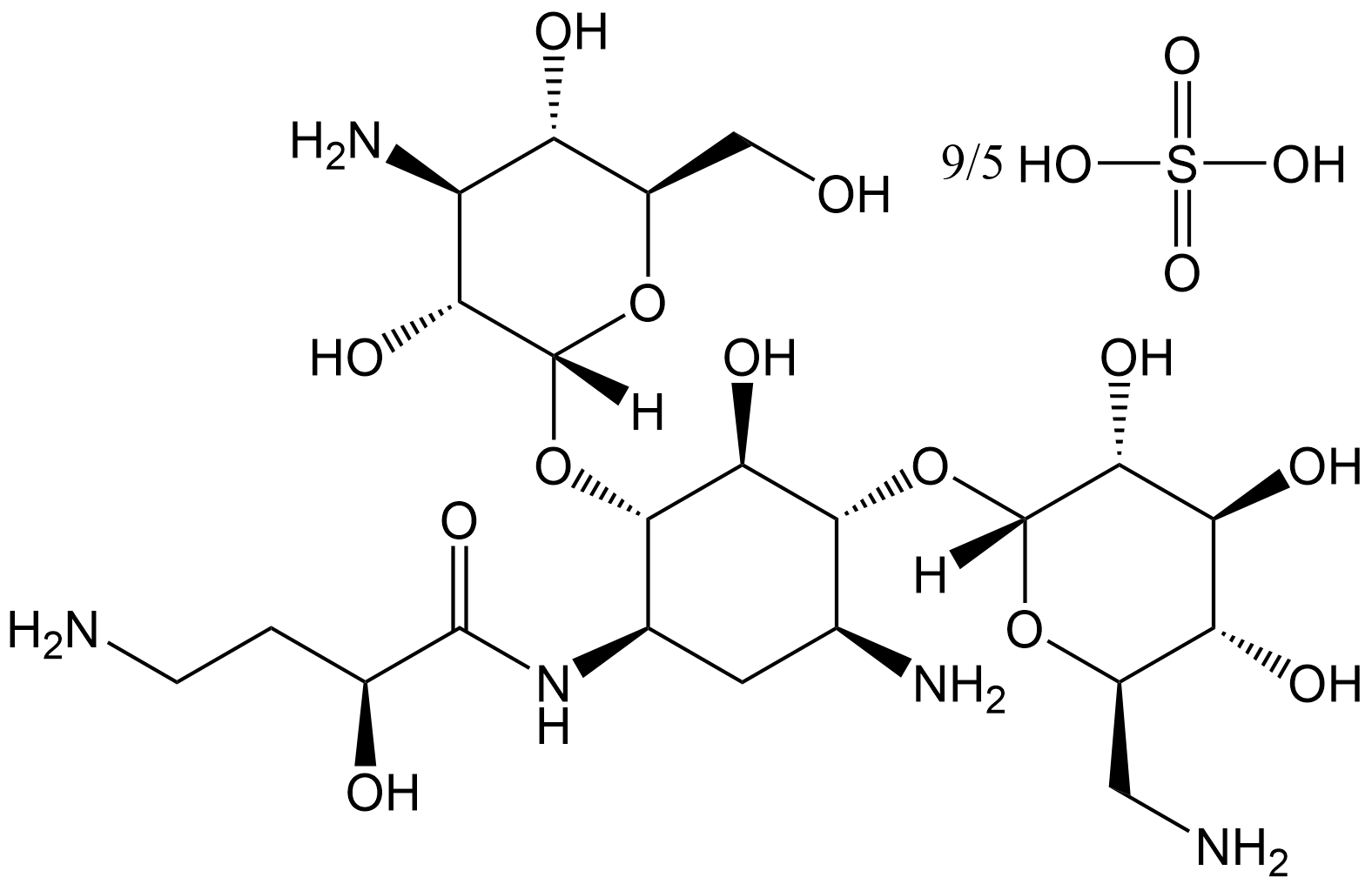
Amikacin Sulfate
Amikacin Sulfate (CAS No. 149022-22-0) is the sulfate form of amikacin (Amikacin, CAS No.: Amikacin, Cat. No.: B3431). Amikacin is an aminoglycoside antibiotic whose core biological function is to exert dose-dependent bactericidal activity against Mycobacterium avium (Mycobacterium avium, MAC) and Staphylococcus aureus (Staphylococcus aureus). FITC fluorescent conjugation does not substantially alter its antibacterial activity. The minimum inhibitory concentration (MIC) against M. avium is 1 mg/ml. In vitro, at 64 mg/L, free amikacin can reduce M. avium colony-forming units (CFU) from 300 in the control group to 1, and reduce S. aureus CFU from 124 in the control group to 2.5. At the same dose, the bactericidal effect of amikacin - FITC on both bacteria shows no significant difference from the free form.
In cell culture systems, the commonly used concentration range is 25~100 mg/L. Within this range, amikacin - FITC can be efficiently taken up by mouse monocyte-macrophage RAW 264.7-derived dendritic cells (DCs) via passive transmembrane diffusion. After 24 h exposure to 50 mg/L, the intracellular drug concentration can reach 15~29 mg/ml, far exceeding its MIC against M. avium. At 50 and 100 mg/L, it does not affect DC viability, nor does it upregulate secretion of the inflammatory marker MCP-1 or gene expression of CCR-2, showing no significant cytotoxicity or pro-inflammatory effects.
In animal experiments, its most common application is a disseminated non-tuberculous mycobacterial (NTM) infection model established by intranasal inoculation of M. avium in C57BL/6 mice (typical granulomas form in the lungs, spleen, and liver after 6 months of infection), used to validate DC-mediated targeted drug delivery and in vivo therapeutic efficacy. The median lethal dose (LD50) of commercially available amikacin sulfate by intravenous injection in mice is 181 mg/kg, corresponding to a serum level of approximately 1~3 mg/ml. At 24 h after tail-vein injection of DCs loaded with amikacin - FITC, the drug concentration in bronchoalveolar lavage fluid (BALF) of infected mice reaches 0.5 mg/ml. After accounting for the dilution factor of lavage fluid, the drug concentration in the lung lining fluid reaches several times the MIC, and drug enrichment is detected only in granulomas of infected tissues, with none detected in serum, achieving targeted delivery to the infection site and maintenance of an effective therapeutic concentration.
Clinically, amikacin is a core drug for treating infections such as non-tuberculous mycobacteria. Conventional use is limited by the need for high systemic doses and the propensity to cause ototoxicity and nephrotoxicity; related studies focus on cell carrier-mediated targeted delivery to improve its therapeutic index.
References:
[1] Montes-Worboys A, Brown S, Regev D, Bellew BF, Mohammed KA, Faruqi I, Sharma P, Moudgil B, Antony VB. Targeted delivery of amikacin into granuloma. Am J Respir Crit Care Med. 2010 Dec 15;182(12):1546-53. doi: 10.1164/rccm.200912-1888OC. Epub 2010 Aug 6. PMID: 20693382; PMCID: PMC3029940.
[2] Jain G, Jaimes EA. Nicotine signaling and progression of chronic kidney disease in smokers. Biochem Pharmacol. 2013 Oct 15;86(8):1215-23. doi: 10.1016/j.bcp.2013.07.014. Epub 2013 Jul 26. PMID: 23892062; PMCID: PMC3838879.
[3] Forsberg KJ, Bhatt IV, Schmidtke DT, Javanmardi K, Dillard KE, Stoddard BL, Finkelstein IJ, Kaiser BK, Malik HS. Functional metagenomics-guided discovery of potent Cas9 inhibitors in the human microbiome. Elife. 2019 Sep 10;8:e46540. doi: 10.7554/eLife.46540. PMID: 31502535; PMCID: PMC6739867.
| Storage | -20℃, sealed storage, away from moisture and light |
| M.Wt | 762.14 |
| Cas No. | 149022-22-0 |
| Formula | C22H43N5O13•9/5H2O4S |
| Synonyms | BAY 41-6551 sulfate |
| Canonical SMILES | OS(O)(=O)=O.[H][C@]1(O[C@H]2[C@@H](C[C@H](N)[C@@H](O[C@@]3([H])O[C@H](CN)[C@@H](O)[C@H](O)[C@H]3O)[C@@H]2O)NC(=O)[C@@H](O)CCN)O[C@H](CO)[C@@H](O)[C@H](N)[C@H]1O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |