Entecavir
Entecavir ( CAS No. 142217-69-4) is a potent and selective inhibitor of HBV DNA polymerase (reverse transcriptase), whose core bioactivity is to inhibit replication of chronic hepatitis B virus (HBV). Its targets include the priming of HBV reverse transcriptase, and the synthesis steps of negative- and positive-strand DNA. It is effective against both wild-type HBV and lamivudine-resistant HBV (M204V/L180M mutations). In HepG2.2.15 cells, the EC?? for inhibition of HBV replication is 3.75 nM, and the EC?? for lamivudine-resistant strains increases by about 8-fold; no definitive MIC data are available. In vitro cell experiments are designed around the EC?? in the 0.1~10 nM range, while in animal experiments, rat/dog models use doses of 1~10 mg/kg/day. In a woodchuck chronic HBV infection model, oral administration can significantly reduce viral load and cccDNA levels. Clinically effective therapeutic concentrations correspond to oral doses of 0.5 mg/day (taken on an empty stomach) in nucleos(t)ide-na?ve adults, and 1 mg/day in lamivudine-resistant patients or those with decompensated liver disease. The steady-state peak plasma concentration is about 8.24 ng/mL. After 2 years of treatment, 81% of patients can achieve HBV DNA levels below 300 copies/mL, and the 5-year resistance rate in nucleos(t)ide-na?ve patients is only 0.9%. Development of resistance requires multiple mutations in HBV polymerase (e.g., T184G/S202I/M250V). Entecavir has a good safety profile, with occasional thrombocytopenia and lactic acidosis (high-risk populations require monitoring). It is indicated for patients with chronic HBV infection (with viral replication, elevated ALT, or histologically active liver disease) and for those with decompensated cirrhosis.
References:
[1] Opio CK, Lee WM, Kirkpatrick P. Entecavir. Nat Rev Drug Discov. 2005 Jul;4(7):535-6. doi: 10.1038/nrd1780. PMID: 16075486.
[2] Zoulim F. Entecavir: a new treatment option for chronic hepatitis B. J Clin Virol. 2006 May;36(1):8-12. doi: 10.1016/j.jcv.2006.01.010. Epub 2006 Mar 3. PMID: 16515882.
[3] Shepherd J, Gospodarevskaya E, Frampton G, Cooper K. Entecavir for the treatment of chronic hepatitis B infection. Health Technol Assess. 2009 Oct;13 Suppl 3:31-6. doi: 10.3310/hta13suppl3/05. PMID: 19846026.
[4] Keating GM. Entecavir: a review of its use in the treatment of chronic hepatitis B in patients with decompensated liver disease. Drugs. 2011 Dec 24;71(18):2511-29. doi: 10.2165/11208510-000000000-00000. PMID: 22141390.
[5] Yu Y, Feng H. Entecavir-associated thrombocytopenia. Int J Immunopathol Pharmacol. 2021 Jan-Dec;35:20587384211059676. doi: 10.1177/20587384211059676. PMID: 34823407; PMCID: PMC8674478.
[6] Li P, Wang Y, Yu J, Yu J, Tao Q, Zhang J, Lau WY, Zhou W, Huang G. Tenofovir vs Entecavir Among Patients With HBV-Related HCC After Resection. JAMA Netw Open. 2023 Oct 2;6(10):e2340353. doi: 10.1001/jamanetworkopen.2023.40353. PMID: 37906195; PMCID: PMC10618847.
[7] Henriquez-Camacho C, Hijas-Gomez AI, Risco Risco C, Ruiz Lapuente MA, Escudero-Sanchez R, Cuerda VM. Lamivudine and Entecavir for Acute Hepatitis B: A Systematic Review and Meta-Analysis. Viruses. 2023 Nov 10;15(11):2241. doi: 10.3390/v15112241. PMID: 38005918; PMCID: PMC10675181.
[8] Lumley SF, Delphin M, Mokaya JF, Tan CCS, Martyn E, Anderson M, Li KC, Waddilove E, Sukali G, Downs LO, Said K, Okanda D, Campbell C, Harriss E, Shimakawa Y, Matthews PC. A systematic review and meta-analysis of the risk of hepatitis B virus (HBV) resistance in people treated with entecavir or tenofovir. J Clin Virol. 2024 Oct;174:105711. doi: 10.1016/j.jcv.2024.105711. Epub 2024 Jun 28. Erratum in: J Clin Virol. 2024 Oct;174:105716. doi: 10.1016/j.jcv.2024.105716. PMID: 38991458.
| Physical Appearance | A solid |
| Storage | -20°C |
| M.Wt | 277.28 |
| Cas No. | 142217-69-4 |
| Formula | C12H15N5O3 |
| Synonyms | BMS200475;SQ34676 |
| Solubility | ≥37.3 mg/mL in DMSO; insoluble in EtOH; insoluble in H2O |
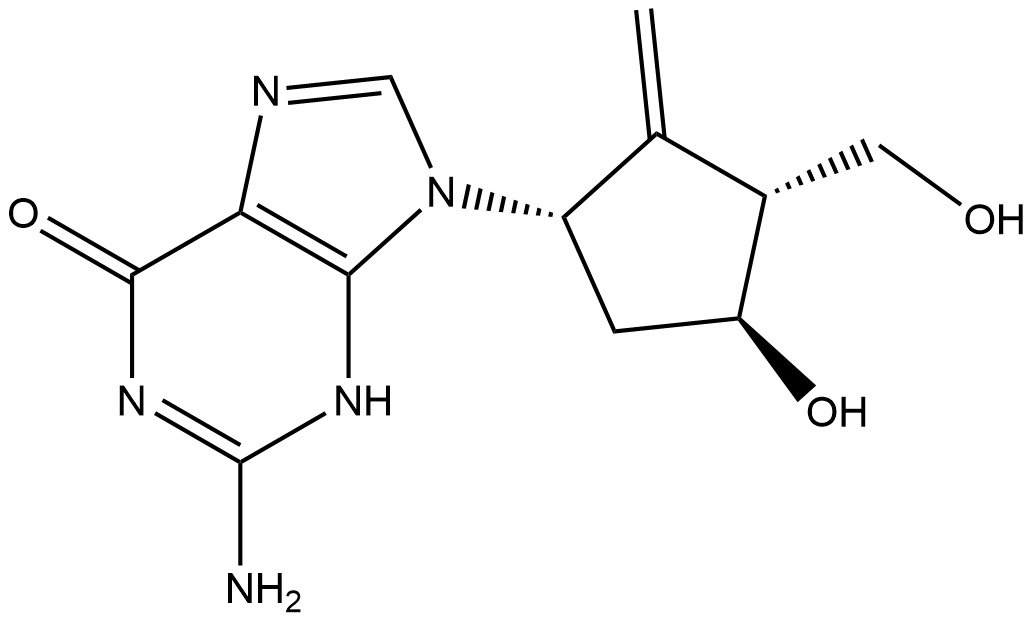
| Chemical Name | 2-amino-9-((1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl)-3,9-dihydro-6H-purin-6-one |
| Canonical SMILES | C=C1[C@H](CO)[C@@H](O)C[C@@H]1N(C=N2)C3=C2C(N=C(N)N3)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure