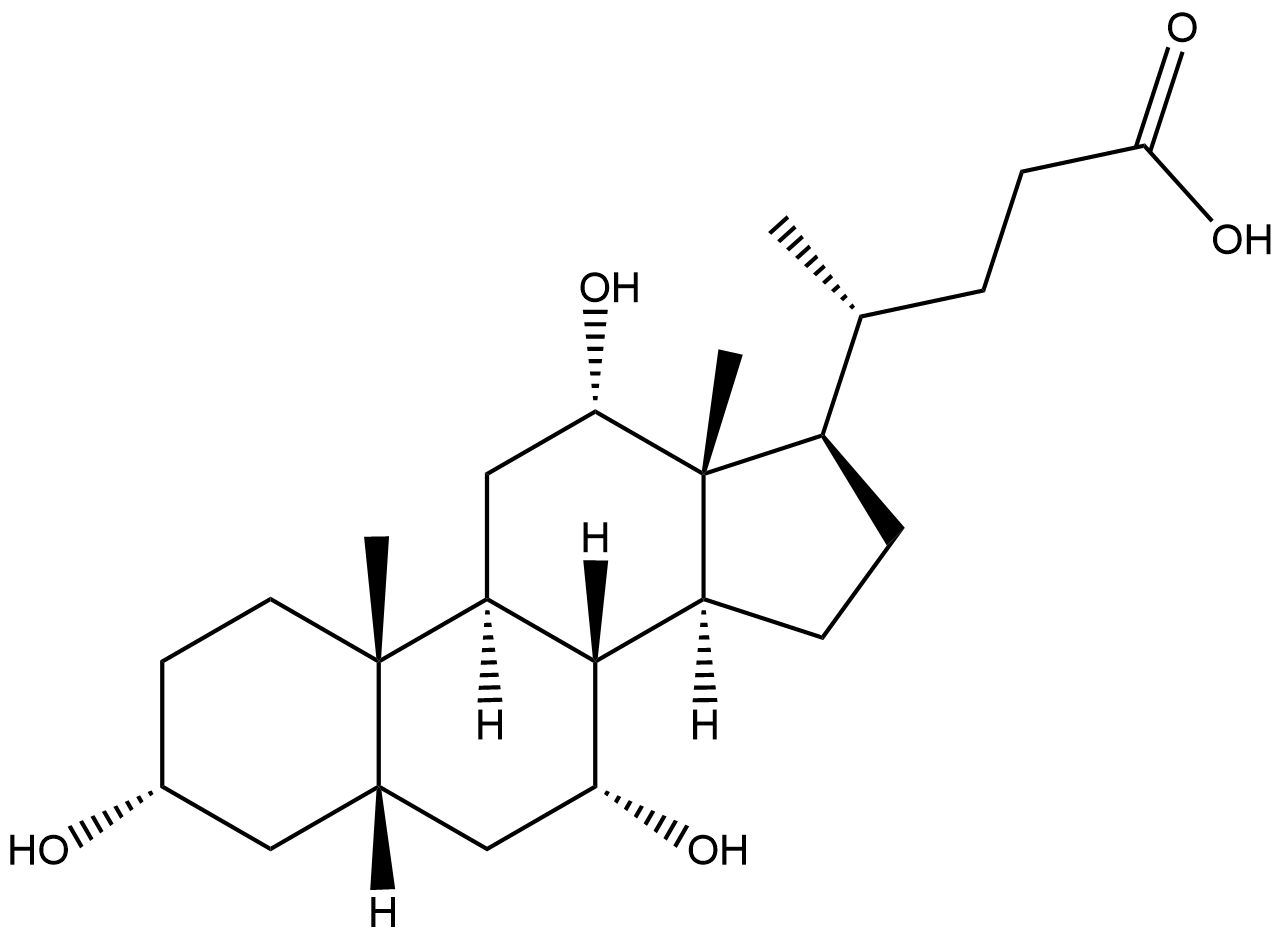
Allocholic acid
Allocholic acid is a bile acid found in vertebrates. Its molecular structure is an isomer of bile acid. Due to the isomerization of the hydroxyl group position on the steroid ring, it forms a unique configuration and is also the conjugate acid of isocholic acid. Allocholic acid has a relatively low content under normal physiological conditions, but it is re-expressed during liver regeneration and carcinogenesis, suggesting that it may be involved in the metabolic regulatory network of tissue repair and tumorigenesis. As a specific stimulant of the mature olfactory system, Allocholic acid can efficiently activate olfactory receptors and mediate olfactory signal transduction. Its mechanism of action may be related to the interaction with G protein-coupled receptors in the nasal mucosa. From the perspective of metabolite distribution, it possesses the attributes of metabolites in Marine organisms, rats and humans. It may not only serve as a secondary metabolite in Marine organisms but also play a role in the hepatointestinal circulation of mammals.
| Physical Appearance | A solid |
| Storage | Store at -20°C for 3 years. |
| M.Wt | 408.57 |
| Cas No. | 2464-18-8 |
| Formula | C24H40O5 |
| Solubility | ≥53.5 mg/mL in DMSO; ≥52.2 mg/mL in EtOH; insoluble in H2O |
| Chemical Name | (R)-4-((3R,5R,7R,8R,9S,10S,12S,13R,14S,17R)-3,7,12-trihydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)pentanoic acid |
| Canonical SMILES | O[C@H]1[C@]2([C@]3([C@@](C)([C@@]([C@@H](CCC(O)=O)C)(CC3)[H])[C@@H](O)C[C@@]2([C@]4(C)[C@@](C1)(C[C@H](O)CC4)[H])[H])[H])[H] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure