Eltrombopag Olamine
Description: IC50 Value: 0.27 uM (EC50 in murine BAF3 cells) [1] Eltrombopag is a new, orally active thrombopoietin-receptor (c-mpl) agonist that stimulates thrombopoiesis. Potential advantages of eltrombopag may include a sustained platelet response and a good tolerability profile. in vitro: Eltrombopag demonstrated a half maximal effective concentration (EC50) of 0.27 uM in murine BAF3 cells transfected with the luciferase reporter gene under direction of the STAT-activated IRF-1 promoter and human TpoR (BAF3/IRF-1/hTpoR) [1]. Eltrombopag stimulates the growth of TPO-dependent cell lines via JAK2 and STAT signaling pathways and stimulates isolated human CD34+ cells to become megakaryocytes and produce platelets [2]. in vivo: Twelve weeks of antiviral therapy, with concurrent receipt of eltrombopag or placebo, were completed by 36%, 53%, and 65% of patients receiving 30 mg, 50 mg, and 75 mg of eltrombopag, respectively, and by 6% of patients in the placebo group [3]. Eltrombopag was administered as once-daily oral capsules for 10 days at doses of 5, 10, 25, 30, 50, and 75 mg. The pharmacokinetics of eltrombopag was dose dependent and linear, and eltrombopag increased platelet counts in a dose-dependent manner [4]. Toxicity: There were no apparent differences in the incidence or severity of adverse events in subjects receiving active or placebo study medication [4]. Long-term treatment with eltrombopag was generally safe, well tolerated, and effective in maintaining platelet counts in the desired range [5]. Clinical trial: Eltrombopag In Cord Blood Or Haploidentical Bone Marrow Transplantation. Phase 2
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 564.63 |
| Cas No. | 496775-62-3 |
| Formula | C29H36N6O6 |
| Synonyms | Eltrombopag diethanolamine salt;Promacta;Revolade;SB-497115GR |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥14.12 mg/mL in DMSO with gentle warming |
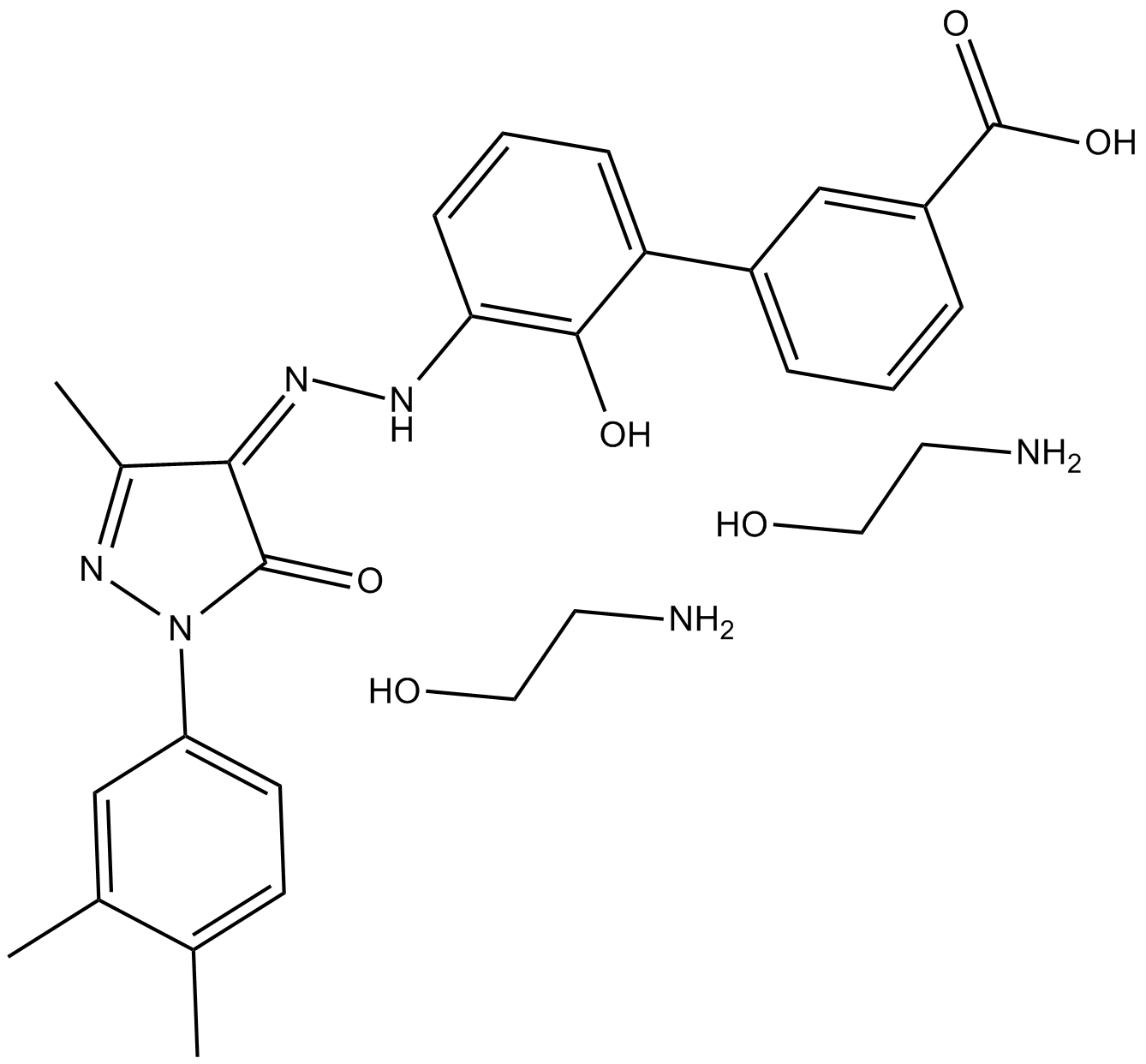
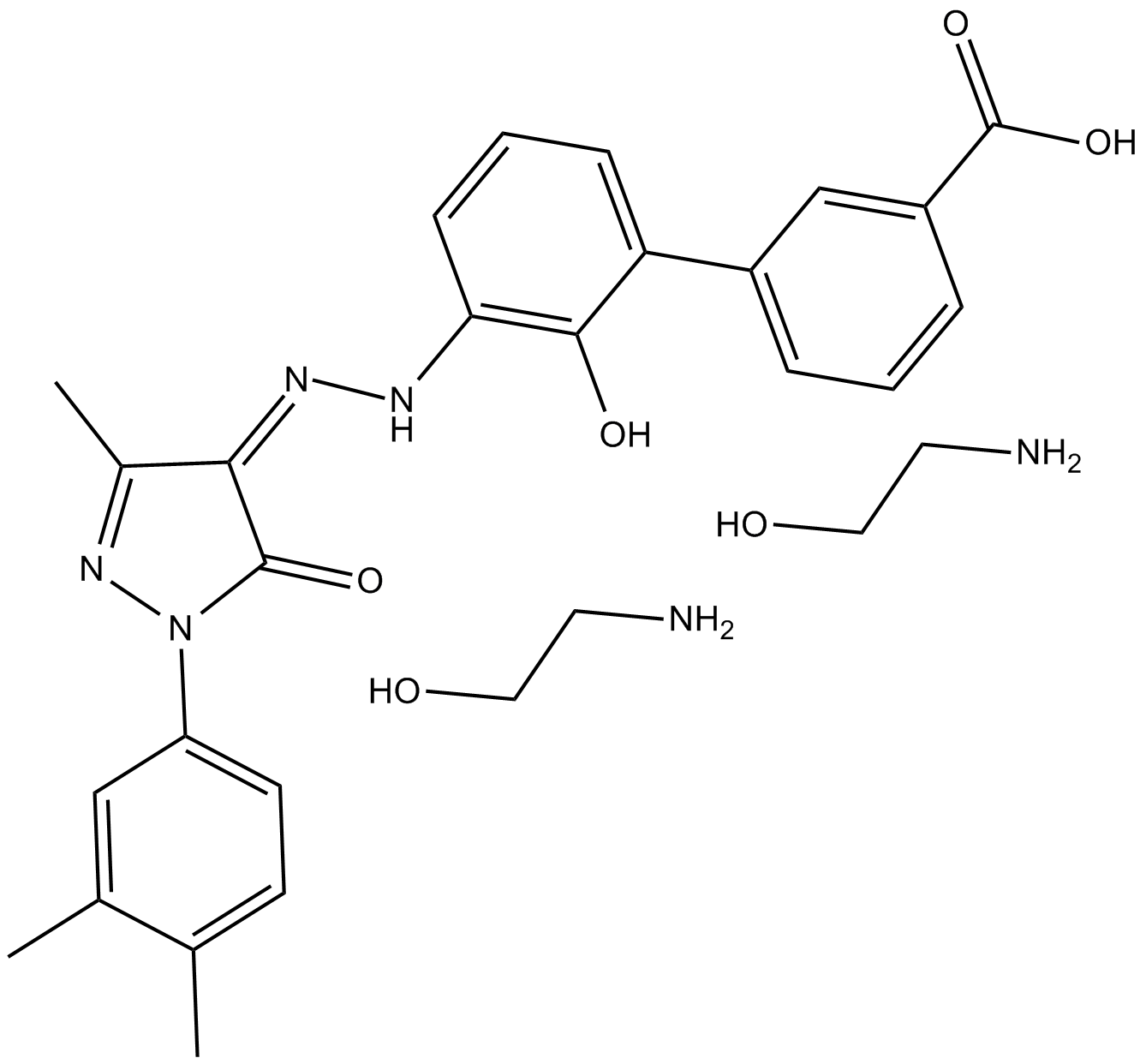
| Chemical Name | 2-aminoethanol;3-[(5E)-5-[[2-(3,4-dimethylphenyl)-5-methyl-3-oxo-1H-pyrazol-4-yl]hydrazinylidene]-6-oxocyclohexa-1,3-dien-1-yl]benzoic acid |
| Canonical SMILES | CC1=C(C=C(C=C1)N2C(=O)C(=C(N2)C)NN=C3C=CC=C(C3=O)C4=CC(=CC=C4)C(=O)O)C.C(CO)N.C(CO)N |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure