PF-04971729
Ertugliflozin (CAS No. 1210344-57-2) is a highly selective sodium-glucose co-transporter 2 (SGLT2) inhibitor (with >2000-fold higher selectivity for SGLT2 than SGLT1). Its core biological functions include lowering blood glucose in patients with type 2 diabetes (T2D), reducing body weight, providing cardiovascular protection (reducing the risk of hospitalization for heart failure), and renal protection. It also shows anti-inflammatory and mucosal repair activity in ulcerative colitis (UC) models. Its mechanism is specific inhibition of SGLT2-mediated renal glucose reabsorption. It is commonly used for disease model treatment; common models include acetic acid-induced UC rat models, cardiac pressure overload models, and T2D-related models (e.g., high-fat diet-induced diabetes models), which are used to evaluate anti-inflammatory mucosal repair, cardiovascular protection, and glucose-lowering/weight-loss effects, respectively.
Common applications and concentrations: In animal studies, oral dosing in UC models is 1, 5, 10 mg/kg/day (the efficacy at 10 mg/kg is comparable to sulfasalazine 100 mg/kg); dosing concentrations for cardiac models have not been clearly defined. Clinically, the commonly used dose in T2D patients is 5 mg or 15 mg once daily orally, as monotherapy or add-on therapy; it is also applicable to T2D patients with concomitant atherosclerotic cardiovascular disease. Effective therapeutic concentrations: Clinically, 5 mg and 15 mg once daily can significantly reduce glycated hemoglobin and reduce body weight (in the VERTIS FACTORIAL study, the 15 mg group lost 3.7 kg, 4.2% of baseline body weight); the 15 mg dose is superior for weight reduction and can reduce the risk of hospitalization for heart failure (hazard ratio 0.70, 95% CI 0.54-0.90). In animal studies, oral 10 mg/kg can inhibit the NF-κB signaling pathway in UC models, downregulate miR-155 expression, promote M2 macrophage polarization, repair the intestinal mucosal barrier, and improve myocardial remodeling in cardiac pressure overload models. In terms of safety, it may increase fracture risk (RR 2.47, not statistically significant), and there are no clear concentration-associated data related to serious adverse reactions.
References:
[1] Cannon CP, Pratley R, Dagogo-Jack S, Mancuso J, Huyck S, Masiukiewicz U, Charbonnel B, Frederich R, Gallo S, Cosentino F, Shih WJ, Gantz I, Terra SG, Cherney DZI, McGuire DK; VERTIS CV Investigators. Cardiovascular Outcomes with Ertugliflozin in Type 2 Diabetes. N Engl J Med. 2020 Oct 8;383(15):1425-1435. doi: 10.1056/NEJMoa2004967. Epub 2020 Sep 23. PMID: 32966714.
[2] Lazzaroni E, Ben Nasr M, Loretelli C, Pastore I, Plebani L, Lunati ME, Vallone L, Bolla AM, Rossi A, Montefusco L, Ippolito E, Berra C, D'Addio F, Zuccotti GV, Fiorina P. Anti-diabetic drugs and weight loss in patients with type 2 diabetes. Pharmacol Res. 2021 Sep;171:105782. doi: 10.1016/j.phrs.2021.105782. Epub 2021 Jul 22. PMID: 34302978.
[3] Zhang YS, Zheng YD, Yuan Y, Chen SC, Xie BC. Effects of Anti-Diabetic Drugs on Fracture Risk: A Systematic Review and Network Meta-Analysis. Front Endocrinol (Lausanne). 2021 Oct 14;12:735824. doi: 10.3389/fendo.2021.735824. PMID: 34721294; PMCID: PMC8553257.
[4] Nikolaou PE, Mylonas N, Makridakis M, Makrecka-Kuka M, Iliou A, Zerikiotis S, Efentakis P, Kampoukos S, Kostomitsopoulos N, Vilskersts R, Ikonomidis I, Lambadiari V, Zuurbier CJ, Latosinska A, Vlahou A, Dimitriadis G, Iliodromitis EK, Andreadou I. Cardioprotection by selective SGLT-2 inhibitors in a non-diabetic mouse model of myocardial ischemia/reperfusion injury: a class or a drug effect? Basic Res Cardiol. 2022 May 17;117(1):27. doi: 10.1007/s00395-022-00934-7. PMID: 35581445.
[5] Pang B, Zhang LL, Li B, Sun FX, Wang ZD. The sodium glucose co-transporter 2 inhibitor ertugliflozin for Alzheimer's disease: Inhibition of brain insulin signaling disruption-induced tau hyperphosphorylation. Physiol Behav. 2023 May 1;263:114134. doi: 10.1016/j.physbeh.2023.114134. Epub 2023 Feb 19. PMID: 36809844.
[6] Kongmalai T, Hadnorntun P, Leelahavarong P, Kongmalai P, Srinonprasert V, Chirakarnjanakorn S, Chaikledkaew U, McKay G, Attia J, Thakkinstian A. Comparative cardiovascular benefits of individual SGLT2 inhibitors in type 2 diabetes and heart failure: a systematic review and network meta-analysis of randomized controlled trials. Front Endocrinol (Lausanne). 2023 Dec 20;14:1216160. doi: 10.3389/fendo.2023.1216160. PMID: 38179304; PMCID: PMC10765518.
[7] Fouad MR, Rabie MA, Zaki HF, Salama RM. Ertugliflozin ameliorates experimentally-induced colitis in rats by regulating the interplay between M1/M2 macrophage polarization, tight junction proteins, and MicroRNA 155 expression. Int Immunopharmacol. 2025 Nov 14;165:115505. doi: 10.1016/j.intimp.2025.115505. Epub 2025 Sep 6. PMID: 40915189.
| Storage | Store at -20°C |
| M.Wt | 436.88 |
| Cas No. | 1210344-57-2 |
| Formula | C22H25ClO7 |
| Synonyms | Ertugliflozin;PF04971729;PF 04971729 |
| Solubility | ≥50.8 mg/mL in DMSO; ≥51.5 mg/mL in EtOH; insoluble in H2O |
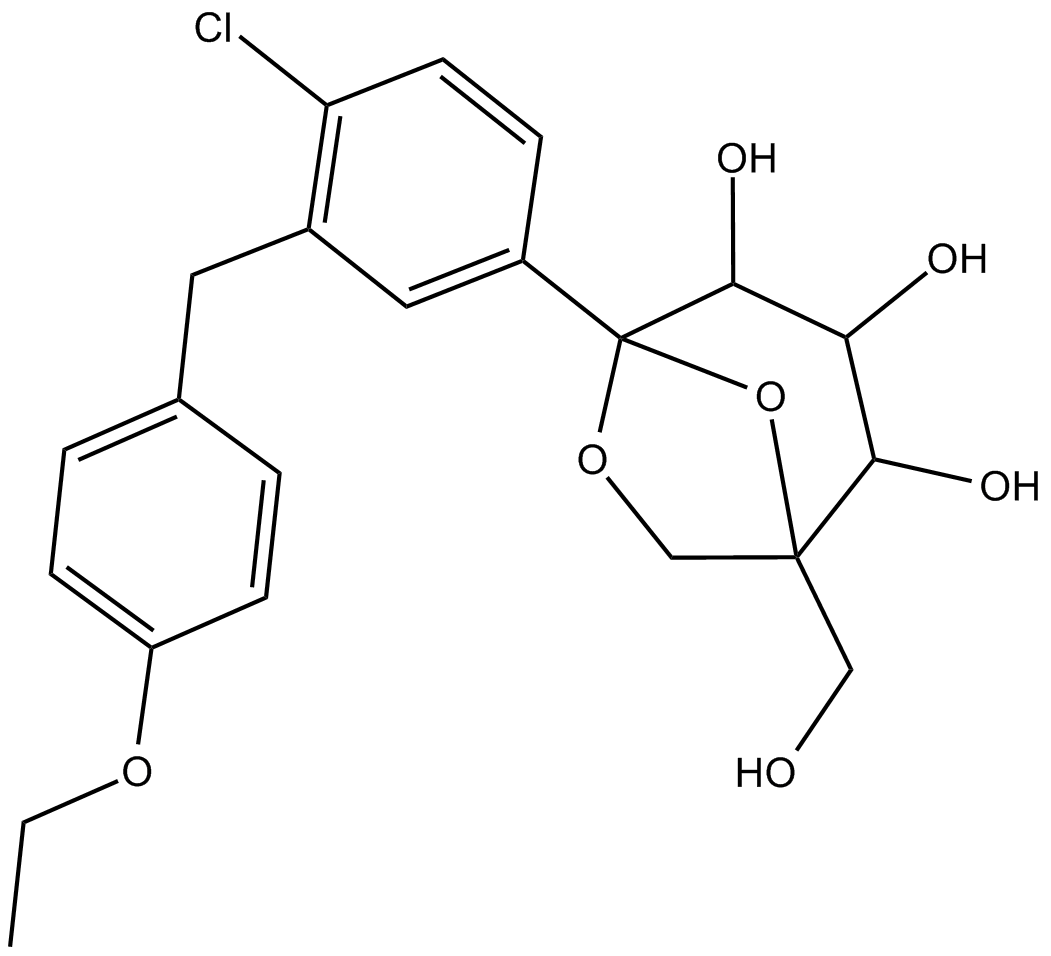
| Chemical Name | (1S,2S,3S,4R,5S)-5-(4-chloro-3-(4-ethoxybenzyl)phenyl)-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol |
| Canonical SMILES | O[C@H]1[C@@]2(O[C@@](CO)(CO2)[C@@H](O)[C@@H]1O)C3=CC(CC4=CC=C(OCC)C=C4)=C(Cl)C=C3 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure