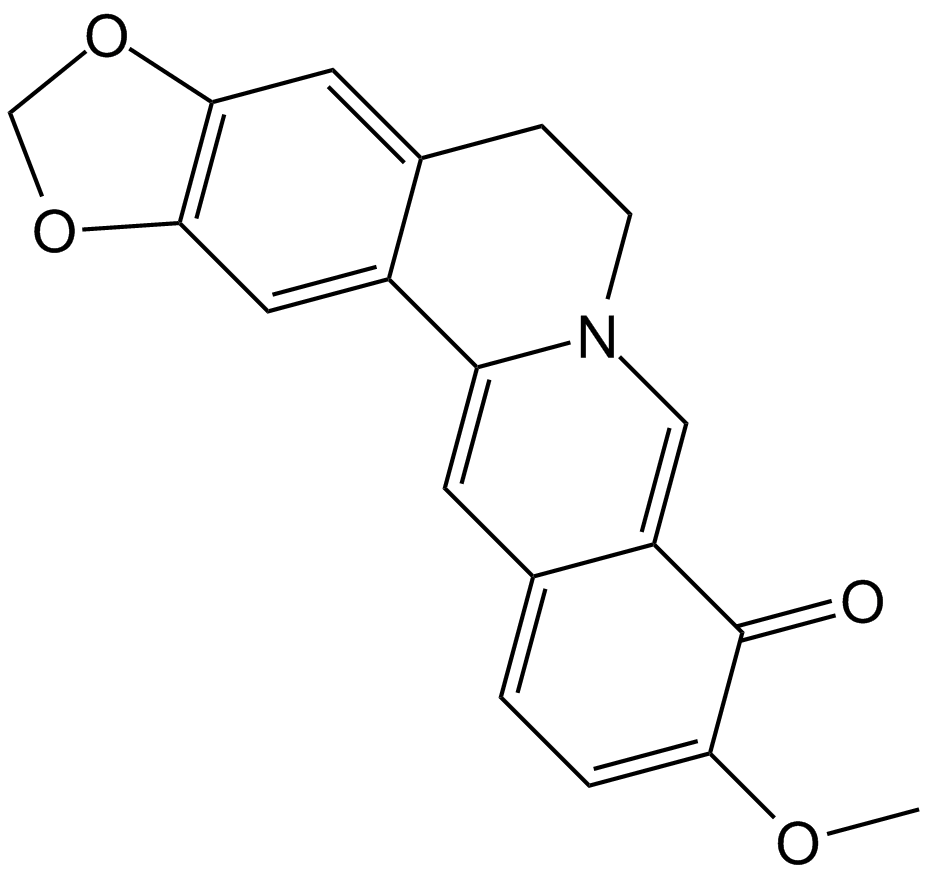
Berberrubine chloride
Berberrubine chloride (CAS No. 15401-69-1) is the hydrochloride salt form of Berberrubine (CAS No. 17388-19-1). Berberrubine is a natural isoquinoline alkaloid derived from traditional Chinese medicines such as Coptis chinensis (berberine, Berberine (CAS No. 2086-83-1) major metabolite), with core biological functions including antibacterial, anti-inflammatory, hypoglycemic, anti-hyperuricemia, anti-thrombosis, and inhibition of tumor proliferation such as colorectal cancer and non-small cell lung cancer (NSCLC); it can also activate glutathione S-transferase Mu2 (GSTM2) to exert tumor-suppressive effects; its targets and mechanisms are diverse: inhibition of IMP dehydrogenase 2 (IMPDH2, competitive binding, with selectivity for IMPDH2 more than 15-fold higher than IMPDH1), thioredoxin reductase (TrxR, specifically targeting the Sec498 residue), vitamin K epoxide reductase (VKOR) and γ-glutamyl carboxylase (GGCX), activation of GSTM2 (via activation by the SP1 transcription factor and DNA demethylation), inhibition of NF-κB nuclear translocation, the JAK2/STAT3 signaling pathway, and urate transporters URAT1/GLUT9, upregulation of urate excretion transporters OAT1/3/ABCG2, and it can also inhibit topoisomerase II-mediated DNA cleavage; key activity data: IMPDH2 inhibition IC₅₀ 2.37 μM, TrxR inhibition IC₅₀ 5.0 μM, MIC values against fungi such as Candida albicans of 1-16 μg/mL (the 13-(4 - isopropylbenzyl) derivative is the most potent); commonly used animal models include the AOM/DSS-induced colorectal cancer model, the PO/HX-induced hyperuricemia model, the carrageenan-induced thrombosis model, the DSS-induced ulcerative colitis model, and the NSCLC xenograft model.
Common applications and concentrations: in cell culture, SW620/LS174T colorectal cancer cells 10-80 μM, A549 NSCLC cells 20-50 μM, ARPE-19 retinal pigment epithelial cells 0.2-25 μM, BFTC 905 bladder cancer cells 50 μM; in animal studies, oral dosing in the colorectal cancer model 25-50 mg/kg/d, oral dosing in the hyperuricemia model 6.25-25 mg/kg/d, oral dosing in the thrombosis model 50-100 mg/kg/d, oral dosing in the colitis model 100-200 mg/kg/d; no direct clinical application concentrations; effective therapeutic concentrations: at the cellular level, 10-80 μM can inhibit tumor cell proliferation, and 20 μM combined with cisplatin can enhance NSCLC chemosensitivity; at the animal level, 25-50 mg/kg/d can significantly inhibit colorectal tumor growth, 25 mg/kg/d can reduce serum uric acid levels in hyperuricemic mice by more than 75%, and 100 mg/kg orally can inhibit thrombosis without increasing bleeding risk.
References:
[1] Park KD, Lee JH, Kim SH, Kang TH, Moon JS, Kim SU. Synthesis of 13-(substituted benzyl) berberine and berberrubine derivatives as antifungal agents. Bioorg Med Chem Lett. 2006 Aug 1;16(15):3913-6. doi: 10.1016/j.bmcl.2006.05.033. Epub 2006 May 30. PMID: 16730982.
[2] Cui HS, Hayasaka S, Zhang XY, Hayasaka Y, Chi ZL, Zheng LS. Effect of berberrubine on interleukin-8 and monocyte chemotactic protein-1 expression in human retinal pigment epithelial cell line. Life Sci. 2006 Aug 1;79(10):949-56. doi: 10.1016/j.lfs.2006.05.004. Epub 2006 May 12. PMID: 16797033.
[3] Li R, Wu J, He Y, Hai L, Wu Y. Synthesis and in vitro evaluation of 12-(substituted aminomethyl) berberrubine derivatives as anti-diabetics. Bioorg Med Chem Lett. 2014 Apr 1;24(7):1762-5. doi: 10.1016/j.bmcl.2014.02.032. Epub 2014 Feb 21. PMID: 24613165.
[4] Lin G, Yu Q, Xu L, Huang Z, Mai L, Jiang L, Su Z, Xie J, Li Y, Liu Y, Lin Z, Chen J. Berberrubine attenuates potassium oxonate- and hypoxanthine-induced hyperuricemia by regulating urate transporters and JAK2/STAT3 signaling pathway. Eur J Pharmacol. 2021 Dec 5;912:174592. doi: 10.1016/j.ejphar.2021.174592. Epub 2021 Oct 23. PMID: 34699754.
[5] Yang S, Cao S, Li C, Zhang J, Liu C, Qiu F, Kang N. Berberrubine, a Main Metabolite of Berberine, Alleviates Non-Alcoholic Fatty Liver Disease via Modulating Glucose and Lipid Metabolism and Restoring Gut Microbiota. Front Pharmacol. 2022 Jul 8;13:913378. doi: 10.3389/fphar.2022.913378. PMID: 35873595; PMCID: PMC9304582.
[6] Shen CH, Wu JY, Wang SC, Wang CH, Hong CT, Liu PY, Wu SR, Liu YW. The suppressive role of phytochemical-induced glutathione S-transferase Mu 2 in human urothelial carcinoma cells. Biomed Pharmacother. 2022 Jul;151:113102. doi: 10.1016/j.biopha.2022.113102. Epub 2022 May 17. PMID: 35594716.
[7] Wang C, Yuan Z, Xie J, Lei Y, Li Y, Huang J, Kong W, Jiang J. Integrated metabolomics and molecular docking reveal berberrubine inhibits thrombosis by regulating the vitamin K catalytic cycle in mice. Eur J Pharmacol. 2023 Jan 5;938:175436. doi: 10.1016/j.ejphar.2022.175436. Epub 2022 Dec 5. PMID: 36481237.
[8] He X, Cui J, Ma H, Abuduaini N, Huang Y, Tang L, Wang W, Zhang Y, Wang Y, Lu W, Feng B, Huang J. Berberrubine is a novel and selective IMPDH2 inhibitor that impairs the growth of colorectal cancer. Biochem Pharmacol. 2023 Dec;218:115868. doi: 10.1016/j.bcp.2023.115868. Epub 2023 Oct 21. PMID: 37871880.
[9] Rao J, Wang T, Yu L, Wang K, Qiu F. Inactivation of CYP2D6 by Berberrubine and the Chemical Mechanism. Biochemistry. 2024 Dec 3;63(23):3078-3089. doi: 10.1021/acs.biochem.4c00450. Epub 2024 Nov 21. PMID: 39569501.
[10] Chu Y, Nie Q, Zhou X, Yang J, Fang J, Zhang J. Berberrubine as a novel TrxR inhibitor enhances cisplatin sensitivity in the treatment of non-small cell lung cancer. Bioorg Chem. 2025 May;158:108329. doi: 10.1016/j.bioorg.2025.108329. Epub 2025 Mar 3. PMID: 40056602.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 357.79 |
| Cas No. | 15401-69-1 |
| Formula | C19H16ClNO4 |
| Synonyms | Beroline; 9-Berberoline |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥6.42 mg/mL in DMSO with gentle warming and ultrasonic |
| Chemical Name | 9-hydroxy-10-methoxy-5,6-dihydro-[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinolin-7-ium chloride |
| Canonical SMILES | OC1=C(OC)C=CC(C1=C2)=CC3=[N+]2CCC4=CC(OCO5)=C5C=C43.[Cl-] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure