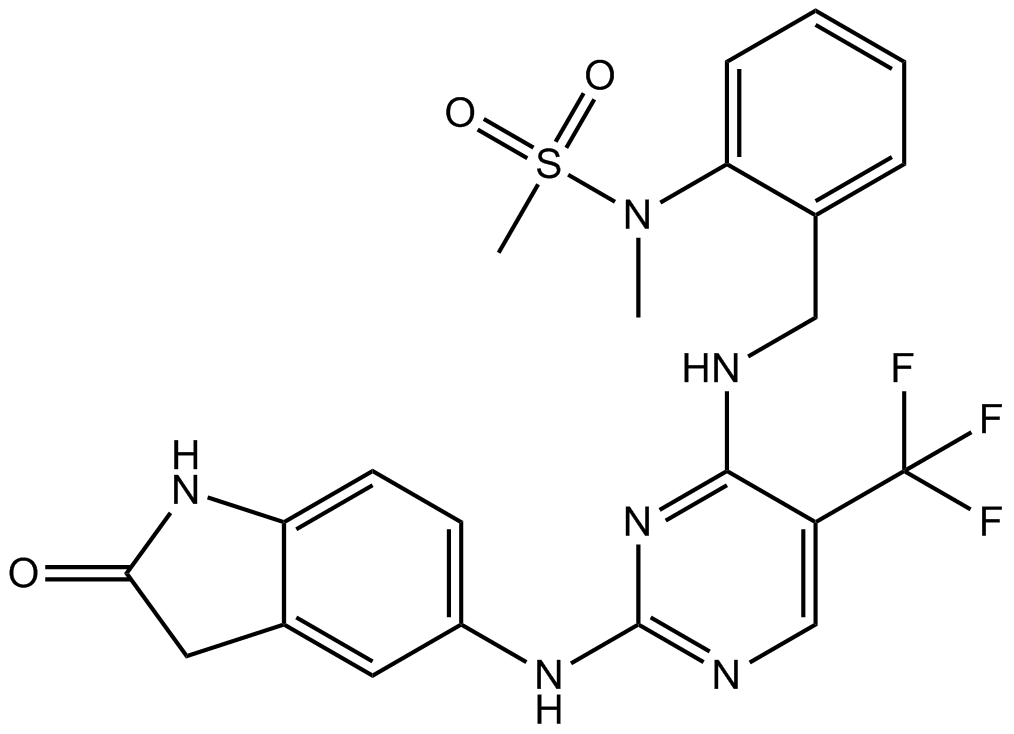
Fenretinide
Fenretinide(4HPR) is an inhibitor of Focal adhesion kinase (FAK) [1].
Fenretinideis a vitamin A analogue, it has been shown toinhibit the growth of many tumor cells, including small-cell lung cancer, malignant hemopoietic cells, and breast cancer cells. Fenretinide may also protectwomen against the development of ovarian cancer. The effect of Fenretinide on several gynecologic cancer cell lines shows the IC50 values of Fenretinide are only 0.3 and 0.4μM in two ovarian cancer cell lines(222and UCI 101) and are from 1 to 10μM in other ovarian cancer cell lines and cervical, endometrial cancer cell lines [2].
Fenretinide has also been shown to induce apoptosis inhuman prostate carcinoma cells (HPC).The IC50s of Fenretinide in LNCaP, DU145, and PC-3 are 0.9±0.16μM, 4.4±0.45μM and 3.0±1.0μM,respectively.Fenretinide induces this apoptosis through increasingROS and increasing enzymatic labeling of DNA breaks and formation of a DNA ladder. It is also reported that Fenretinide can impair prostate cancer cell migration and invasion by interfering with FAK/AKT/GSK3β pathway and β-catenin stability [1, 3].
References:
[1] Roberto Benelli, Stefano Monteghirfo, Roberta Venè, Francesca Tosettiand Nicoletta Ferrari.The chemopreventive retinoid 4HPR impairs prostate cancer cell migration and invasion by interfering with FAK/AKT/GSK3β pathway andβ-catenin stability. Molecular Cancer.2010, 9:142-154.
[2] Anita L. Sabichi, Denver T. Hendricks, Mary A. Bober, Michael J. Birrer. Retinoic acid receptorβexpression and growthinhibition of gynecologic cancer cells by thesynthetic retinoidn-(4-hydroxyphenyl) retinamide. Journal of the National Cancer Institute. 1998, 90(8): 597-605.
[3] Shi-Yong Sun, Ping Yue, and Reuben Lotan. Induction of apoptosis by n-(4-hydroxyphenyl)retinamide andits association with reactive oxygen species, nuclearretinoic acid receptors, and apoptosis-related genes in human prostate carcinoma cells.Molecular Pharmacology. 1999, 55:403–410.
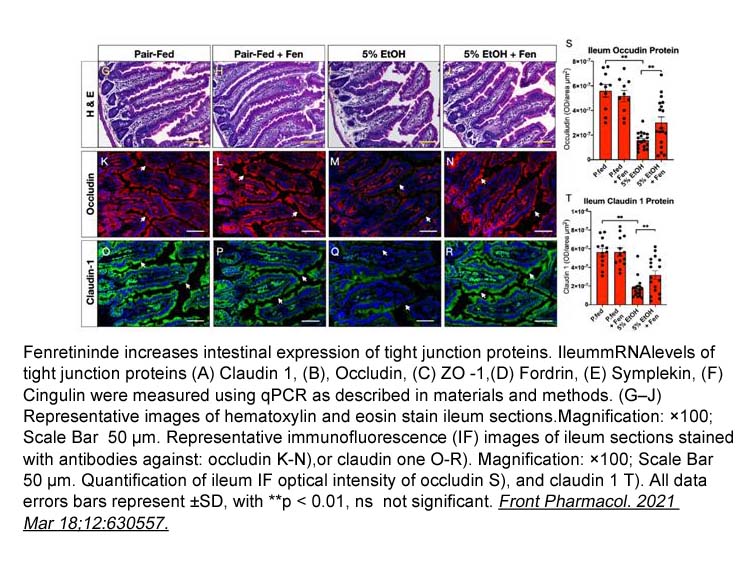
- 1. Xiao-Han Tang, Marta Melis, et al. "Fenretinide Improves Intestinal Barrier Function and Mitigates Alcohol Liver Disease." Front Pharmacol. 2021 Mar 18;12:630557. PMID:33815111
- 2. Britt EL, Raman S, et al. "Combination of fenretinide and ABT-263 induces apoptosis through NOXA for head and neck squamous cell carcinoma treatment." PLoS One. 2019 Jul 5;14(7):e0219398. PMID:31276572
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 391.55 |
| Cas No. | 65646-68-6 |
| Formula | C26H33NO2 |
| Synonyms | 4-HPR; (4-Hydroxyphenyl)retinamide |
| Solubility | insoluble in H2O; ≥19.6 mg/mL in DMSO; ≥47.8 mg/mL in EtOH with gentle warming |
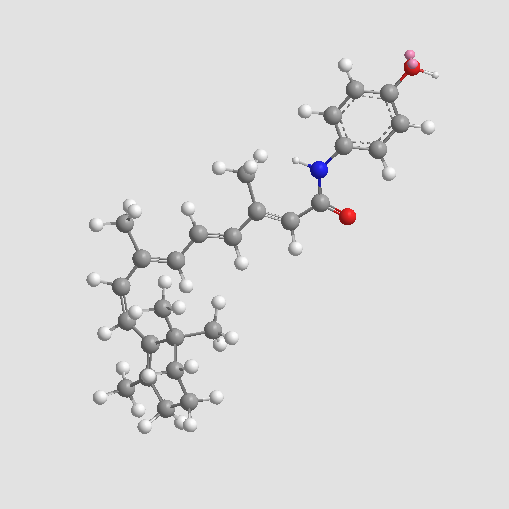
| Chemical Name | (2E,4E,6E,8E)-N-(4-hydroxyphenyl)-3,7-dimethyl-9-(2,6,6-trimethylcyclohexen-1-yl)nona-2,4,6,8-tetraenamide |
| Canonical SMILES | CC1(C)C(C=CC(C)=CC=CC(C)=CC(Nc(cc2)ccc2O)=O)=C(C)CCC1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1-3]: | |
|
Cell lines |
T-ALL cell lines, CCRF-CEM leukemia cells, CCRF-CEM and Jurkat cell, OVCAR-5 cell |
|
Preparation method |
The solubility of this compound in DMSO is > 19.6 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
>1 μM, 3 days |
|
Applications |
Fenretinide inhibited the growth of many tumor cells, including small-cell lung cancer, malignant hemopoietic cells, and breast cancer cells. The IC50 values of Fenretinide were 0.3 and 0.4 μM in 222 and UCI 101 ovarian cancer cell lines. Fenretinide showed antitumor activity in selected T-ALL cell lines. Fenretinide inhibited DES activity in CCRF-CEM leukemia cells in a dose and time dependent manner, leading to a concomitant increase of the endogenous cellular dhCer content. Fenretinide (3 μM) induced dhCer accumulation in both CCRF-CEM and Jurkat cells. Fenretinide (> 1 μM) inhibited OVCAR-5 cell proliferation and viability, with 70-90% growth inhibition at 10 μM. Fenretinide (1 μM) significantly inhibited OVCAR-5 invasion after 3 days preincubation. |
| Animal experiment [4,5]: | |
|
Animal models |
HFD-fed male C57Bl/6 mice, NOD/SCID mice |
|
Dosage form |
Intraperitoneal injection, 10 mg/kg |
|
Application |
Fenretinide (10 mg/kg, i.p.) selectively inhibited ceramide accumulation HFD-fed male C57Bl/6 mice. Fenretinide treatment improved glucose tolerance and insulin sensitivity. Addition of 25 mg/kg ketoconazole to Fenretinide in NOD/SCID mice increased 4-HPR plasma levels. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Apraiz, Aintzane., et al. Dihydroceramide accumulation and reactive oxygen species are distinct and nonessential events in 4-HPR-mediated leukemia cell death. Biochemistry and Cell Biology (2012), 90(2), 209-223. [2]. Golubkov V, et al. Action of fenretinide (4-HPR) on ovarian cancer and endothelial cells. Anticancer Res. 2005 Jan-Feb;25(1A):249-53. [3]. Anita L. Sabichi, Denver T. Hendricks, Mary A. Bober, Michael J. Birrer. Retinoic acid receptorβexpression and growthinhibition of gynecologic cancer cells by thesynthetic retinoidn-(4-hydroxyphenyl) retinamide. Journal of the National Cancer Institute. 1998, 90(8): 597-605. [4]. Bikman, Benjamin T., et al. Fenretinide Prevents Lipid-induced Insulin Resistance by Blocking Ceramide Biosynthesis. Journal of Biological Chemistry (2012), 287(21), 17426-17437. [5]. Cooper JP, et al. Fenretinide metabolism in humans and mice: utilizing pharmacological modulation of its metabolic pathway to increase systemic exposure. Br J Pharmacol. 2011 Jul;163(6):1263-75. |
|
| Description | Fenretinide is a synthetic retinoid derivative. | |||||
| Targets | RAR | |||||
| IC50 | ||||||
Quality Control & MSDS
- View current batch:
Chemical structure

Related Biological Data
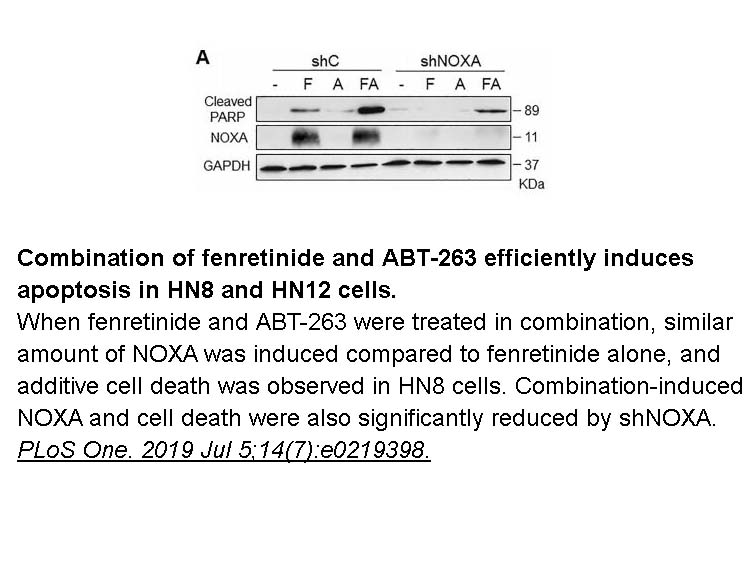
Related Biological Data