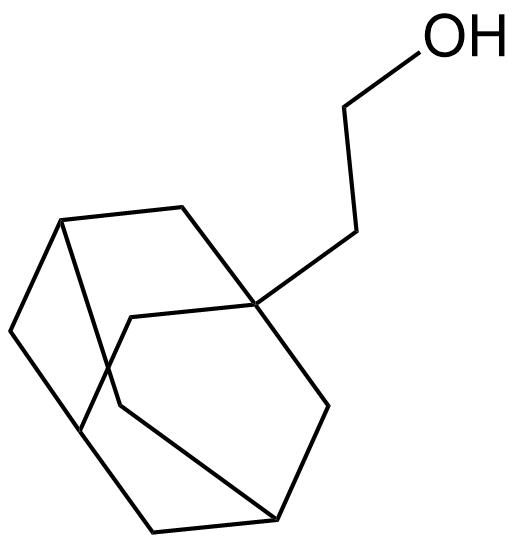
1-Adamantaneethanol
The core biological function of 1-Adamantaneethanol (CAS No. 6240-11-5) is as a key intermediate for the synthesis of antibacterial and immunomodulatory adamantane derivatives. Its derivatives can enhance antigen peptide binding and exchange (HLA-DM-independent) by acting on the P1 pocket of MHC II molecules (selective for MHC II alleles with a large P1 pocket such as DR1), and also exhibit antibacterial activity against Gram-positive bacteria such as Staphylococcus aureus, Bacillus subtilis, and Bacillus cereus; the derived N-substituted phthalimide compounds have MIC values of 8-100 μg/mL (15 μg/mL against Bacillus cereus; 20 μg/mL against Staphylococcus aureus NTCC 4163). It is mainly used to assist immunomodulation (co-injection with a targeting peptide can enhance T-cell priming in mice, as a model therapeutic adjuvant). Common concentrations: in cell culture, >10⁻⁵ M can significantly enhance MHC II-mediated peptide exchange; in animal experiments, it is used for immune enhancement by co-injection with peptides. In terms of effective concentration, the MIC values of antibacterial derivatives are the effective concentrations for inhibiting bacterial growth, and in immunomodulation, concentrations of 10⁻⁵ M and above can effectively promote peptide binding to MHC II.
References:
[1] Orzeszko A, Kamińska B, Orzeszko G, Starościak BJ. Synthesis and antimicrobial activity of new adamantane derivatives II. Farmaco. 2000 Sep-Oct;55(9-10):619-23. doi: 10.1016/s0014-827x(00)00075-6. PMID: 11152243.
[2] Call MJ. Small molecule modulators of MHC class II antigen presentation: mechanistic insights and implications for therapeutic application. Mol Immunol. 2011 Sep;48(15-16):1735-43. doi: 10.1016/j.molimm.2011.05.022. Epub 2011 Jun 14. PMID: 21676462.
[3] de Souza TG, Granado R, Benaim G, de Souza W, Benchimol M. Effects of SQ109 on Trichomonas vaginalis. Exp Parasitol. 2023 Jul;250:108549. doi: 10.1016/j.exppara.2023.108549. Epub 2023 May 16. PMID: 37196704.
| Storage | Store at -20°C |
| M.Wt | 180.29 |
| Cas No. | 6240-11-5 |
| Formula | C12H20O |
| Synonyms | Tricyclo[3.3.1.13,7]decane-1-ethanol; 2-(adamant-1-yl)ethanol; 1-(2-Hydroxyethyl)-adamantan |
| Solubility | ≥28.2 mg/mL in DMSO; ≥15.05 mg/mL in EtOH; insoluble in H2O |
| Chemical Name | 2-(adamantan-1-yl)ethan-1-ol |
| Canonical SMILES | OCCC12CC3CC(CC(C3)C1)C2 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure