Clodronate (sodium salt hydrate)
Clodronate (sodium salt hydrate) (CAS No.: 88416-50-6) is a first-generation, non-nitrogenous bisphosphonate widely used in biomedical research for its anti-resorptive, anti-inflammatory, and analgesic properties, particularly in the context of bone metabolism and neuroinflammatory signaling. As a high-affinity binder of hydroxyapatite, it inhibits crystal growth and suppresses bone resorption through actions on osteoclasts and osteoblasts, while intracellular metabolism generates a non-hydrolyzable ATP analog that induces cytotoxic effects in osteoclasts and macrophage-lineage cells. In addition to its skeletal effects, clodronate functions as a selective, potent, and reversible inhibitor of the vesicular nucleotide transporter, competitively interacting with chloride-binding sites to block vesicular ATP release from neurons, thereby modulating purinergic signaling pathways implicated in chronic neuropathic and inflammatory pain. Its activity is typically observed in the nanomolar to low micromolar range in vitro, depending on the assay system and biological endpoint. Clodronate is also frequently employed in liposome-encapsulated formulations to achieve selective depletion of macrophages or macrophage-like cells in experimental models, enabling studies of immune cell function, tumor microenvironment dynamics, and inflammation. In vitro and in vivo applications commonly include osteoporosis models, pain signaling studies, and immune modulation assays, with experimental concentrations or dosing regimens adjusted according to specific study designs and research objectives.
| Physical Appearance | A solid |
| Storage | -20°C |
| M.Wt | 360.92 |
| Cas No. | 88416-50-6 |
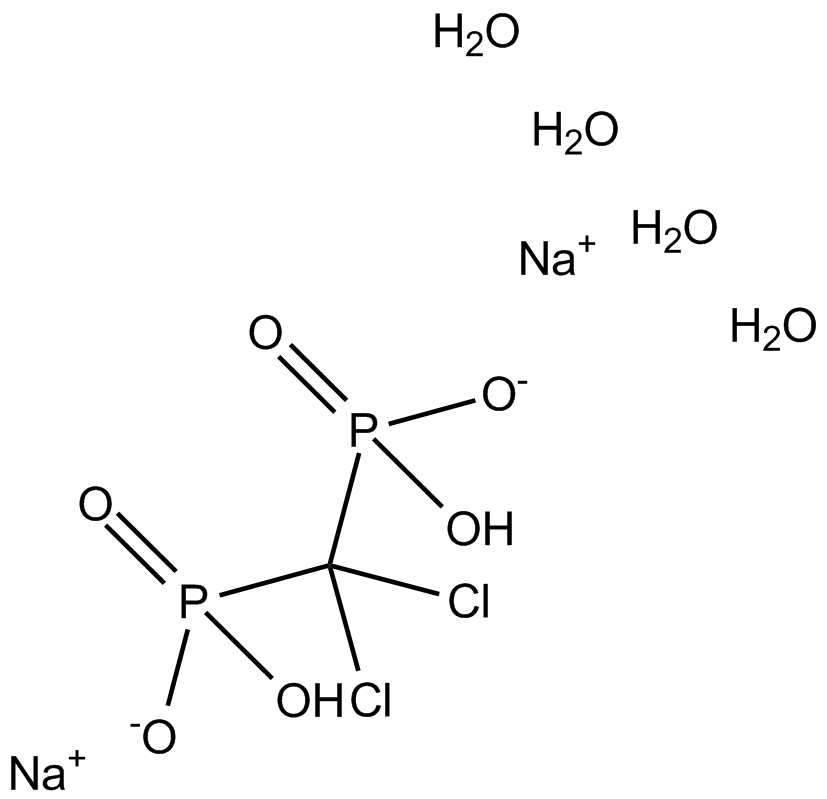
| Formula | CH10Cl2Na2O10P2 |
| Synonyms | Clodronic acid |
| Solubility | insoluble in EtOH; insoluble in DMSO; ≥26.5 mg/mL in H2O |
| Chemical Name | sodium (dichloromethylene)bis(hydrogenphosphonate) tetrahydrate |
| Canonical SMILES | O=C1OC2(C3=CC=C(C=C3OC4=C2C=CC(OC(C)=O)=C4)OC(C)=O)C5=C1C=C(C(O)=O)C=C5.O=C6OC7(C8=CC=C(C=C8OC9=C7C=CC(OC(C)=O)=C9)OC(C)=O)C%10=C6C=CC(C(O)=O)=C%10 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |